A drop of water, a grain of salt, the graphite in a pencil core, and the glass in a phone screen seem completely different. Yet all of them are built from tiny particles too small to see directly. That is one of the most powerful ideas in science: huge, visible objects can be explained by the way invisible atoms are arranged. Scientists use models to make those arrangements easier to picture, test, and compare.
Matter is anything that has mass and takes up space. Everything around you is matter: air, metal, food, water, plastic, and rock. Scientists explain matter by saying it is made of atoms. Because atoms are much too small to see with ordinary eyes, scientists build models. A model is not the actual thing. Instead, it is a useful representation that helps us understand what the real thing is like.
For example, a globe is a model of Earth. It shows shape and location, but it does not include every tree, road, or building. In the same way, an atomic model shows how atoms are put together in a substance, even though the atoms in the model are often drawn much larger and farther apart than they really are.
Model is a simplified representation of an object, system, or idea that helps people explain and predict what happens.
Atom is the tiny building block of matter that makes up elements and substances.
Molecule is a group of atoms joined together as a small unit.
Extended structure is a substance made of a repeating arrangement of atoms or particles that continues in many directions, rather than existing as one small separate molecule.
When scientists make models of substances, they ask questions such as: Which atoms are present? How many of each kind are there? Are they arranged as small separate groups, or in a repeating pattern? The answers help explain why substances have different properties.
An element is a pure substance made of only one kind of atom. Oxygen, carbon, hydrogen, and iron are examples of elements. Each element has its own symbol, such as \(\textrm{O}\), \(\textrm{C}\), \(\textrm{H}\), and \(\textrm{Fe}\).
A single atom is one particle of an element. But atoms often combine with other atoms. When atoms join in small groups, they can form a molecule. Some molecules contain only one element, such as oxygen gas, \(\textrm{O}_2\), which has two oxygen atoms. Other molecules contain more than one element, such as water, \(\textrm{H}_2\textrm{O}\), which has hydrogen and oxygen atoms.
It is important to separate these ideas clearly. An element tells you the kind of atom. A molecule tells you that atoms are grouped together in a small unit. Not every substance is made of separate molecules, though. Some substances are built as giant repeating structures instead.
From earlier studies of matter, you may remember that substances have properties such as hardness, melting point, and conductivity. Those properties do not come from size alone. They are connected to the kinds of atoms present and how those atoms are arranged.
Atoms are often represented in diagrams as circles or spheres. Different colors are commonly used for different elements, but the color is just part of the model. Real atoms are not tiny colored balls. The color choice simply helps us keep track of which atom is which.
A chemical formula is a compact model. It tells the kinds of atoms in a substance and how many of each are present in one molecule or repeating unit. In middle school, one of the most useful skills is learning how to read that information correctly.
In \(\textrm{H}_2\textrm{O}\), the small \(2\) means there are two hydrogen atoms and one oxygen atom. In \(\textrm{CO}_2\), there is one carbon atom and two oxygen atoms. If an element symbol has no small number after it, that means there is just one atom of that element in the formula.
Here are some examples:
| Formula | Atoms Present | What It Represents |
|---|---|---|
| \(\textrm{O}_2\) | \(2\) oxygen atoms | One molecule of oxygen |
| \(\textrm{H}_2\textrm{O}\) | \(2\) hydrogen, \(1\) oxygen | A water molecule |
| \(\textrm{CO}_2\) | \(1\) carbon, \(2\) oxygen | A carbon dioxide molecule |
| \(\textrm{CH}_4\) | \(1\) carbon, \(4\) hydrogen | A methane molecule |
| \(\textrm{NaCl}\) | \(1\) sodium, \(1\) chlorine | The repeating ratio in table salt |
Table 1. Chemical formulas can be read as models showing the kinds and numbers of atoms, or in some cases the repeating ratio of atoms in a larger structure.
Notice something important about \(\textrm{NaCl}\). For water or carbon dioxide, the formula describes one small molecule. For table salt, the formula tells the ratio of sodium atoms to chlorine atoms in the repeating structure. This is one reason scientists need more than one kind of model.
Interpreting a formula
Suppose you are given the formula \(\textrm{NH}_3\).
Step 1: Identify the element symbols.
\(\textrm{N}\) stands for nitrogen and \(\textrm{H}\) stands for hydrogen.
Step 2: Read the small numbers.
There is no small number after \(\textrm{N}\), so there is \(1\) nitrogen atom. The \(3\) after \(\textrm{H}\) means there are \(3\) hydrogen atoms.
Step 3: Describe the model.
The formula represents a molecule made of \(1\) nitrogen atom and \(3\) hydrogen atoms.
