Why does a metal bench feel colder than a wooden bench on the same day? Why does a pizza box keep food warm for a while, but not forever? Why can a tiny insulated bottle keep a drink hot during a long bus ride? These questions all connect to one big idea: thermal energy transfer. Engineers study how thermal energy moves so they can design devices that either slow it down or speed it up, depending on the job that needs to be done.
Every day, people depend on devices that control thermal energy transfer. A refrigerator slows the movement of thermal energy into cool food. A frying pan helps thermal energy move quickly from the stove to food. A winter coat reduces thermal energy transfer from your body to cold air. A car radiator helps thermal energy leave the engine so it does not overheat.
Designing these devices is not guesswork. Scientists and engineers apply evidence, test ideas, compare materials, and improve prototypes. A successful design matches its purpose. If the goal is to keep cocoa warm, the device should minimize thermal energy transfer. If the goal is to cool a computer chip, the device should maximize thermal energy transfer away from the chip.
Energy can move from one place to another and can change form, but it is conserved. Thermal energy is related to the motion of particles, so understanding particle motion helps explain why some objects warm up or cool down faster than others.
One important idea in physical science is that kinetic energy is the energy of motion. Even particles too small to see are moving. Their motion helps explain temperature and thermal energy transfer.
Thermal energy is the total energy connected to the motion of particles in matter. Particles in solids vibrate in place. Particles in liquids slide past one another. Particles in gases move more freely and spread out more. When particles move faster on average, they have more kinetic energy.
Temperature tells us about the average kinetic energy of particles in a substance. If one cup of water is hotter than another, the particles in the hotter cup are moving faster on average. Thermal energy naturally tends to move from a warmer object or area to a cooler one.
For example, if a hot spoon is placed in cooler water, energy moves from the spoon to the water until their temperatures become closer. You do not need to calculate the total amount of energy transferred to understand the process. What matters here is the direction of transfer and the factors that make it faster or slower.
Thermal energy transfer is the movement of thermal energy from a warmer object or area to a cooler one.
Temperature is a measure of the average kinetic energy of particles.
Kinetic energy is the energy an object or particle has because of its motion.
This connection between motion and temperature helps explain why certain designs work. A device that slows particle interactions, traps air, or reflects radiation can reduce thermal energy transfer. A device that uses metal, increases contact, or increases surface area can help thermal energy transfer more quickly.
[Figure 1] shows that thermal energy moves in three main ways: conduction, convection, and radiation. A good designer needs to know which kind of transfer matters most in a device.
Conduction is thermal energy transfer through direct contact. If you touch a cold metal railing, energy from your hand moves into the metal. Metals are often good conductors of thermal energy, which is why metal pans heat food effectively.
Convection happens in liquids and gases when warmer, less dense parts rise and cooler, denser parts sink. This creates a moving current. Boiling water is a familiar example. Warm water rises from the bottom of the pot while cooler water sinks to be heated.
Radiation transfers energy by electromagnetic waves and does not require direct contact or matter between objects. The Sun warms Earth mostly by radiation. A campfire can warm your hands even if you are not touching the flames.
