A campfire, a loaf of bread in the oven, and a bicycle left out in the rain all have something important in common: the substances involved do not just look different afterward. Their properties can change in ways that reveal that entirely new substances have formed. Scientists often cannot decide by sight alone, so they compare evidence from before and after substances interact.
Every day, substances mix, heat up, cool down, burn, dissolve, and react. Some of these changes are only physical changes, which means the substance stays the same even if its form changes. Other changes are chemical reactions, which produce one or more new substances. Knowing the difference matters in cooking, medicine, manufacturing, environmental science, and safety.
If a solid candle wax melts, it is still wax. If the wax burns, new substances form. If salt dissolves in water, the salt can still be recovered, so that is not usually evidence of a chemical reaction. But if iron reacts with oxygen in moist air and forms rust, the new material has different properties from the original iron. The key question is this: Do the before-and-after properties show that a new substance formed?
Matter is anything that has mass and takes up space. Matter is made of tiny particles called atoms, and atoms can join to form molecules or other kinds of particle arrangements. When substances react chemically, atoms are rearranged into new combinations, but the atoms themselves do not disappear.
To answer the key question, scientists gather data. They may measure density, test whether a substance dissolves, compare melting point or boiling point, notice odor, or observe whether a substance is flammable. These properties help identify a substance, much like fingerprints help identify a person.
A substance is a kind of matter with a specific set of properties. Water, oxygen, sugar, and copper are all examples of substances. Because matter is made of atoms and molecules, each substance has its own structure, and that structure helps explain its properties. For example, water, written as \(\textrm{H}_2\textrm{O}\), has properties very different from oxygen gas, written as \(\textrm{O}_2\), even though both contain oxygen atoms.
Some properties can be observed directly, such as smell. Others can be measured. For example, density compares mass to volume and can be calculated using the formula \(\rho = \dfrac{m}{V}\), where \(\rho\) is density, \(m\) is mass, and \(V\) is volume. If a sample has a mass of \(20 \textrm{ g}\) and a volume of \(10 \textrm{ cm}^3\), then its density is \(\rho = \dfrac{20}{10} = 2 \textrm{ g/cm}^3\).
Chemical reaction is the process in which atoms are rearranged and new substances form.
Physical property is a characteristic of a substance that can be observed or measured without changing it into a different substance.
Chemical property is a characteristic that describes how a substance can form new substances. In this lesson, flammability is a chemical property used as evidence.
When scientists compare substances before and after interaction, they ask whether the material after the change has the same set of properties as before. If the properties stay the same, that suggests no new substance formed. If key properties are different, that is evidence that a chemical reaction may have occurred.
Particle models make this idea easier to understand, as [Figure 1] shows. In a physical change, the particles may spread out, move closer together, or change arrangement, but the substance itself remains the same. In a chemical reaction, atoms are rearranged into new particles, so the properties can change.
Think about ice melting. Solid water and liquid water are both \(\textrm{H}_2\textrm{O}\). The particles move differently, but no new substance appears. Now think about burning wood. The original materials in the wood react with oxygen from the air and produce new substances such as gases and ash. Those products have different properties from the starting materials.
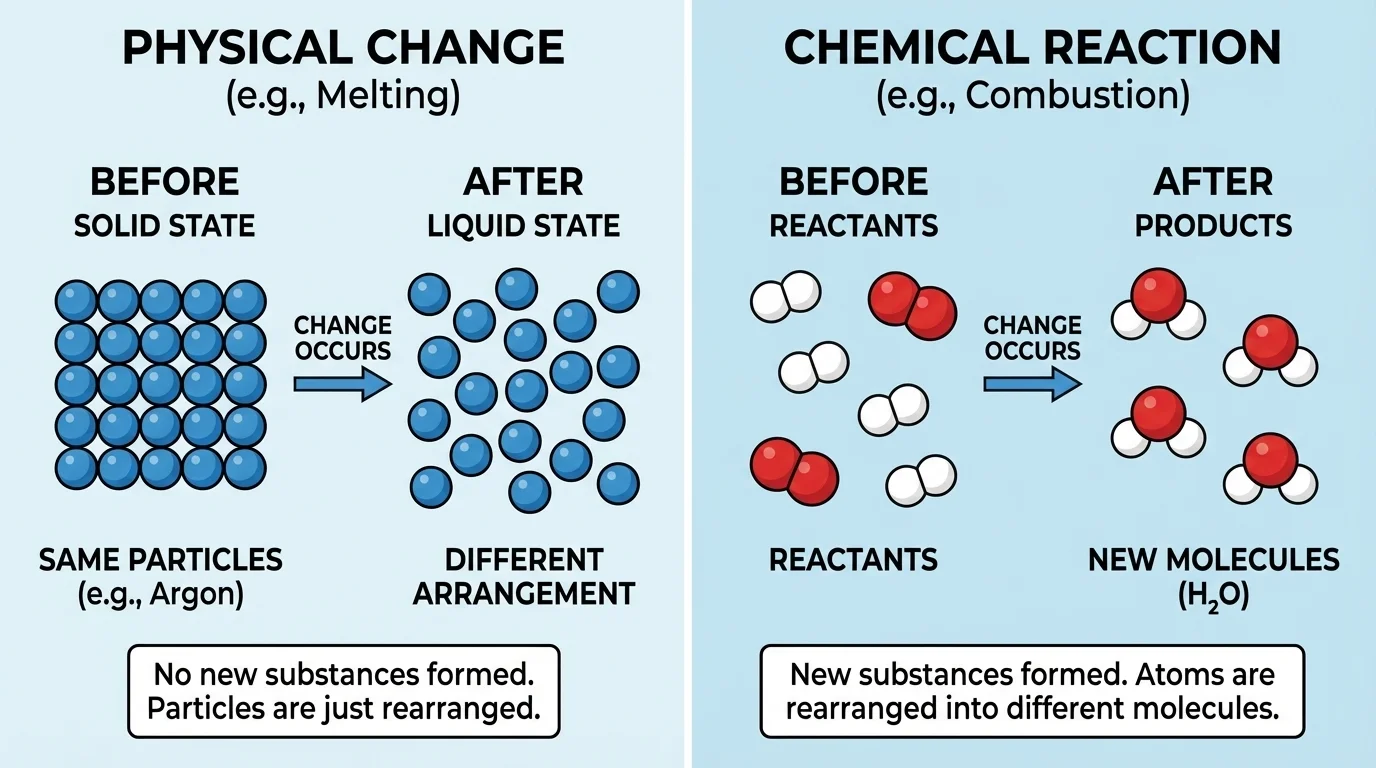
This difference explains why scientists focus on evidence from properties. A substance that changes only in size, shape, or state may still be the same substance. A substance that ends up with a different density, different melting point, different odor, or different flammability may be a new substance.