Formulas are powerful because they let scientists communicate atomic composition quickly. But a formula alone does not always show the shape or pattern. For that, diagrams and physical models are often more helpful.
[Figure 1] When scientists model a simple molecule, they often want to show both the types of atoms and the way the atoms are arranged. A ball-and-stick model uses spheres for atoms and sticks for connections. A structural drawing uses symbols and lines. A space-filling model shows atoms as touching spheres to suggest how much space they take up.
Consider water, \(\textrm{H}_2\textrm{O}\). A model of water shows one oxygen atom connected to two hydrogen atoms. Carbon dioxide, \(\textrm{CO}_2\), has one carbon atom and two oxygen atoms. Oxygen gas, \(\textrm{O}_2\), has two oxygen atoms together. Methane, \(\textrm{CH}_4\), has one carbon atom and four hydrogen atoms. Even without describing every detail of bonding, these models help us count atoms and compare structures.
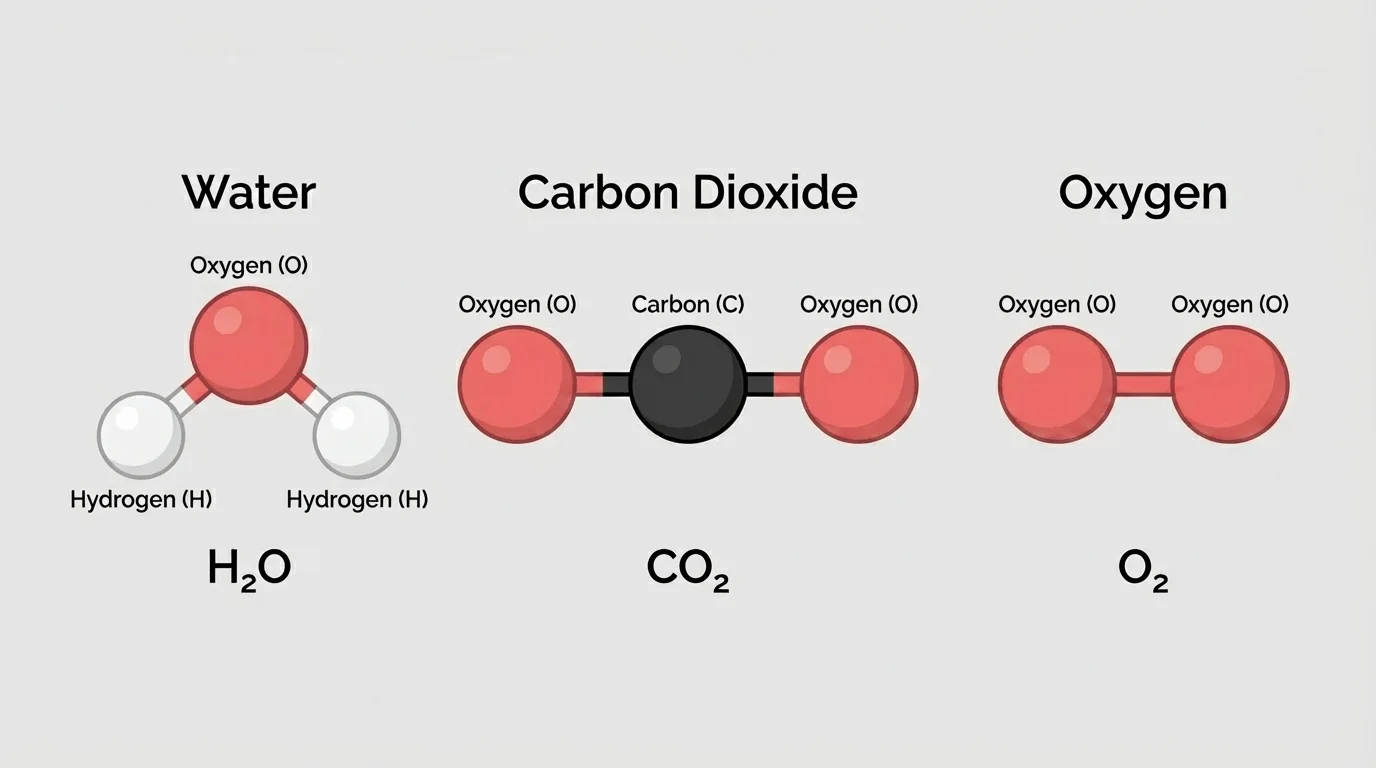
A model can also help prevent a common mistake: students sometimes think \(\textrm{CO}_2\) means one carbon atom floating near two separate oxygen atoms. A molecule model makes it clear that the atoms are part of one unit. The arrangement matters because the substance acts as a specific material, not as three separate atoms.
