Have you ever seen an ice cube disappear into water or watched cookie dough turn into cookies? This is an important scientific idea: matter can change when it gets warmer or cooler. We can often see these changes with our eyes. A material may melt, freeze, get harder, get softer, change shape, or even turn into something new.
Everything around us is made of matter. Matter can be a solid, a liquid, or a gas. A solid keeps its shape, like a toy block or an ice cube. A liquid flows and takes the shape of its cup, like water or milk. A gas spreads out, like the air in a room or steam above hot soup.
When matter changes, we can look for clues. We may notice a different shape, a different feel, a different smell, or a different color. Some changes happen fast, and some happen slowly. Heating and cooling are two important ways to make these changes happen.
Heating means adding heat to something. Cooling means removing heat from something. A reversible change can be changed back. An irreversible change cannot be changed back easily or at all.
Scientists pay attention to what they can observe. That means what they can see, touch, smell, or hear. If butter gets soft on warm toast, that is an observable change. If water in a cup turns into ice in the freezer, that is an observable change too.
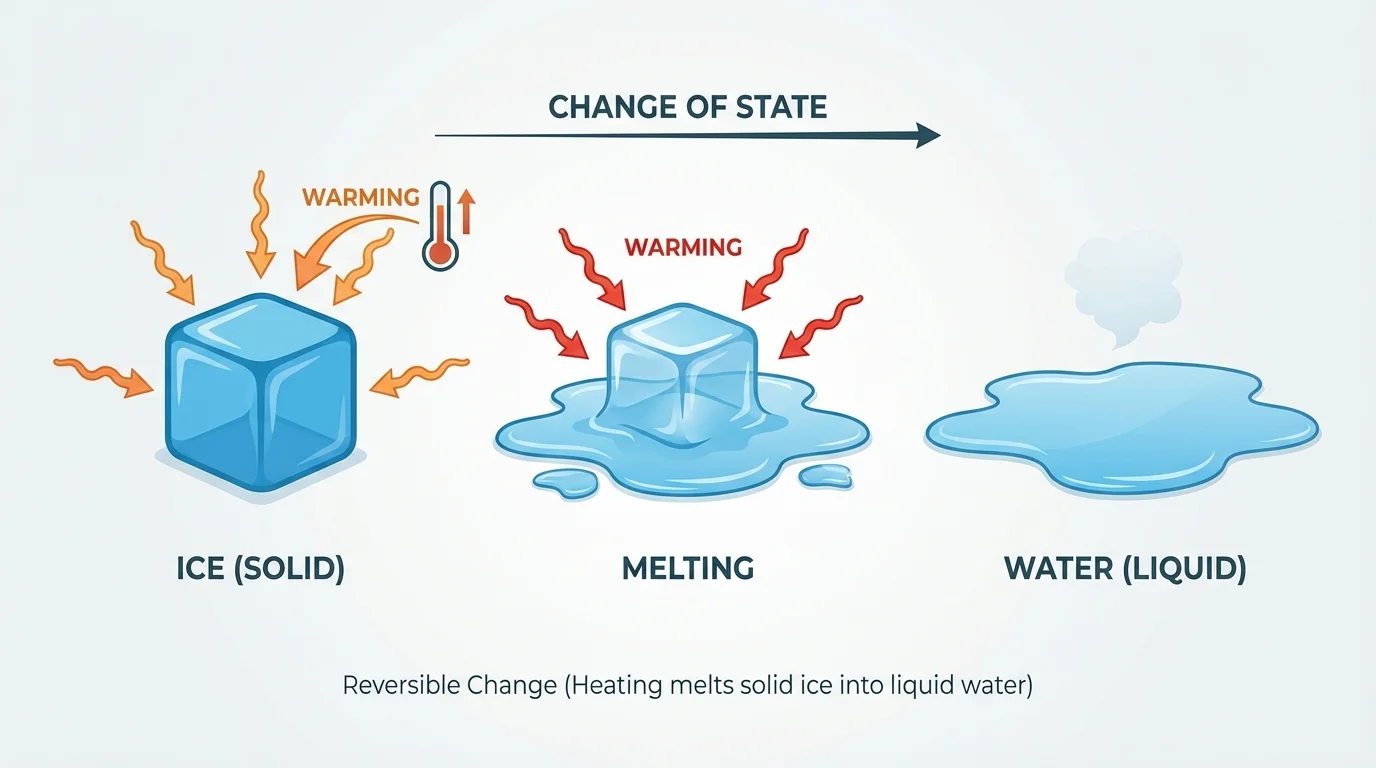
[Figure 1] Melting happens when heat makes some solids turn into liquids. Ice cream left in the sun melts. Butter in a warm pan melts. Chocolate in your hand can even start to melt a little.
