A puddle disappears after rain, metal feels colder than wood in the same room, and water can boil furiously while staying at the same temperature. These everyday events may seem unrelated, but they all connect to one powerful idea: matter is made of tiny particles, and those particles respond in predictable ways when thermal energy is added or removed.
To understand these changes, scientists build models. A model is a simplified way to explain something that is too small, too large, or too complex to observe directly. In this lesson, the model is based on particles such as atoms and molecules. Even though we cannot see these particles with our eyes, we can use evidence to describe how they move and how that motion changes the temperature and state of a substance.
When a bowl of hot soup gives off steam, the particles in the liquid are moving fast enough that some escape into the air as a gas. When water on a winter road freezes, thermal energy is removed, the particles slow down, and they become locked into a more fixed arrangement. These changes are not random. They follow patterns that can be predicted.
A good scientific model helps answer questions like these: If thermal energy is added to ice, what happens first? If more thermal energy is added after the ice melts, what changes next? If thermal energy is removed from water vapor, does it always cool first, or can it change into liquid while the temperature stays the same? The answers depend on particle motion and the state of the substance.
All matter is made of tiny particles. Some substances are made of individual atoms, and others are made of molecules, which are groups of atoms joined together. Water, for example, is made of molecules of \(\textrm{H}_2\textrm{O}\). Oxygen gas is made of molecules of \(\textrm{O}_2\). Iron is made of iron atoms.
A pure substance is matter made of only one kind of particle. That means every particle in a sample is the same. Pure water contains only \(\textrm{H}_2\textrm{O}\) molecules. Pure oxygen contains only \(\textrm{O}_2\) molecules. A mixture, by contrast, contains more than one kind of particle. This lesson focuses on pure substances because their changes of state follow clearer patterns.
Thermal energy is the total energy connected to the motion of particles in a substance. Temperature is a measure of the average kinetic energy of those particles. State of matter means whether a substance is a solid, liquid, or gas.
It is important to notice that thermal energy and temperature are related, but they are not exactly the same thing. Temperature tells how energetic the particles are on average. Thermal energy involves the energy of all the particles together. For middle school science, the key idea is that when particles move faster, temperature usually increases, and when particles move slower, temperature usually decreases.
