Have you ever watched ice melt, water boil, or vinegar fizz with baking soda and wondered whether some of the matter disappears? It can look that way. A solid can turn into a liquid. A liquid can seem to vanish into the air. Two powders or liquids can bubble and foam. But scientists have found something powerful: no matter what kind of change happens, the total amount of matter stays the same if all of it is kept in the system and measured carefully.
This idea is called conservation of matter. It means matter is not created from nothing and does not vanish into nothing during ordinary changes. Sometimes matter changes shape, size, state, or even forms new substances with different properties. Even then, the total amount is still there. To prove that, scientists do not just guess. They measure, record data, and make graphs.
Think about cooking popcorn. The kernels heat up and change. Think about chocolate melting in your hand. Think about a fizzy tablet dropped into water. In each case, matter is changing in a way you can observe. Some changes are physical changes, where the same substance is still present but in a different form. Some are chemical reactions, where new substances form and may have new smells, colors, temperatures, or gases.
No matter which kind of change happens, science asks the same question: if we measure everything before and after, do we get the same total amount of matter? The answer is yes. That is a big idea because it helps scientists understand what is happening, design fair experiments, and trust their evidence instead of only their eyes.
Conservation of matter means the total amount of matter stays the same during heating, cooling, and mixing, even if the matter changes form or becomes new substances.
Physical change is a change in size, shape, or state that does not make a new substance.
Chemical reaction is a change in which substances mix and form one or more new substances with different properties.
Sometimes your eyes can fool you. Steam rising from hot water may look like the water is gone. Bubbles from a reaction may make it seem like matter appeared from nowhere. As [Figure 1] shows, a careful scientist knows that visible changes are clues, but measurements are better evidence.
To test conservation of matter, scientists use a balance to measure the mass of a system before and after a change. The key idea is to measure the whole setup, not just one part of it. For example, if water is heated in a sealed container, the scientist measures the container and everything inside it before heating and again after heating.
If the measurements are the same, that is evidence that the total amount of matter is conserved. Suppose a sealed container and its contents measure \(120 \textrm{ g}\) before heating. After heating, the container and contents still measure \(120 \textrm{ g}\). The substances may look different inside, but the total amount has not changed.

Scientists often organize their measurements in a table first. Then they may make a graph to compare the starting and ending measurements. This makes patterns easier to spot. If the bars on a graph are the same height before and after each change, that is visual evidence that matter is conserved.
You already know that solids, liquids, and gases are all forms of matter. Matter takes up space and can be measured. When matter changes from one state to another, it is still matter.
A good measurement depends on careful procedures. The same balance should be used if possible. The container should not spill. If a lid is part of the setup, it must stay with the setup the whole time. Scientists repeat measurements because one result is helpful, but repeated results are stronger evidence.
There are many ways matter can change. One common change is heating. Heating can melt butter, warm soup, or change liquid water to water vapor. Cooling can freeze juice into ice pops or make steam turn back into liquid drops. These are changes of state. The substance is still the same substance. Water stays \(H_2O\) whether it is ice, liquid water, or water vapor.
Another common change is dissolving. When salt is stirred into water, the salt seems to disappear, but it has not vanished. Its particles spread through the water. If the saltwater is measured in a closed container, the total amount is still the same as the amount of water plus the amount of salt before mixing.
Some changes make new substances. These are chemical reactions. For example, baking soda mixed with vinegar can form bubbles of gas. The materials after the reaction are not exactly the same as the materials before. New substances have formed. Even so, if the reaction happens in a closed system, the total amount of matter is still conserved.
Different kinds of changes, same total matter
Physical changes and chemical reactions do not look the same, but both follow the same rule. In a physical change, the particles are rearranged in state or position. In a chemical reaction, atoms are rearranged into new substances. In either case, the total amount of matter remains constant when all of the matter is included in the measurement.