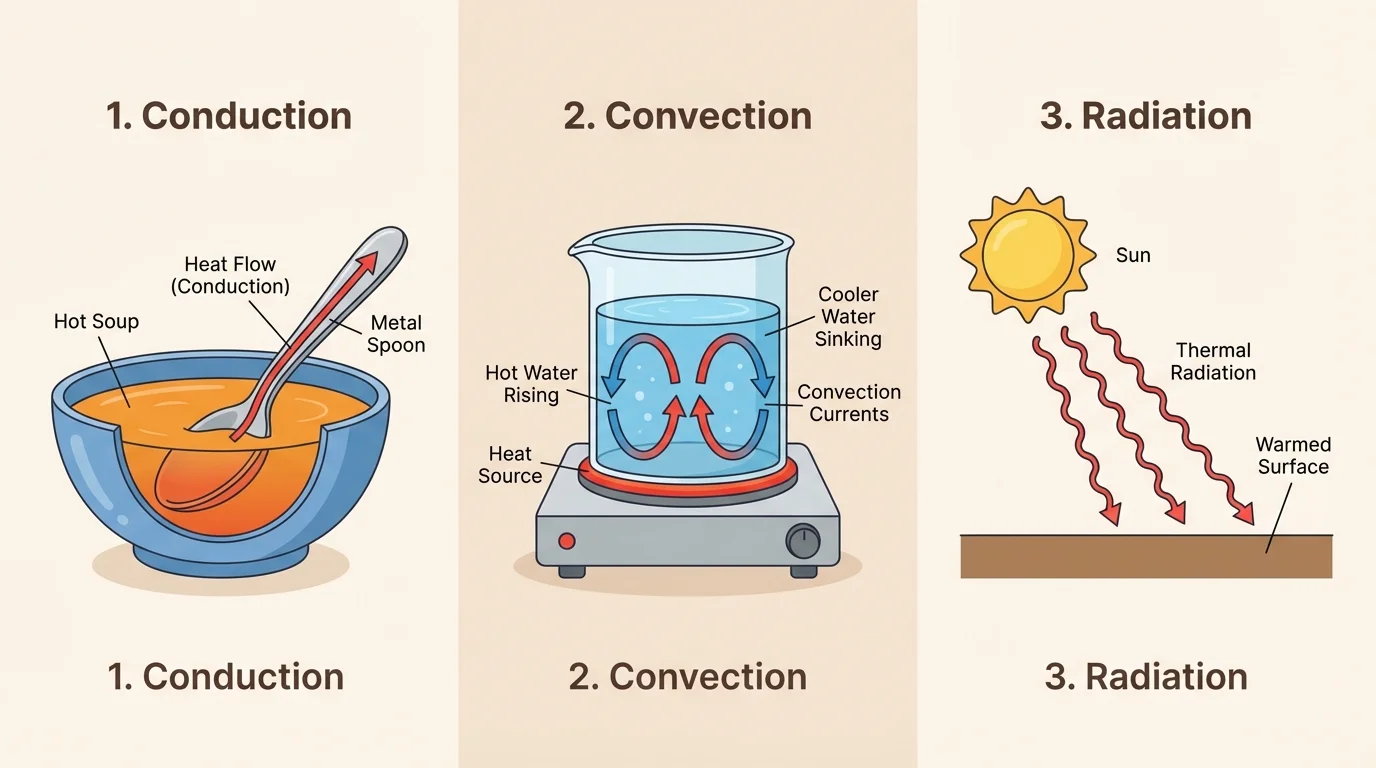
These three processes often happen at the same time. A mug of hot tea loses energy by conduction through the cup, by convection into the surrounding air, and by radiation from its warm surface. Engineers often design a device to reduce all three at once or to increase one of them on purpose.
Later, when testing a device, it is useful to ask which type of transfer is most important. For example, in an insulated cup, conduction through the wall and convection at the opening are major concerns, while in a solar cooker, radiation is especially important. The three-panel view in [Figure 1] helps separate these processes clearly.
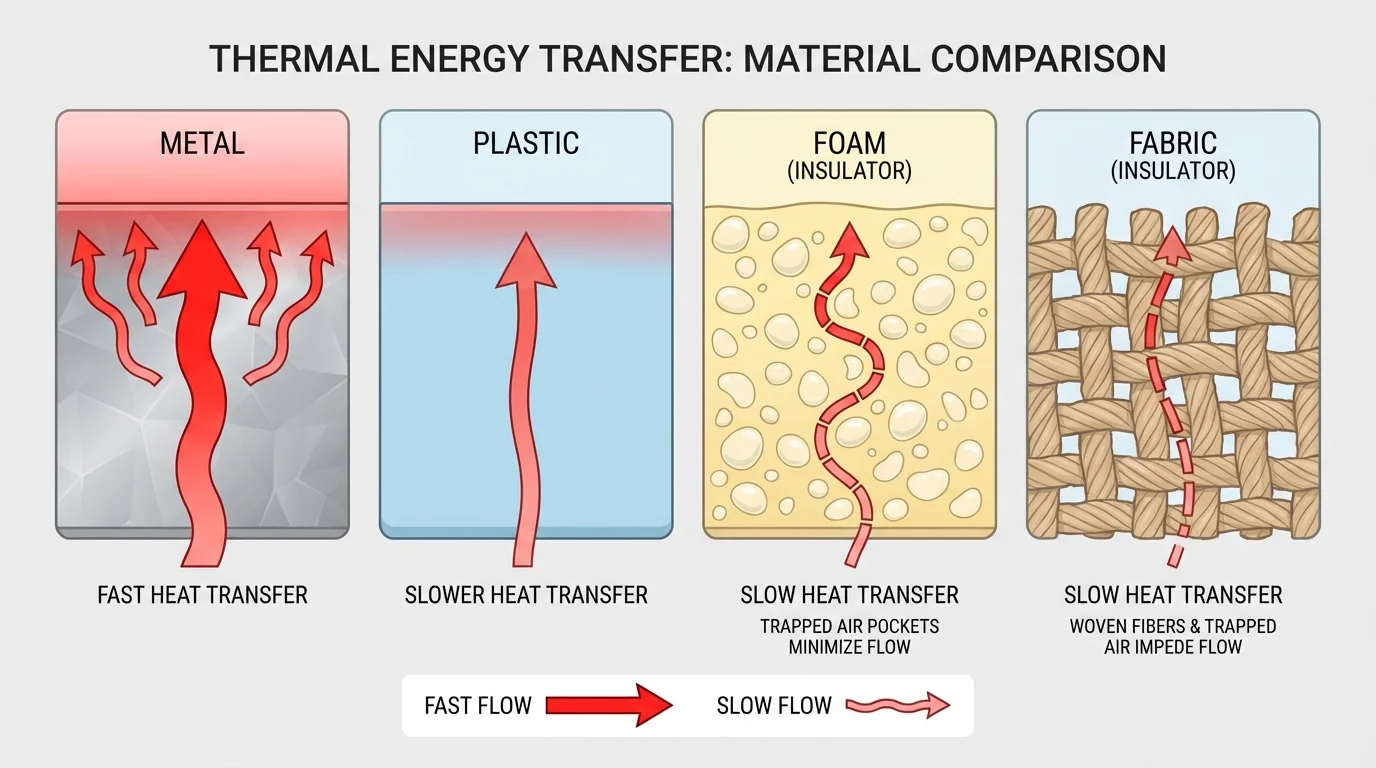
[Figure 2] illustrates how the material used in a device makes a huge difference. Some materials are strong insulators, which slow thermal energy transfer. Others are strong conductors, which allow energy to move more easily.
Metals such as aluminum, copper, and steel are usually good conductors. They are useful when you want energy to move quickly, such as in cookware or radiators. Materials like foam, wood, plastic, wool, and rubber are better insulators. They are useful when you want to slow transfer, such as in lunch boxes, oven mitts, or cooler walls.
Air can also act as an insulator when it is trapped in small spaces. That is why foam works well and why puffy jackets can keep people warm. The air pockets slow conduction and reduce convection. Multiple layers can make a design more effective because each layer adds resistance to energy transfer.