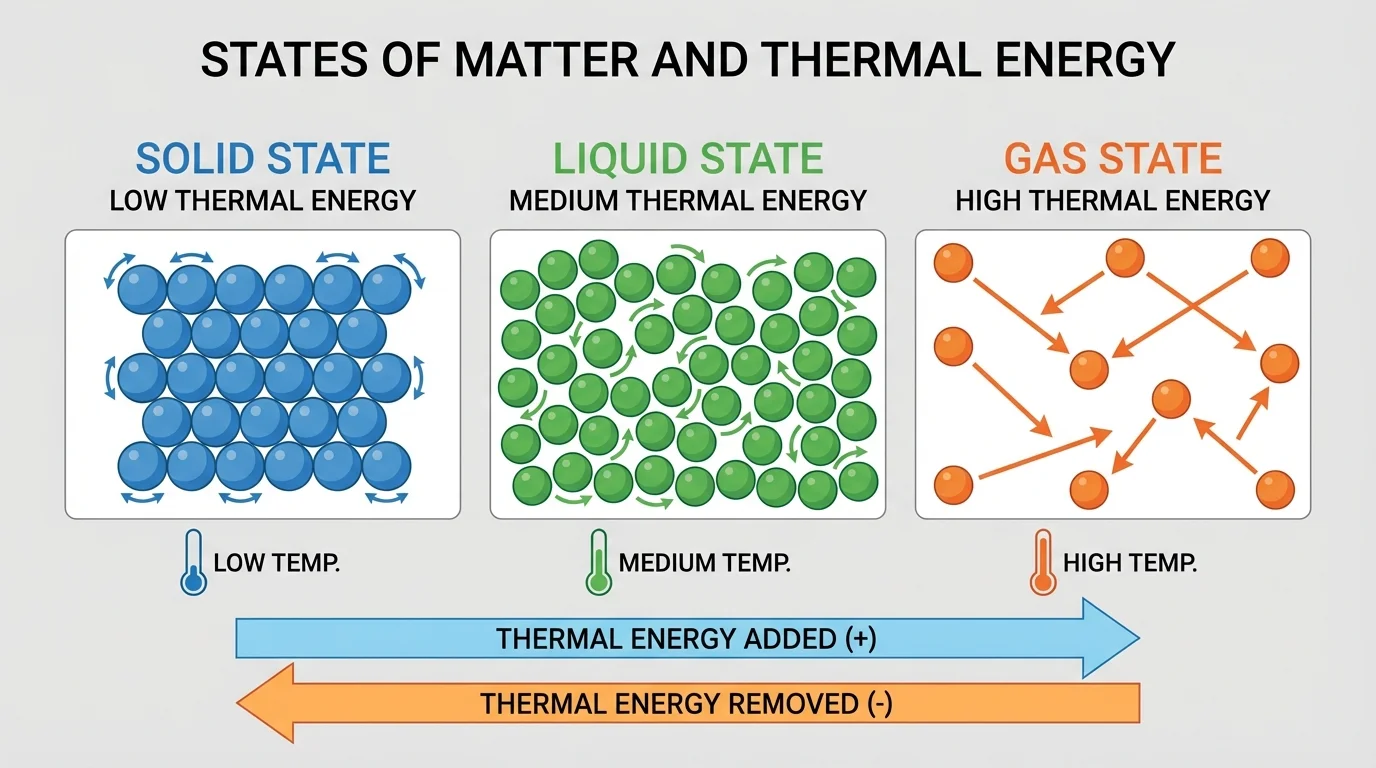
[Figure 1] shows that in each state of matter, particles have a different arrangement and different kinds of motion. These differences explain why solids keep their shape, liquids flow, and gases spread out to fill a container.
In a solid, particles are packed closely together in fixed positions. They still move, but mostly by vibrating in place. Because they cannot move past each other easily, a solid keeps its shape. In a liquid, particles are still close together, but they can slide past one another. That is why liquids flow and take the shape of their container. In a gas, particles are much farther apart and move freely in many directions. That is why gases expand to fill all available space.
| State | Particle arrangement | Particle motion | Visible behavior |
|---|---|---|---|
| Solid | Very close together, fixed pattern | Vibrate in place | Keeps its own shape |
| Liquid | Close together, not fixed | Slide past one another | Flows, takes container shape |
| Gas | Far apart | Move freely and quickly | Spreads to fill container |
Table 1. Comparison of particle arrangement, motion, and behavior in the three common states of matter.
These particle differences are the basis of the model. If thermal energy is added, particles usually move more. If thermal energy is removed, particles usually move less. Once enough change happens, the substance may change state.
When thermal energy is added to a pure substance, one of two main things can happen. The particles can move faster, which usually raises the temperature, or the substance can change state, which changes how the particles are arranged. When thermal energy is removed, the opposite can happen: particles can slow down, lowering the temperature, or the substance can change into a state with particles that are more tightly connected.
This means a scientist does not just ask, "Is the substance getting hotter or colder?" A better question is, "What are the particles doing?" If the particles are moving faster in the same state, the temperature rises. If the particles are rearranging during a phase change, the temperature may stay the same for a while even though thermal energy is still being added or removed.
The particle-motion model explains temperature and state changes together. Faster particle motion usually means higher temperature. Slower particle motion usually means lower temperature. But during a change of state, energy may go into changing how particles are connected or spaced rather than making them move faster.
This is one reason water is so interesting. Ice, liquid water, and water vapor are all the same pure substance: \(\textrm{H}_2\textrm{O}\). The particles are the same in all three cases. What changes is their motion, spacing, and arrangement.