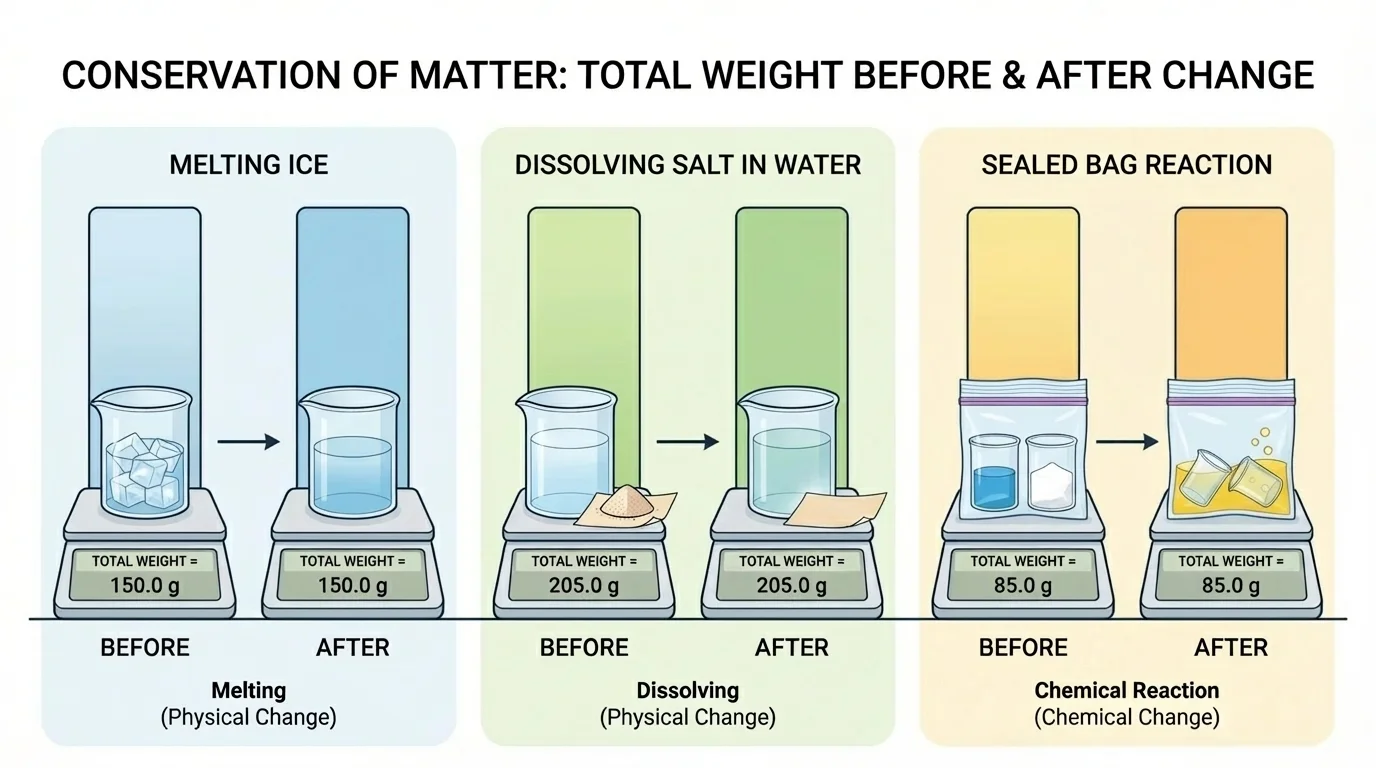
As [Figure 2] helps illustrate, the type of change does not matter when we talk about the total amount of matter. Melting, freezing, evaporating, dissolving, and reacting may all look different, but the same scientific rule applies.
Sometimes people think matter was lost because they did not measure all of it. This often happens when a gas escapes into the air. If vinegar and baking soda react in an open cup, bubbles rise and some gas leaves the cup. If you measure only the cup and what remains in it, the amount may seem smaller afterward. But the matter did not disappear; some of it moved into the air.
A closed system is a setup where matter cannot escape. A sealed plastic bag, a jar with a tight lid, or another closed container helps scientists measure all the matter before and after a change. In a closed system, gases stay trapped inside, so the total amount can be measured more accurately.

An open system is not wrong to observe, but it can be misleading if you want to test conservation of matter. If some matter leaves the container, then you are not measuring the whole system anymore. That is why scientists are careful about what counts as the system in an investigation.
Later, when you look at reaction data, remember this lesson: if gas escapes, the measurement of the remaining container may change even though the total matter in the complete system has not changed.
In [Figure 3], scientists compare data from many trials and examine pairs of bars that show the amount before and after several different changes. A graph can make the evidence easier to understand, and equal bar heights support the idea that matter is conserved.
Suppose students measure three setups in closed containers. Setup A contains ice that melts. Setup B contains water and salt that are mixed. Setup C contains vinegar and baking soda reacting in a sealed bag. Their data might look like this:
| Setup | Change observed | Before | After |
|---|---|---|---|
| A | Ice melts | \(85 \textrm{ g}\) | \(85 \textrm{ g}\) |
| B | Salt dissolves in water | \(92 \textrm{ g}\) | \(92 \textrm{ g}\) |
| C | Vinegar and baking soda react | \(140 \textrm{ g}\) | \(140 \textrm{ g}\) |
In each case, the before and after measurements match. We can describe that relationship with a simple sentence in math form: \[\textrm{total before} = \textrm{total after}\] If Setup B starts with \(60 \textrm{ g}\) of water and \(32 \textrm{ g}\) of salt, then the total before mixing is \(60 + 32 = 92\), so the sealed container should still measure \(92 \textrm{ g}\) after the salt dissolves.