Other design features matter too. Thickness often affects how quickly thermal energy moves. A thicker insulated wall usually slows transfer more than a thin one. Surface color matters for radiation: dark surfaces absorb more radiant energy, while shiny or reflective surfaces reflect more. Surface area matters because more exposed area can increase energy transfer.
Shape and structure matter as well. A lid on a cup reduces convection by trapping warm air. A narrow opening lets less warm air escape than a wide opening. Fins on a heat sink increase surface area, allowing more energy to move into the air. The material comparison in [Figure 2] helps explain why engineers often combine several materials in one design instead of relying on only one.
| Design feature | If the goal is to minimize transfer | If the goal is to maximize transfer |
|---|---|---|
| Material | Use foam, plastic, fabric, wood | Use metal |
| Thickness | Increase insulation thickness | Use thinner conducting path when safe |
| Surface area | Reduce exposed area | Increase exposed area |
| Color and finish | Use reflective or shiny surfaces | Use dark surfaces when absorbing radiation is useful |
| Air movement | Block drafts, trap air, add lids | Allow airflow to carry energy away |
Table 1. Common design choices for reducing or increasing thermal energy transfer.
[Figure 3] shows that sometimes the best device is one that keeps temperatures from changing quickly. An insulated bottle, a house wall, a cooler, or a sleeping bag all work by reducing thermal energy transfer.