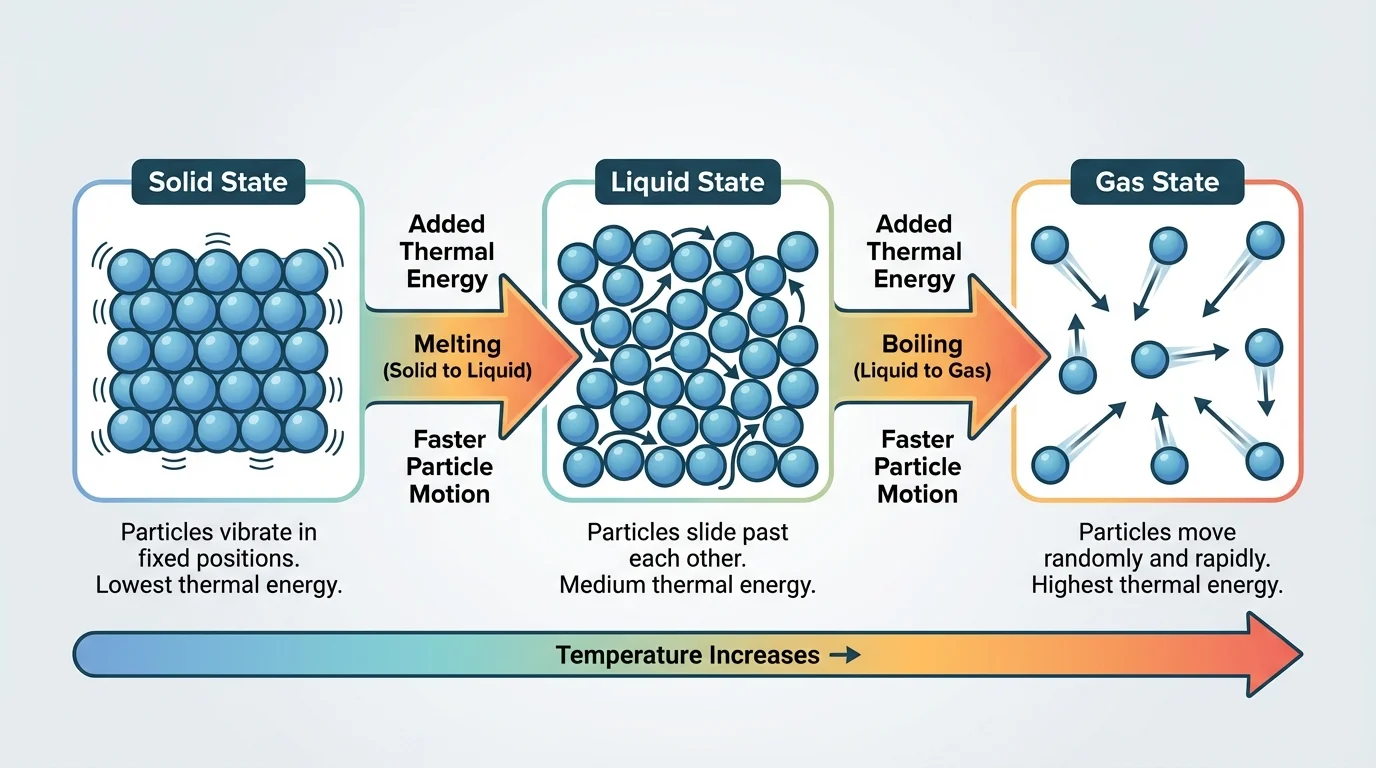
Scientists use a step-by-step model to predict what happens as a pure substance is heated or cooled, as [Figure 2] illustrates. The model is simple enough to use with many substances, including water, oxygen, and metals.
If thermal energy is added: first ask what state the substance is in. If it is staying in the same state, its particles move faster and the temperature rises. If it reaches the point where it changes state, the energy goes into changing particle arrangement. For example, a solid may melt into a liquid, or a liquid may boil into a gas.
If thermal energy is removed: again ask what state the substance is in. If the state stays the same, particles slow down and temperature falls. If the substance reaches a phase-change point, the particles rearrange into a different state. A gas may condense into a liquid, or a liquid may freeze into a solid.
This model allows predictions without complicated formulas. Students can trace the path: same state means change in particle speed and temperature; changing state means change in arrangement, often with no temperature change for a time.
A phase change is a change from one state of matter to another. For pure substances, these changes happen at specific temperatures under the same conditions. The most common phase changes are melting, freezing, vaporization, condensation, sublimation, and deposition.
Melting happens when a solid gains enough thermal energy that its particles are no longer locked in place. They can move past each other, and the substance becomes a liquid. Ice melting into liquid water is a familiar example.
Freezing is the reverse. As thermal energy is removed, liquid particles slow down and become more fixed in position. The liquid becomes a solid. Water freezing into ice on a cold night is an example.
Boiling or vaporization happens when liquid particles gain enough energy to escape into the gas state throughout the liquid. Evaporation is a similar change from liquid to gas, but it happens only at the surface and can occur below the boiling point. A wet shirt drying on a line is evaporation.
