A small packet can warm frozen hands in winter, while another can help cool an injured ankle in seconds. That seems almost like magic, but it is really chemistry and engineering working together. When certain substances react, they can transfer thermal energy to the surroundings or take thermal energy in. A good design project turns that scientific idea into a useful device.
A design project is more than building something once. It involves planning, constructing, testing, and improving. In this topic, the goal is to make a device that either gets warmer because a reaction releases energy or gets colder because a reaction absorbs energy. Students act like engineers: they define a problem, build a prototype, collect evidence, and make changes based on the results.
These ideas matter in real life. People use warming devices when camping, working outside, or during power outages. Cooling devices are used in sports medicine, first aid, and shipping items that must stay cold. The same basic science supports all of these tools.
Matter is made of atoms, and atoms are not destroyed in ordinary chemical reactions. Instead, atoms in the starting substances are rearranged to make new substances. Energy can also change form during reactions.
When you design a thermal device, you are not just asking, "Does it work?" You are also asking, "How much material should be used?" "How fast does the temperature change?" and "What temperature does the device reach?" Those are the main criteria for this topic.
A chemical reaction happens when substances change into different substances. During the reaction, atoms are rearranged into new groupings, as [Figure 1] shows. The total number of each type of atom stays the same before and after the reaction, even though the substances themselves change.
For example, when iron in some hand warmers reacts with oxygen in the air, new substances form and thermal energy is released. In a simple way, we can describe that as iron and oxygen reacting to form iron oxide: \[4\textrm{Fe} + 3\textrm{O}_2 \rightarrow 2\textrm{Fe}_2\textrm{O}_3\] The important idea is not memorizing the equation, but understanding that the atoms in iron and oxygen are still present after the reaction. They have simply been rearranged.
Some reactions are exothermic reactions. That means they release thermal energy to the surroundings, so the device feels warmer. Other reactions are endothermic reactions. That means they absorb thermal energy from the surroundings, so the device feels colder.
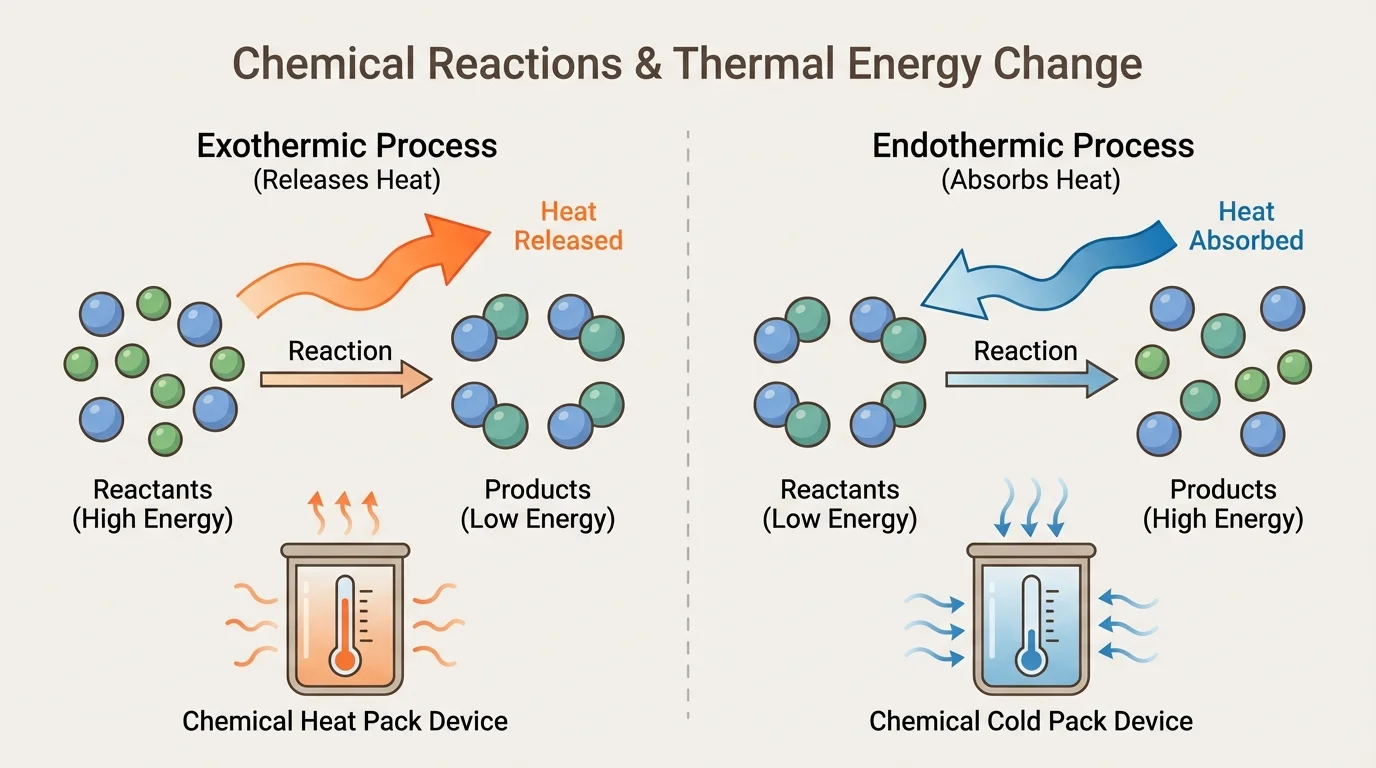
You can think of thermal energy transfer like money moving between accounts. In an exothermic reaction, energy moves out of the reacting substances into the surroundings. In an endothermic reaction, energy moves from the surroundings into the reacting substances. The matter changes, and energy transfer happens at the same time.