To minimize transfer, designers often choose insulating materials, add layers, trap air, reduce openings, and reflect radiation. A thermos is a great example because it combines several ideas at once. It may have insulated walls, a tight lid, and reflective inner surfaces. Each feature reduces a different path that thermal energy could take.
If you were designing a lunch container to keep soup warm, you would want a thick insulating wall, a snug lid, and materials that do not conduct energy quickly. If you were designing a shelter for a cold environment, you might include layers, trapped air, and a shape that reduces heat loss.
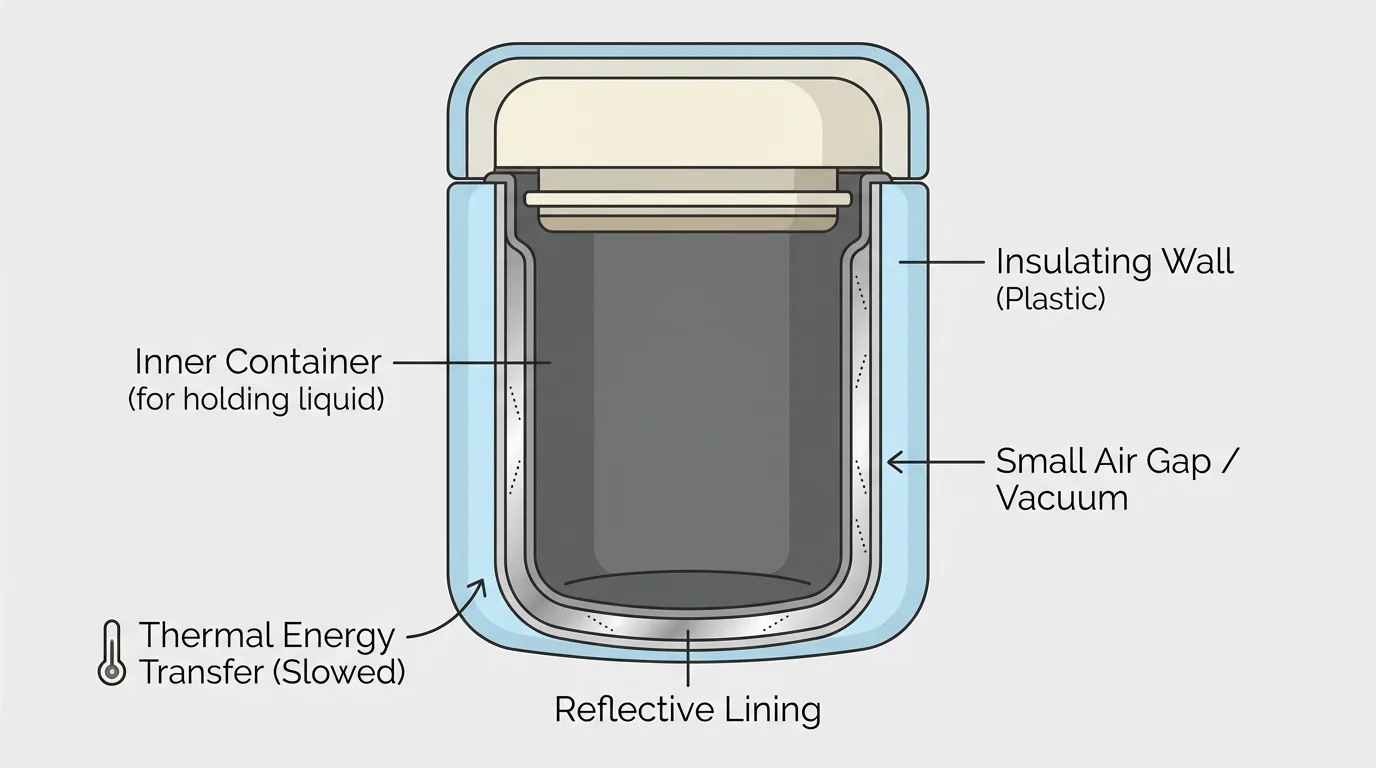
Minimizing transfer does not mean stopping it completely. No real device is perfect. Over time, thermal energy still moves from warmer to cooler areas. The goal is to slow that process enough to make the device useful.
Case study: Improving a hot-drink container
Step 1: Identify the problem
A plain paper cup lets a hot drink cool too quickly.