Heating can also make things softer. A crayon left in a hot car may bend more easily. Cheese on pizza becomes soft and stretchy in the oven. Water can get so warm that some of it becomes water vapor, which is a gas.
Sometimes heating makes a very different kind of change. An egg in a pan changes from runny to firm. Cake batter in an oven becomes cake. Bread in a toaster can turn into toast. These are changes we can observe, but they do not go back to the way they were before.
Heating can do different jobs
Heat does not always cause the same kind of change. Sometimes it only changes the form of a substance, like ice becoming water. Other times it makes a new material with new properties, like batter becoming a cupcake. Looking closely helps us decide what kind of change happened.
We use heating every day. Grown-ups cook food to make it safe and tasty. Warm sunlight dries puddles after rain. Warm air can soften wax. When we think about heat, we think about what changes we can see.
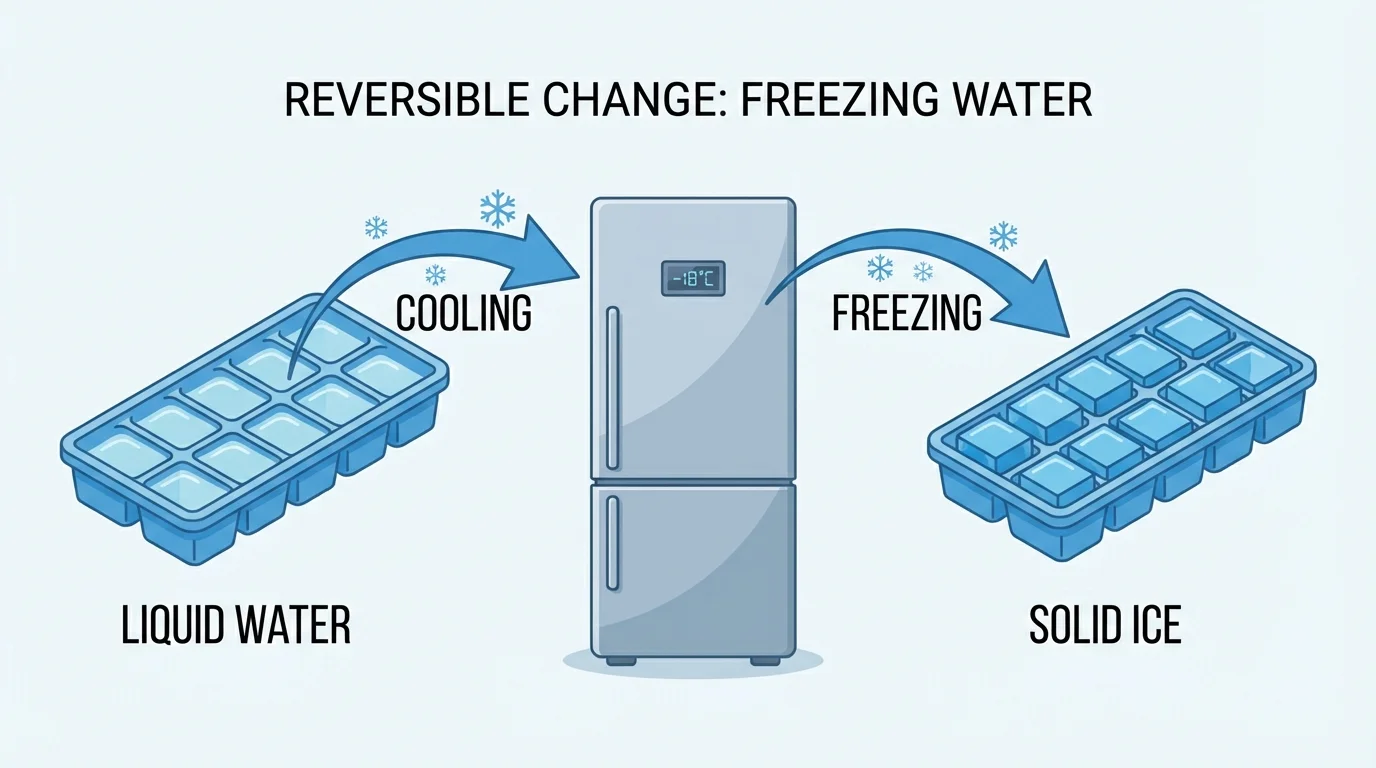
[Figure 2] Freezing happens when cooling makes some liquids turn into solids. Water in an ice tray freezes in the freezer. Melted juice can freeze into popsicles. Melted wax can cool and become hard again.
Cooling can make things harder or less bendy. Warm butter becomes firmer in the refrigerator. Melted chocolate can cool into a solid candy shape. Cooling can also make water droplets appear on the outside of a cold cup. That happens because water in the air turns into tiny liquid drops.