Scientists choose different models for different purposes. If the goal is to count atoms, a formula may be enough. If the goal is to compare shapes, a drawing is better. If the goal is to picture packed particles, a space-filling model can help. No single model does everything perfectly.
Oxygen in the air is usually not made of single oxygen atoms. It is commonly found as \(\textrm{O}_2\), a molecule with two oxygen atoms joined together.
Later, when comparing gases in the atmosphere, the molecular models in [Figure 1] help explain why water vapor, oxygen, and carbon dioxide are all made of atoms but are still different substances. The number and kind of atoms in each molecule are not the same.
[Figure 2] Not all substances exist as separate tiny molecules. Some are arranged in a repeating pattern that continues over and over in many directions. These are called extended structures. Instead of one little group that stands alone, the atoms are part of a much larger connected arrangement.
Table salt is a familiar example. Its formula is \(\textrm{NaCl}\), but a grain of salt is not one single \(\textrm{NaCl}\) molecule. It contains a huge number of sodium and chlorine atoms in a repeating crystal pattern. Quartz and many rocks also have repeating atomic structures. Metals, such as copper or aluminum, can be modeled as large arrangements of atoms packed together.
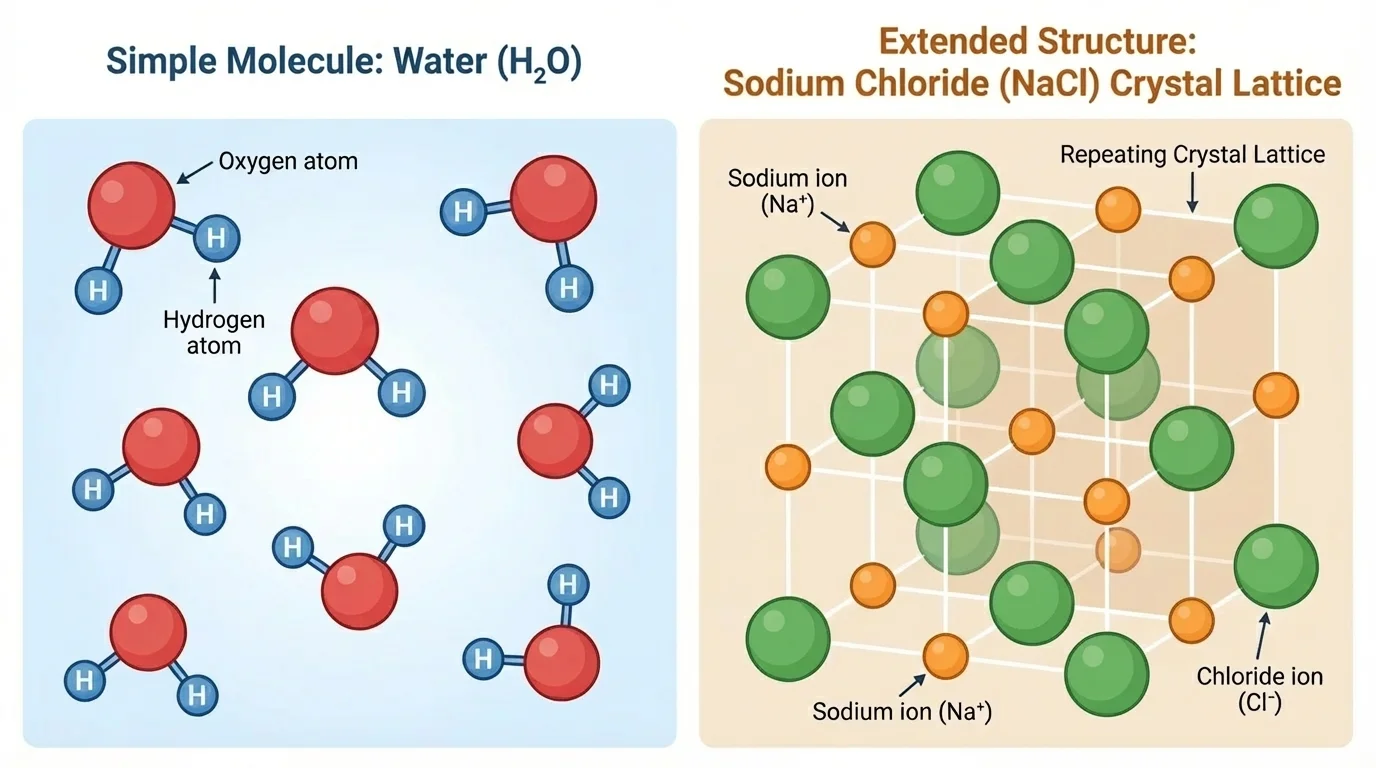
This difference matters. A water sample is made of many separate \(\textrm{H}_2\textrm{O}\) molecules moving around. A crystal of salt is a repeating solid structure. The formula gives useful information in both cases, but the type of model we imagine should not be the same.