A common cooling example involves substances in instant cold packs. When certain solids dissolve in water, the process can absorb thermal energy, making the pack colder. A common warming example is a hand warmer that uses oxidation, a reaction involving oxygen. Later, when comparing devices, these same ideas still matter because every successful design depends on both atom rearrangement and energy transfer.
Thermal energy is the energy associated with the motion of particles in matter and is related to temperature.
Temperature is a measure of how hot or cold something is.
Prototype is an early model of a device built for testing and improvement.
Temperature is what you measure with a thermometer, while thermal energy is the energy associated with the motion of particles in matter. A larger amount of material may undergo a greater total thermal energy change than a smaller amount, even if the temperature change is similar. That is one reason the amount of substance matters in testing.
In this lesson, a device is a simple system made to cause useful warming or cooling through a chemical process. It may be a pouch, cup, packet, or container that safely holds the reacting substances. The device is not just the chemicals. It also includes the way the materials are arranged and the way the user activates it.
For example, a cold pack often keeps water in a separate inner bag and a solid chemical in the outer part. Squeezing the pack breaks the inner bag, the substances mix, and the temperature drops. A heat pack may let air reach iron powder, salt, water, and other materials so oxidation can begin.
Good engineering design includes criteria and constraints. Criteria tell what success looks like. In this topic, the criteria are limited to the mass of the substance used, the time over which the effect lasts or develops, and the temperature reached by the device or sample being tested. Constraints are limits such as safety, available materials, and size of the device.
Some commercial hand warmers can stay warm for several hours because the device is designed to let oxygen enter slowly rather than all at once. The chemistry and the packaging work together.
That is an important point: chemistry alone does not decide whether a device is useful. The same reaction can perform differently depending on container shape, insulation, mixing method, or how much reactant is used.
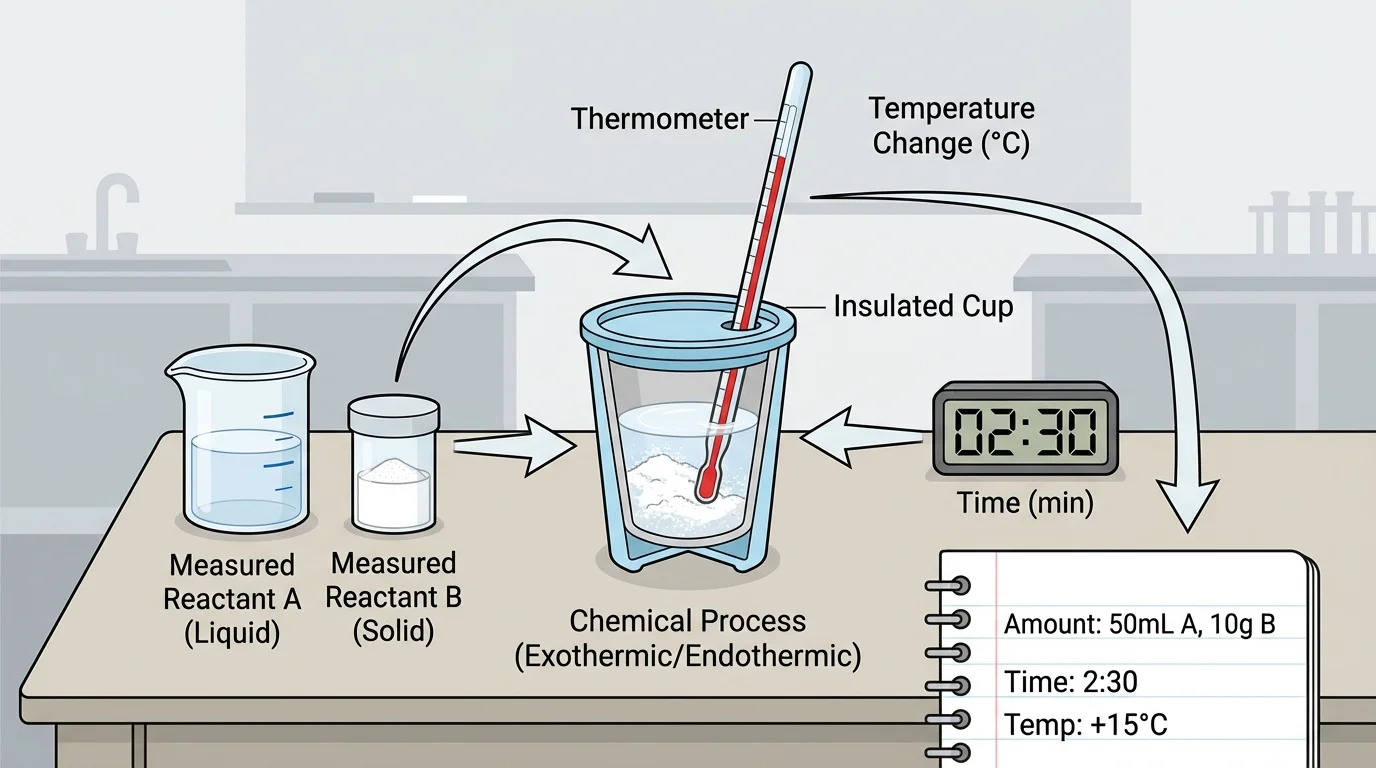
Before building anything, engineers make a plan. For this kind of project, the plan should focus on measurable evidence and include a clear testing setup built around amount, time, and temperature. A strong plan begins with a problem such as: "Design a small device that warms water quickly," or "Design a cold pack that lowers temperature for several minutes."