If you have ever touched a frozen treat, you know cooling changes how matter feels. It may feel colder, harder, and more solid. These are clues that a change happened.
Clouds and rain are connected to cooling. When water vapor high in the sky cools, it can turn into tiny drops of liquid water.
Cooling is useful in real life. We cool food in a refrigerator so it stays fresh longer. We freeze ice packs to help keep lunches cold. We cool water to make ice for drinks on a hot day.
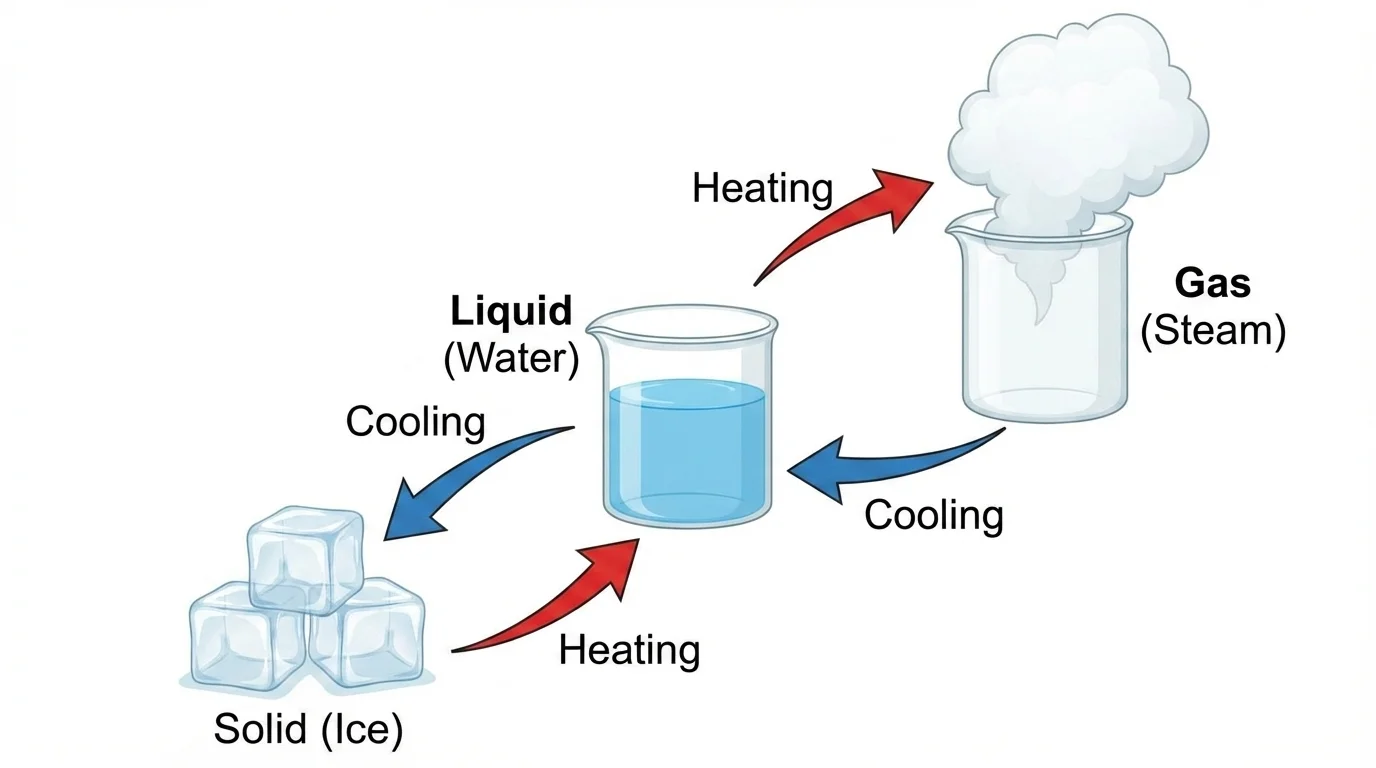
A reversible change is a change that can go back, and water helps us see this clearly. Ice can melt into liquid water. Liquid water can freeze back into ice. If water gets very warm, it can become water vapor, and when that vapor cools, it can turn back into liquid water.
[Figure 3] These changes are exciting because the same substance is still there. With water, the substance is still \(H_2O\). It may look different, but it is still water. Heating and cooling change its form.
Another reversible change happens with wax. Candle wax can melt when heated and harden when cooled. That is like the water changes shown earlier: the material changes form, but it can change back.
Everyday reversible change
Step 1: Put water in a cup and place it in the freezer.
Step 2: The liquid water freezes and becomes solid ice.
Step 3: Take the ice out and leave it in a warm place.
Step 4: The ice melts and becomes liquid water again.
This is a change that can go back and forth.
Not every reversible change happens quickly. Some take time. But if the substance can return to the way it was before, we call the change reversible.
An irreversible change is a change that does not go back. When an egg is cooked, it cannot become a raw egg again. When batter bakes into bread or a muffin, it cannot turn back into wet batter.
Burning is another example. A piece of toast that burns cannot become plain bread again. Wood that burns turns into ash and smoke. These changes make new materials, so they are not easy to reverse.
You already know that matter can be a solid, liquid, or gas. Heating and cooling can move matter from one form to another, but sometimes they also make a completely new kind of material.
We can often tell an irreversible change happened because the substance looks and behaves very differently. It may have a new smell, a new color, or a new texture. Cooked food is often firmer or drier than it was before heating.
Science is happening in kitchens, playgrounds, and backyards all the time. Freezers make ice cubes. Ovens bake bread. The sun warms puddles. Hot soup gives off steam. Heating and cooling help us prepare food, stay comfortable, and understand the world.
It is also important to be safe. Very hot objects can burn skin, and very cold objects can hurt too. Grown-ups use ovens, stoves, and kettles carefully. Scientists observe changes, but they also stay safe while they learn.
When you look at changes in matter, ask two smart questions: What did I observe? and Can the change go back? Those questions help us tell whether heating or cooling caused a reversible or irreversible change.