Condensation is the change from gas to liquid. When water vapor in the air loses thermal energy, it can condense into tiny liquid drops. That is how dew forms on grass and water droplets form on the outside of a cold glass.
Sublimation is when a solid changes directly into a gas without becoming a liquid first. Dry ice, which is solid \(\textrm{CO}_2\), is a well-known example. Deposition is the reverse, when a gas changes directly into a solid. Frost forming from water vapor on a very cold surface is an example.
Real-world example: a freezer tray of water
Step 1: Liquid water starts above its freezing point.
The particles move around each other, so the substance is a liquid.
Step 2: Thermal energy is removed in the freezer.
The particles slow down, so the temperature decreases.
Step 3: At the freezing point, the state begins to change.
The particles rearrange into fixed positions and form solid ice.
Step 4: After all the water freezes, removing more thermal energy makes the ice colder.
The particles in the solid vibrate less vigorously.
The same reasoning works for heating a substance. If you heat ice, the solid first warms. At the melting point, it changes to liquid. After all of it has melted, the liquid warms. At the boiling point, it changes to gas. This is the predictive power of the model.
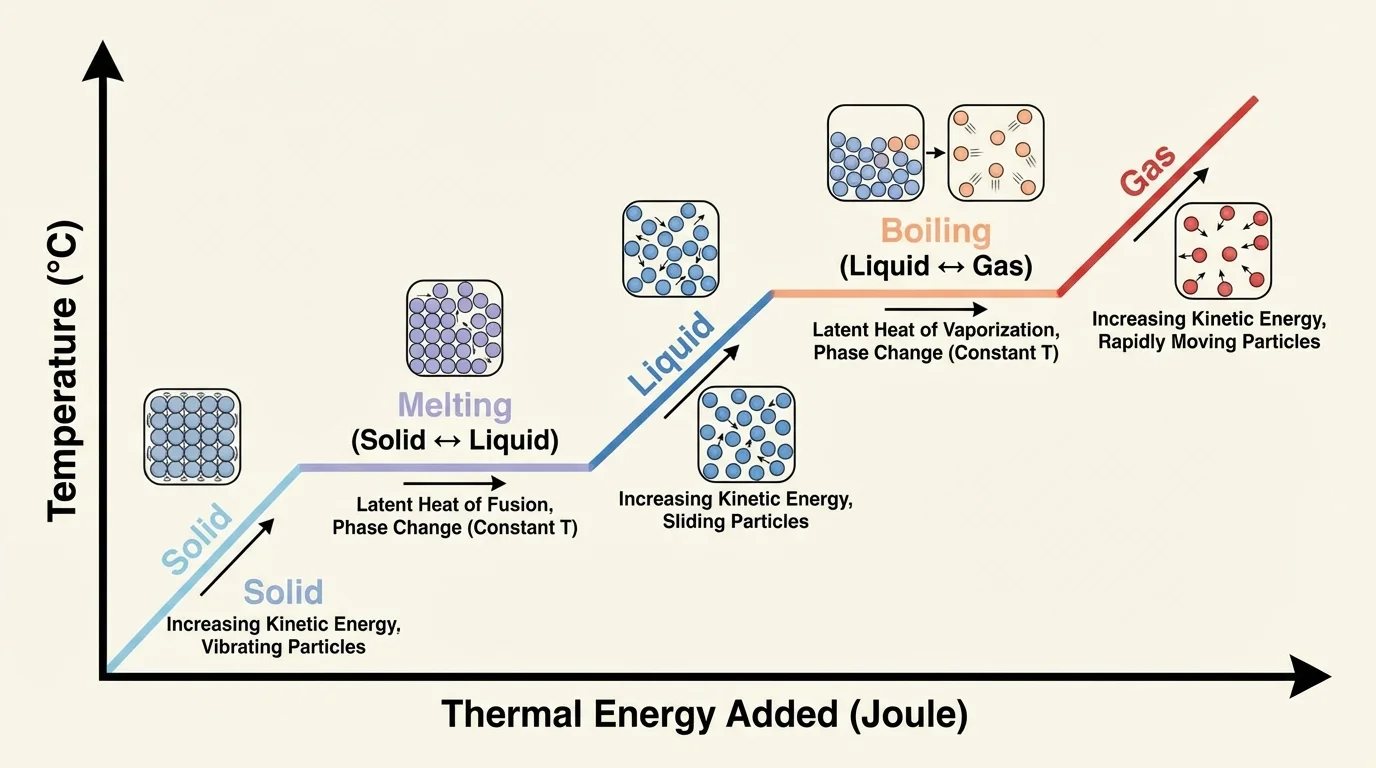
[Figure 3] helps explain one of the most surprising ideas in thermal science: temperature does not always change when thermal energy is added or removed. During melting or boiling, the added energy is used to change particle arrangement instead of increasing average particle motion.