Next, identify the materials. A warming device might use iron powder exposed to air, or another safe classroom-approved chemical process that releases thermal energy. A cooling device might use a dissolving substance that absorbs thermal energy when mixed with water. The exact substances depend on the classroom setup and safety rules.
Then decide what will be measured. Students may record the amount of substance in grams, the time in seconds or minutes, and the temperature in degrees Celsius. These measurements allow fair comparisons between different prototypes. For example, one prototype might use more reacting material, while another uses less but is better insulated.
A useful test table helps organize evidence.
| Prototype | Mass of substance | Starting temperature | Highest or lowest temperature | Time to reach that temperature |
|---|---|---|---|---|
| A | \(20\,\textrm{g}\) | \(22^\circ\textrm{C}\) | \(34^\circ\textrm{C}\) | \(4\,\textrm{min}\) |
| B | \(30\,\textrm{g}\) | \(22^\circ\textrm{C}\) | \(39^\circ\textrm{C}\) | \(3\,\textrm{min}\) |
When students compare prototypes, they should change only one major factor at a time when possible. If the amount of substance changes, the container and starting temperature should stay the same. If the container changes, the amount should stay the same. This makes it easier to tell which change affected the results.
Planning a fair warming-device test
Step 1: Choose a clear question.
Example: Which prototype warms water to a higher temperature using the same starting amount of water?
Step 2: Keep important conditions the same.
Use the same amount of water, the same starting temperature, the same kind of container, and the same thermometer.
Step 3: Change one factor.
For instance, compare \(20\,\textrm{g}\) of reacting substance with \(30\,\textrm{g}\) of the same substance.
Step 4: Record evidence.
Measure the temperature every minute and note how long the device continues warming.
This plan produces evidence that can support a design decision.
A formula can help describe temperature change: \[\Delta T = T_{\textrm{final}} - T_{\textrm{initial}}\] If water starts at \(22^\circ\textrm{C}\) and reaches \(34^\circ\textrm{C}\), then the change is \(34 - 22 = 12^\circ\textrm{C}\). For a cooling device, if a substance starts at \(22^\circ\textrm{C}\) and drops to \(10^\circ\textrm{C}\), then \(10 - 22 = -12^\circ\textrm{C}\). The negative sign shows a decrease in temperature.