Step 2: Apply science ideas
Add a lid to reduce convection, use a sleeve to reduce conduction to the outside, and include a shiny inner layer to reduce radiation.
Step 3: Predict the result
The drink should stay warm longer because fewer paths are available for thermal energy to escape quickly.
This is an example of using evidence about conduction, convection, and radiation to improve a design.
When you look back at [Figure 3], notice that the best minimizing designs often use more than one strategy at once. That is common in engineering: combining simple ideas can create a much stronger solution.
[Figure 4] demonstrates that in other situations, a device should move thermal energy quickly. Cooking pans, radiators, heat sinks in electronics, and some solar water heaters are designed to maximize transfer.
To maximize transfer, designers often use conducting materials such as metal, increase contact between surfaces, increase surface area, and allow moving air or liquid to carry energy away. A metal pan works because thermal energy from the stove moves efficiently into the pan and then into the food.
Electronic devices offer another great example. A computer processor can become too warm during use. Engineers attach a metal heat sink with fins. The metal conducts energy away from the processor, and the fins increase surface area so the air can carry away more thermal energy.
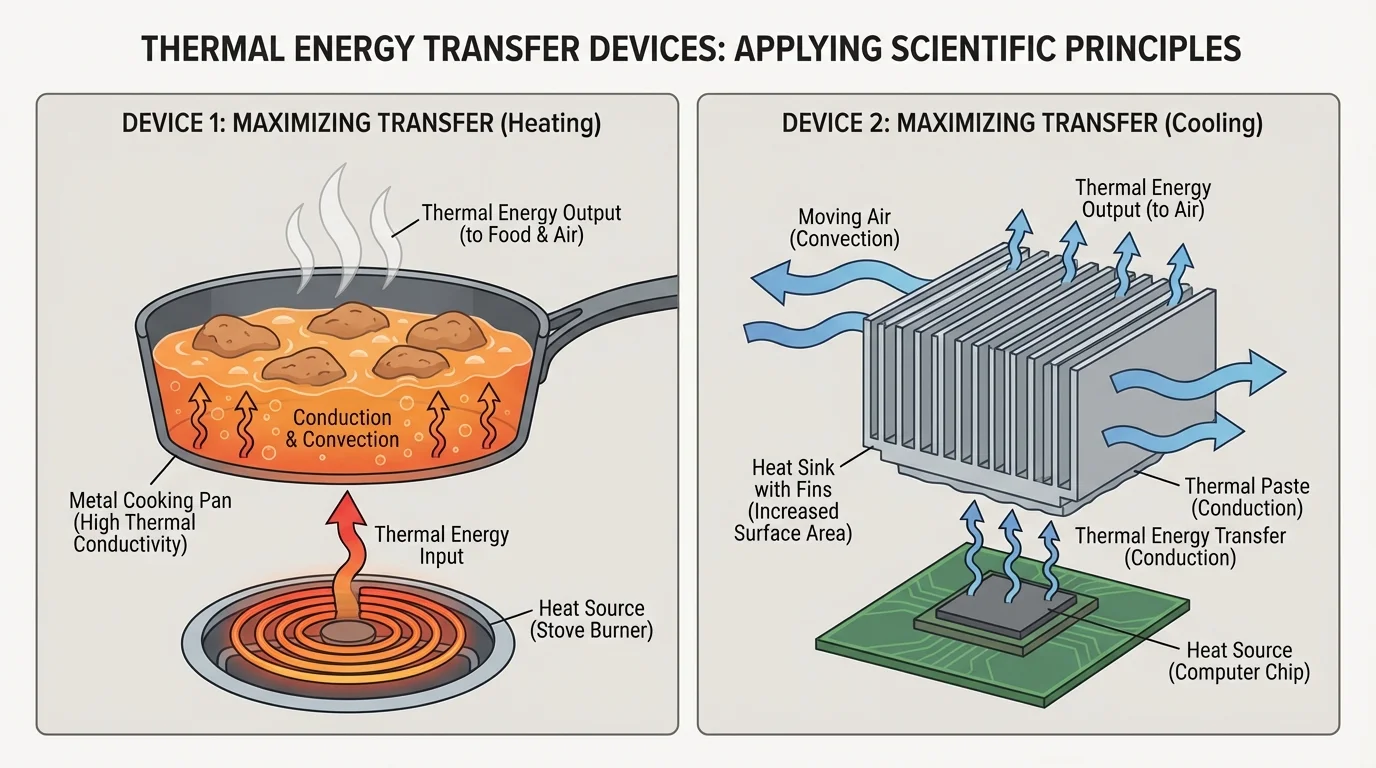
Maximizing transfer can also involve radiation. Dark surfaces can absorb more radiant energy, which is helpful in some solar heaters. In cold climates, black solar collector surfaces can warm water more effectively because they absorb more of the Sun's energy.