For example, a sample of pure water can stay at its boiling point while it is boiling. Even though a stove keeps adding thermal energy, the temperature stays the same until the liquid water has finished changing into gas. The particles are using that energy to separate more completely from one another.
The same thing happens during freezing and condensation when thermal energy is removed. The temperature can remain constant while the state changes because the particles are rearranging into a more organized or closer form. Only after the phase change is complete does the temperature begin to drop again.
This idea also helps explain why steam burns can be severe. Water vapor condenses on skin, and during that condensation it releases thermal energy while changing from gas to liquid. The change of state transfers a large amount of energy even if the temperature reading itself is not changing during the phase change.
Water is a useful example, but the model applies to many pure substances. Oxygen can exist as solid \(\textrm{O}_2\), liquid \(\textrm{O}_2\), or gaseous \(\textrm{O}_2\) depending on temperature and conditions. Iron can be heated until it glows and eventually melts. In each case, the particles are still the same kind of substance. Their energy, spacing, and arrangement change.
Looking back at [Figure 1], the big idea is not just what particles look like in each state, but how those particle patterns help us make predictions. Closely packed particles that only vibrate suggest a solid. Particles that slide past one another suggest a liquid. Widely spaced particles moving freely suggest a gas.
And as [Figure 2] shows, the heating path for a pure substance follows a logical sequence. Add thermal energy and particles move faster until a phase-change point is reached. Continue adding energy and the state changes. Remove thermal energy and the reverse sequence occurs.
Liquid nitrogen is so cold that it can quickly remove thermal energy from many materials. It is the same substance, \(\textrm{N}_2\), that makes up most of the air, but in a much colder liquid state.
Different pure substances melt and boil at different temperatures, but the particle model still works. That is why scientists can use a common explanation for many materials while also recognizing that each substance has its own specific properties.
Weather depends heavily on these ideas. Water evaporates from oceans, lakes, and soil. Higher particle motion allows water molecules to escape into the air. Later, water vapor cools and condenses to form clouds. If enough thermal energy is removed, precipitation may freeze into snow or sleet.
Cooking also depends on thermal energy and state changes. When water boils in a pot, it changes from liquid to gas. When butter melts in a pan, solid fat changes to liquid because its particles gain enough energy to move more freely. Bakers also pay attention to steam because water vapor trapped in dough can help bread rise.
Refrigerators and air conditioners work by moving thermal energy from one place to another. A refrigerant substance changes state inside the system. It evaporates in one part of the system and condenses in another. Those changes of state are useful because they involve transfers of thermal energy.
Engineers and manufacturers also use these ideas. Metals are melted and reshaped. Plastic materials soften when heated and harden again when cooled. Understanding particle motion helps scientists choose the right substance for the right job.
Energy can be transferred from warmer objects to cooler ones. Thermal energy naturally moves from a place with higher temperature to a place with lower temperature until conditions become more balanced.
Safety matters, too. Frostbite can happen when thermal energy leaves body tissues very quickly. Burns can happen when thermal energy enters tissues quickly, including from steam. Even road salt connects to this topic because it changes how water freezes, which is why it helps melt ice on roads.
One common mistake is thinking that particles in a solid do not move. They do move; they vibrate in place. Another mistake is thinking that when a substance melts or boils, its particles become different particles. They do not. Ice, liquid water, and steam are all made of \(\textrm{H}_2\textrm{O}\) molecules.
A third misunderstanding is believing that adding thermal energy always increases temperature immediately. As [Figure 3] demonstrates, temperature can stay the same during melting or boiling because the energy is changing the state rather than speeding up the particles.
It is also important not to confuse heat feeling with actual temperature. A metal chair and a plastic chair outside may be at the same temperature, yet the metal may feel colder because thermal energy moves differently between your skin and the material. Science models help explain the cause instead of relying only on what we sense.