Once the plan is ready, build the prototype carefully. Use safe materials, closed containers if needed, and accurate measuring tools. Label each prototype clearly so the data do not get mixed up. Even a very simple device can produce useful evidence if the testing is careful.
Start each trial with the same starting conditions whenever possible. Measure the initial temperature, activate the device, and begin timing right away. Then record the temperature at regular intervals, such as every \(30\,\textrm{s}\) or every \(1\,\textrm{min}\).
Repeated trials matter because one test can be affected by small mistakes. If prototype A is tested three times and reaches \(33^\circ\textrm{C}\), \(34^\circ\textrm{C}\), and \(35^\circ\textrm{C}\), the average is \[\frac{33 + 34 + 35}{3} = \frac{102}{3} = 34^\circ\textrm{C}\] That average gives a better picture of performance than one trial alone.
Why fair testing matters
A fair test compares prototypes in a way that isolates the effect of one design choice. If several factors change at once, such as amount, container, and starting temperature, then the results do not clearly show what caused the difference.
During testing, students should watch for signs that the device is not working as intended. A leak, poor sealing, uneven mixing, or delayed timing can all affect the outcome. Careful notes are part of good science and engineering.
The most important part of engineering is what happens after the first test. Evidence should guide improvements, and data comparisons can show how two versions of a device perform differently over time. If one design heats faster but does not stay warm very long, while another heats more slowly but lasts longer, the better choice depends on the goal.
Suppose a heating device using \(20\,\textrm{g}\) of reacting substance raises the temperature by \(8^\circ\textrm{C}\) in \(2\,\textrm{min}\), but a version using \(30\,\textrm{g}\) raises it by \(15^\circ\textrm{C}\) in \(3\,\textrm{min}\). If the goal is a larger temperature increase, the second design may be better. If the goal is to use less material, the first might be preferred.

Modification means changing the design based on what the data show. A student might increase the amount of reacting substance, improve insulation, change how the substances mix, or adjust the size of the container. The key is that each change should have a reason supported by test results.
This is where science and engineering connect directly. Science explains why a reaction releases or absorbs thermal energy. Engineering uses that scientific knowledge to produce a device that meets a need. Looking back to the particle changes described earlier, the energy behavior of the reaction helps explain why one prototype warms or cools more than another.
Using evidence to improve a cooling device
Step 1: Identify the problem in the first prototype.
The pack becomes cold, but only for \(2\,\textrm{min}\).
Step 2: Look at the data.
The temperature drops from \(21^\circ\textrm{C}\) to \(11^\circ\textrm{C}\), but quickly rises again.
Step 3: Make one change.
Increase the amount of dissolving substance or improve the container so the cooling lasts longer.
Step 4: Retest using the same method.
Compare the new time and temperature results to see whether the modification improved the device.
Evidence, not guessing, should drive the redesign.
Sometimes a modification improves one criterion but worsens another. More material may increase the temperature change, but it may also make the device bulkier or cause the reaction to finish too quickly. Engineers often balance several needs at the same time.
These ideas are not limited to classrooms. Chemical thermal devices appear in sports medicine, outdoor survival, transportation, and emergency response. A runner may use a cold pack after a sprain. A hiker may use a hand warmer on a freezing day. In both cases, the device is designed around how much material to use, how quickly the temperature changes, and how long the effect lasts.
Food and medicine shipping can also involve thermal design. Packages may include chemical packs that help keep contents within a safer temperature range during travel. Designers test these systems carefully because temperature changes over time can affect whether the shipment stays useful.