Extended structures can be represented with repeating cubes, layered diagrams, or network drawings. These models show that the pattern continues beyond the edge of the picture. That idea is important: the diagram on a page is only a small window into a much larger arrangement.
Why repeating structures are different from simple molecules
A simple molecule is one small unit with a specific number of atoms, such as \(\textrm{H}_2\textrm{O}\). An extended structure is more like a repeated building pattern, where the same arrangement continues many times. In a repeating structure, the formula often tells the simplest ratio of atoms, not the total number of atoms in the whole sample.
When you look at a crystal of salt or a piece of metal, you are seeing the result of many atoms arranged in a regular way. Different arrangements of the same atom can produce very different materials. That regularity helps explain why crystals have definite shapes and why metals can be strong and useful for building tools, wires, and machines.
[Figure 3] The same atoms can sometimes be arranged in different ways and produce materials with different properties. This is one reason models matter so much. They do not just help us count atoms; they help us connect structure to what we observe.
Think about carbon. In one arrangement, carbon atoms can form graphite, the material in pencil cores. Graphite is dark, soft, and slippery. In another arrangement, carbon atoms form diamond, which is extremely hard. Both substances are made only of carbon atoms, but the arrangement is different, so the properties are different too.
Another example is water in different states. In ice, water molecules are arranged in a more fixed pattern. In liquid water, the molecules move past one another more freely. The molecules are still \(\textrm{H}_2\textrm{O}\), but their arrangement and motion change the material's properties.
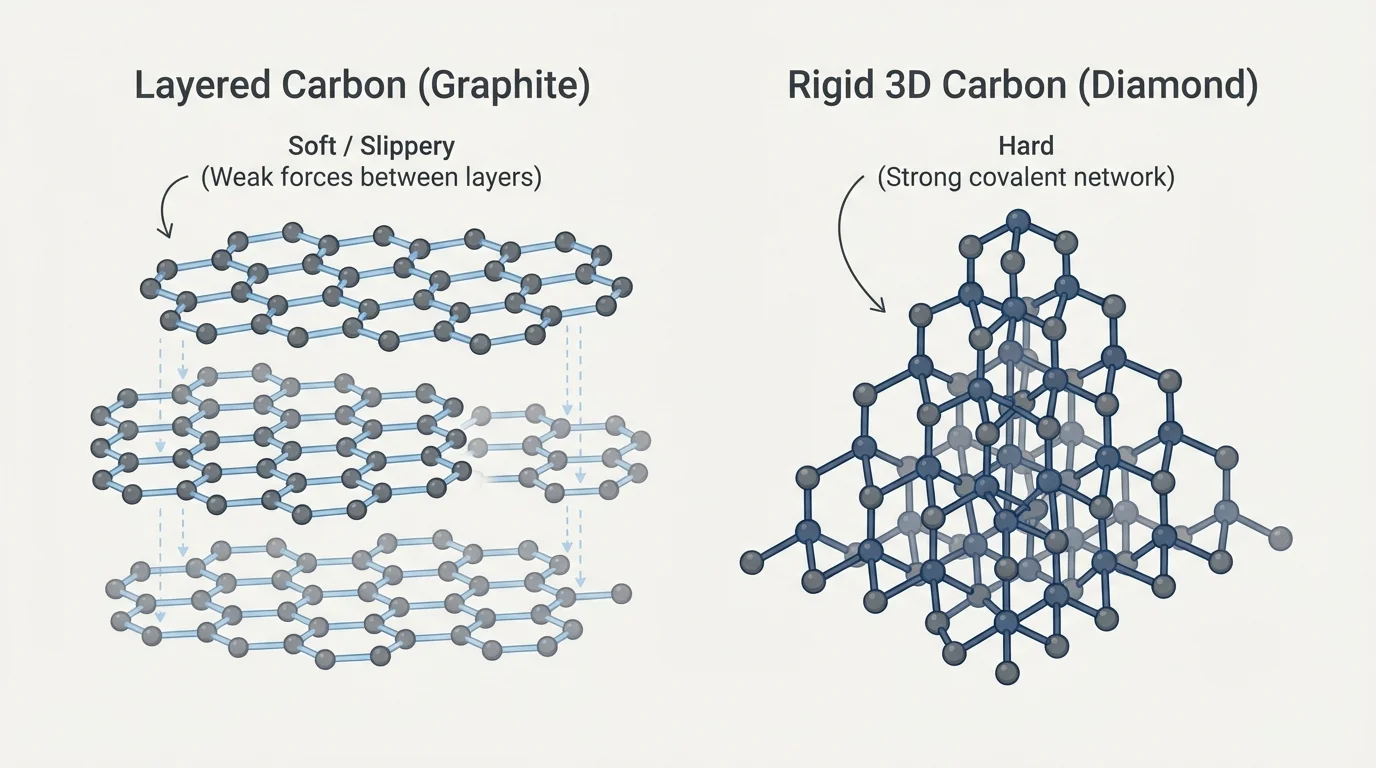
Structure also helps explain why some materials are flexible, some are brittle, and some are easy to shape. Plastics are made of very large molecules arranged in ways that can allow bending. Many ceramics are made from rigid extended structures, which can make them hard but also more likely to crack.