The devices in [Figure 4] show an important engineering idea: the same scientific principles can be used in opposite ways. A designer chooses materials and shapes based on whether the goal is to hold thermal energy in or move it out.
Spacecraft use both strategies at once. Some parts are covered with reflective materials to reduce radiation from the Sun, while other parts are designed to release extra thermal energy so equipment does not overheat.
That is why understanding the purpose of a device comes before building it. There is no single best thermal design for every situation. There is only the design best suited to the problem.
[Figure 5] lays out the process engineers use before building. They define the problem clearly and identify the criteria, which are the things the design must do well, and the constraints, which are the limits such as time, materials, size, or cost.
For example, suppose the goal is to design a device that keeps water cool for the longest time during one class period. Possible criteria might include keeping the temperature low, fitting on a desk, and not leaking. Constraints might include using only classroom materials such as paper, foil, cloth, tape, and plastic cups.
A good plan also identifies variables. The independent variable is what you change, such as the material wrapped around a cup. The dependent variable is what you measure, such as temperature after a certain amount of time. Controlled variables are the things kept the same, such as the amount of water, starting temperature, cup size, and test time.
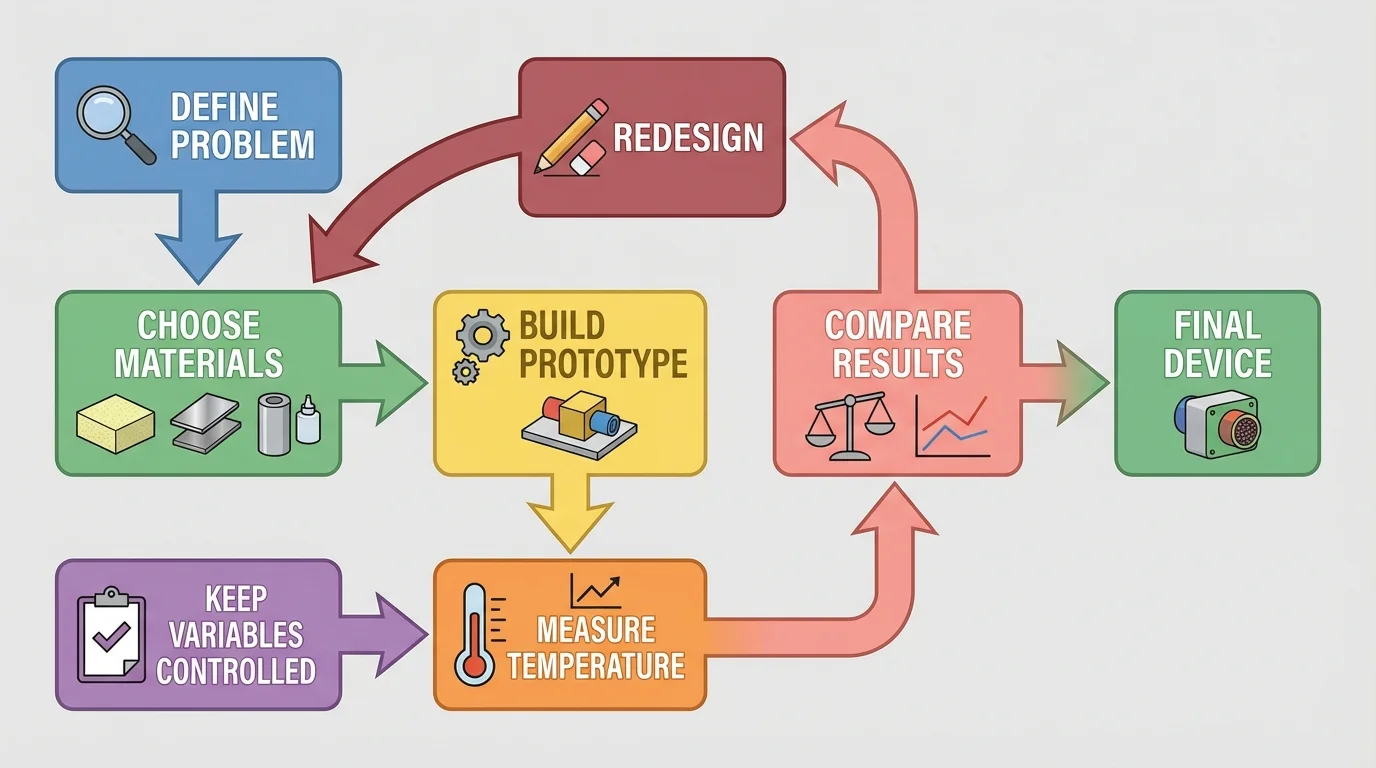
Fair testing is essential. If one cup starts with more water or a different starting temperature, then the comparison is not fair. Reliable evidence comes from changing one major factor at a time while keeping other important factors the same.