Graphs are useful because they let us compare several changes at once. Even though melting, dissolving, and reacting are different processes, the graph pattern is the same. That repeated pattern is strong evidence.
Using measurements as evidence
A sealed bag contains vinegar and baking soda separated at first. The bag and contents are measured before the substances mix, and then measured again after the reaction finishes.
Step 1: Record the first measurement.
The sealed bag and contents measure \(150 \textrm{ g}\) before mixing.
Step 2: Let the substances mix and react.
Bubbles form, which shows a gas is being produced. Because the bag is sealed, the gas stays in the system.
Step 3: Measure the whole bag again.
After the reaction, the sealed bag and contents measure \(150 \textrm{ g}\).
Step 4: Use the data to make a claim.
Since \(150 = 150\), the measurements support the idea that the total amount of matter was conserved.
The bubbling may make the bag look very different, but the evidence shows that the total matter stays the same.
When students build a graph from these data, the "before" bar and the "after" bar for the sealed bag are equal. That matches the pattern we saw in [Figure 3].
Science becomes clearer when you connect it to familiar experiences. Here are several examples of matter being conserved during different kinds of changes.
Melting ice: Put ice cubes in a sealed container and measure the container. After the ice melts, measure it again. The ice changed to liquid water, but the total amount is unchanged.
Chocolate melting: A piece of chocolate softens and melts when warmed. It may spread out and look different, but the same chocolate matter is still there.
Salt dissolving in soup or water: Salt may seem to disappear, but it has mixed throughout the liquid. The total amount remains the same if nothing spills or evaporates out of the measured system.
Fizzing reactions: A bath bomb or baking soda and vinegar can produce bubbles. Those bubbles are matter in the form of gas. If the gas escapes, the remaining container may seem lighter, but the complete system still contains the same total matter.
Some of the most surprising evidence for conservation of matter comes from gases. Because gases spread out and are often invisible, people may think they are "nothing," but gases are matter and must be counted too.
Another useful example is mixing two clear liquids that form a cloudy solid. The appearance changes a lot, which suggests a chemical reaction. Even then, if the setup is closed and measured carefully, the total amount remains the same.
Real experiments are not always perfect. A small difference in measurements does not automatically mean matter was created or destroyed. It may mean there was measurement error. That means the procedure or tool was not perfectly exact.
For example, a drop of liquid could spill. A lid might not be sealed completely. A balance might not be set to zero correctly before measuring. Warm air currents can even affect some very sensitive balances. Scientists think carefully about these possibilities before drawing conclusions.
If one trial gives \(100 \textrm{ g}\) before and \(99 \textrm{ g}\) after, that does not prove matter was destroyed. It may mean some gas escaped, a tiny amount spilled, or the measurement was off by \(1 \textrm{ g}\). Scientists look for repeated, careful results.
Evidence is stronger when the test is fair
A fair test changes one main thing at a time while keeping other conditions as similar as possible. In conservation of matter investigations, students should measure the entire system, use the same equipment, avoid spills, and record data clearly. Good evidence comes from careful methods.
This is also why graphs are helpful. If several careful trials show nearly equal before-and-after measurements, the pattern matters more than one imperfect result. Science relies on evidence collected in a thoughtful way.
This idea is important far beyond the classroom. Cooks measure ingredients and expect the matter to still be present after mixing and heating, even if the food changes texture, color, or smell. Engineers and factory workers track materials as products are made. Recycling centers sort and process matter instead of making it disappear. Environmental scientists study where matter goes in air, water, and soil.
Doctors, pharmacists, and food scientists also depend on careful measurements. If a medicine is mixed into a liquid, the medicine has not vanished; it is spread through the mixture. If a carbonated drink loses bubbles after being opened, gas has moved from the drink into the air. Understanding where the matter goes helps people solve real problems.
The same rule helps explain larger systems in nature. Water changes from ice to liquid to vapor and back again. Carbon moves through living things, air, and oceans. Matter travels and changes, but it is conserved.
"In science, careful measurements can reveal what our eyes alone might miss."
When you examine any change now, ask two questions: What changed, and how can I measure all the matter before and after? That scientific habit turns an interesting observation into evidence.