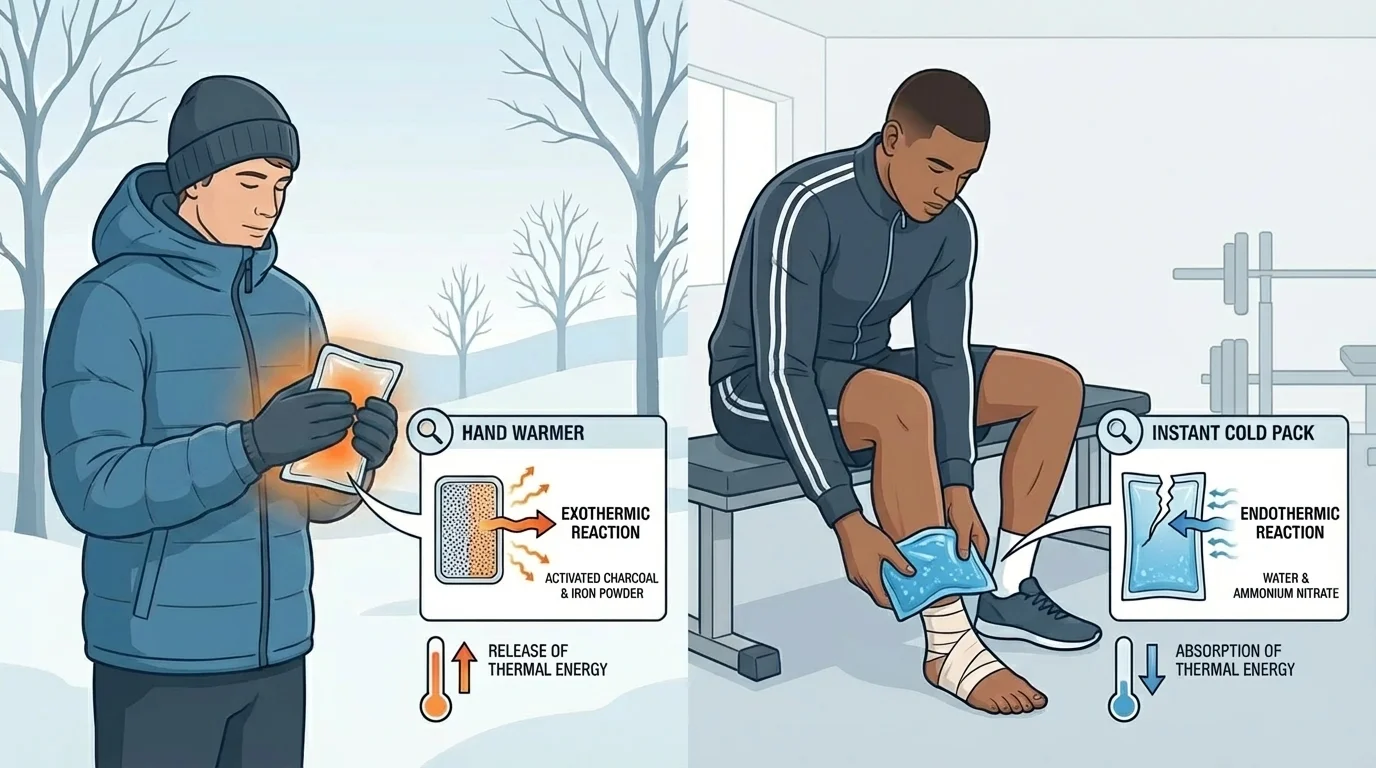
Even though classroom projects are simpler, the reasoning is very similar. A successful design solves a specific problem using scientific evidence. The project is not about making the strongest possible reaction. It is about making a controlled and useful device.
When students compare their results with commercial examples, they often notice that packaging matters greatly. The chemistry may be effective, but poor device design can waste the energy change. That is why a clear testing setup is so important: measuring the effect clearly helps identify which design choices really help.
One common mistake is changing too many things at once. If amount, container, and activation method all change together, then the result is hard to interpret. Another mistake is not measuring temperature at regular times. A device may seem impressive at first but lose effectiveness quickly.
Another problem is not recording the starting temperature. If one trial begins at \(18^\circ\textrm{C}\) and another begins at \(24^\circ\textrm{C}\), the comparison may not be fair. Careful notes, repeated trials, and clear labels make the evidence stronger.
"Good design is improved by good evidence."
Students should also be cautious about making claims that the data do not support. If only amount, time, and temperature were tested, then conclusions should stay focused on those criteria. That keeps the project scientifically honest and within the expected scope.
At the heart of the project is a simple but powerful idea: when substances react, atoms rearrange into new substances, and energy can be transferred. If the process releases thermal energy, a warming device can be designed. If the process absorbs thermal energy, a cooling device can be designed.
The best design is not chosen by opinion. It is chosen by evidence from testing. Students measure how much substance is used, how long the effect lasts or develops, and what temperature change occurs. Then they revise the prototype and test again.
That cycle of building, testing, and improving is one of the most important habits in engineering. It turns chemistry from a set of facts into a practical tool for solving problems.