The contrast in [Figure 3] reminds us not to guess a material's properties from its atoms alone. Knowing the kinds of atoms is important, but knowing how those atoms are arranged is often just as important.
Using structure to explain an observation
A student notices that graphite in a pencil writes easily on paper, but diamond does not.
Step 1: Identify what the two materials have in common.
Both are made of carbon atoms.
Step 2: Identify what is different.
The carbon atoms are arranged differently in the two materials.
Step 3: Connect structure to property.
Graphite's arrangement allows layers to slide more easily, so tiny pieces rub off onto paper. Diamond's arrangement is a rigid network, so it is much harder.
This kind of explanation is one of the main goals of physical science. A model becomes useful when it helps explain a real property we can observe and measure.
By this point, two big categories should be clear: some substances are made of small separate molecules, and others are made of extended repeating structures. Both are made of atoms, but their models look different.
| Feature | Simple Molecules | Extended Structures |
|---|---|---|
| Basic unit | A small group of atoms | A repeating arrangement of atoms or particles |
| Example | \(\textrm{H}_2\textrm{O}\), \(\textrm{CO}_2\), \(\textrm{O}_2\) | Salt crystal, quartz, metals |
| What the formula often means | Exact number of atoms in one molecule | Simplest ratio in the repeating pattern |
| How models are often shown | Ball-and-stick or space-filling models | Lattice or network diagrams |
| Scale | One small unit | Pattern continues through the material |
Table 2. Comparison of simple molecules and extended structures as two major ways atoms can be arranged in substances.
This comparison also explains why one picture is not enough for all of chemistry and materials science. A model for water is different from a model for salt crystal or a metal spoon. Scientists select the model that best fits the structure they are studying.
Atomic models are not just classroom drawings. Engineers, chemists, doctors, and materials scientists use them to design useful things. If a scientist wants to create a stronger building material, a safer medicine, or a better battery, that scientist needs to understand how atoms are arranged.
In medicine, researchers model molecules in order to understand how substances might interact in the body. In environmental science, models of \(\textrm{CO}_2\) and other gases help explain the composition of the atmosphere. In construction, understanding the atomic structure of glass, steel, and concrete helps engineers choose the right material for a job.
Cooking also involves atomic-scale thinking, even if you do not see it. Water molecules move differently in ice, liquid water, and steam. Sugar dissolves because tiny particles spread through water. The textures of foods often depend on the arrangement of molecules and larger structures.
Phone screens often use specially designed glass whose atomic structure helps it resist scratching and breaking better than ordinary window glass.
Even recycling depends on structure. Metals can often be melted and reshaped. Plastics differ because their giant molecules and arrangements differ. Understanding composition and structure helps communities sort, reuse, and redesign materials more effectively.
Models are useful, but they are not perfect copies of reality. In many textbook diagrams, atoms look like hard colored balls touching with sticks between them. Real atoms do not look exactly like that. The size, spacing, and appearance are simplified so the important ideas are easier to understand.
A formula also has limits. It can tell you composition, but it may not fully show shape, motion, scale, or the full complexity of a large structure. That is why scientists often use several models together: formulas, particle drawings, computer images, and physical kits.
Good science involves asking what a model helps you see and what it leaves out. A ball-and-stick model is great for counting and arrangement. A space-filling model is better for showing how atoms occupy space. A repeating lattice diagram is better for a crystal. Choosing the right model is part of scientific thinking.
"Science often advances by making the invisible understandable through models."
When you describe the atomic composition of a substance, you are doing more than naming atoms. You are answering a deeper question: how is matter built? That question connects a glass of water, the air in a room, a grain of salt, and the materials used in spacecraft.