The flow in [Figure 5] also reminds us that design is usually a cycle, not a single attempt. Engineers test, analyze results, and redesign. A first prototype is rarely the final one.
When building a device, start with a clear sketch, a list of materials, and a reason for each design choice. If you use foil, explain whether it reflects radiation. If you use foam, explain that it contains trapped air and reduces conduction. If you use a lid, explain how it reduces convection.
Testing should produce evidence, not just opinions. A statement like "this one felt warmer" is weaker than a measured temperature comparison. A thermometer gives stronger evidence because it provides numerical data. For example, if one container starts at \(60 \,^\circ\textrm{C}\) and ends at \(48 \,^\circ\textrm{C}\) after \(20 \textrm{ min}\), while another ends at \(42 \,^\circ\textrm{C}\), the first container slowed cooling more effectively.
You can also compare temperature change. If the first design changes by \(60 - 48 = 12 \,^\circ\textrm{C}\) and the second changes by \(60 - 42 = 18 \,^\circ\textrm{C}\), the first design does a better job of minimizing thermal energy transfer during that test. This uses data to compare performance without calculating the total thermal energy transferred.
Testing a prototype fairly
Step 1: Keep important conditions the same
Use the same amount of water in each container, the same starting temperature, and the same test time.
Step 2: Change one main design feature
For example, compare a cup wrapped in fabric to a cup wrapped in aluminum foil.
Step 3: Measure and record
Take the final temperatures and compare which design keeps the water warmer or cooler, depending on the goal.
Step 4: Improve the design
If the first design is not effective enough, add a lid, increase thickness, or try a different material.
Testing and redesign are the heart of engineering.
Sometimes observations matter too. You might notice condensation, escaping steam, drafts, or warm outer surfaces. These clues can help explain why a design performed the way it did. A cup that feels warm on the outside may be losing thermal energy quickly through conduction.
Thermal design appears in many careers and technologies. Architects design buildings with insulation, reflective roofs, and sealed windows to reduce unwanted thermal energy transfer. Clothing designers choose fabrics that trap air or release heat depending on weather and activity. Food delivery companies use insulated bags to keep meals at safe temperatures.
In medicine, emergency blankets are often shiny because reflective surfaces reduce energy loss by radiation. In electronics, thermal paste and heat sinks help transfer energy away from important components. In transportation, engines need cooling systems to prevent overheating. These are all examples of designing with the same scientific principles.
Even nature provides ideas. Polar bear fur traps air. Desert animals often avoid the hottest parts of the day, reducing thermal energy gain from the environment. Penguins huddle together to reduce exposed surface area. Engineers often study natural systems and apply similar design ideas in human technology.
Science and engineering work together
Science explains why thermal energy moves and what factors affect it. Engineering uses that knowledge to solve problems by designing, testing, and improving devices. The strongest designs come from evidence, not guessing.
When you understand particle motion, types of thermal energy transfer, and material properties, you can explain why one design works better than another. That is what makes thermal design a scientific process rather than just trial and error.