Two colorless substances can react and produce a substance with a noticeable odor or a different flammability. That is why scientists do not rely on color alone when identifying chemical change.
Later, when you examine data tables, remember the particle idea from [Figure 1]. New particle arrangements lead to new properties, and new properties are the evidence we can measure.
In this topic, scientists limit their analysis to six properties, as [Figure 2] summarizes: density, melting point, boiling point, solubility, flammability, and odor. These are enough to compare substances before and after they interact and decide whether a chemical reaction likely occurred.
Melting point is the temperature at which a solid becomes a liquid. Boiling point is the temperature at which a liquid becomes a gas throughout the liquid. Solubility describes how well a substance dissolves in another substance, often water. Flammability tells whether a substance can burn. Odor is the smell of a substance.
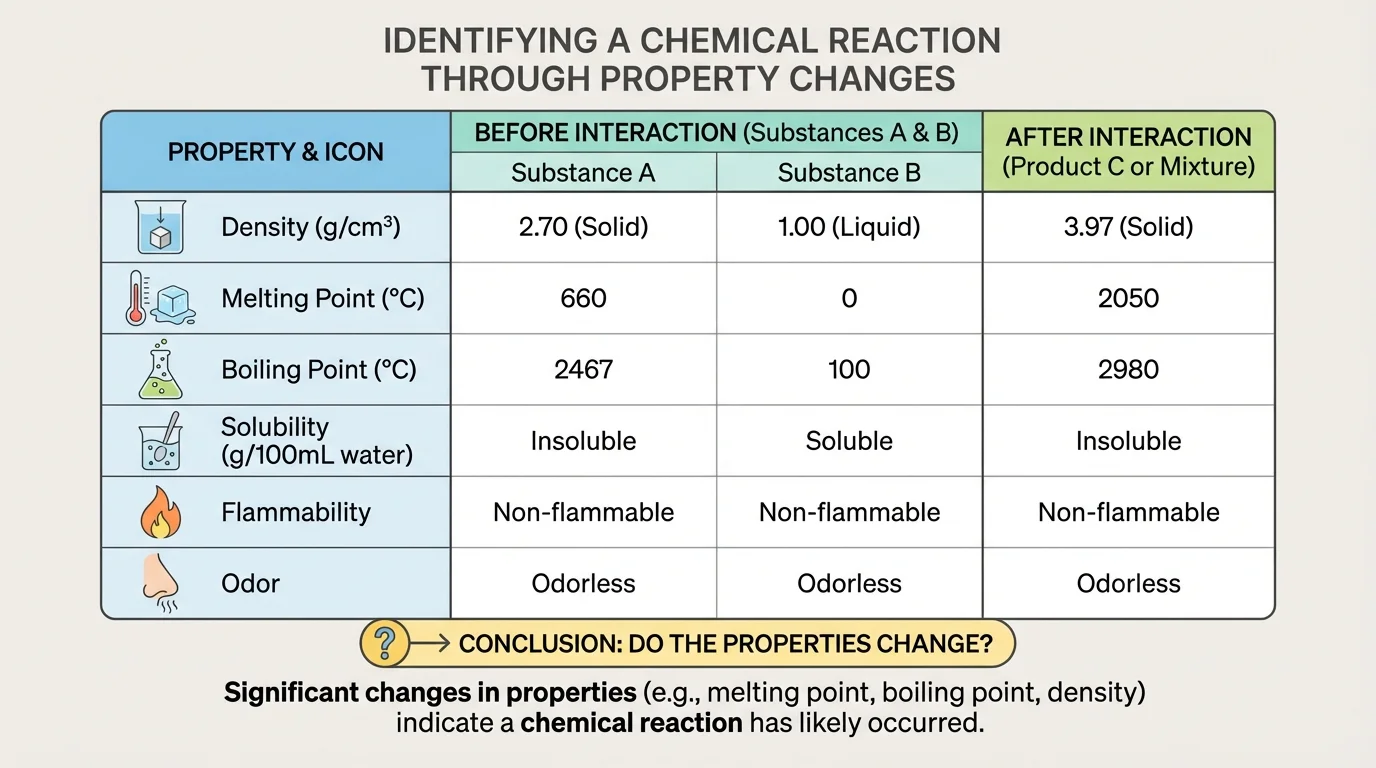
These properties are useful because they help identify substances. For example, pure water has a density close to \(1.0 \textrm{ g/cm}^3\) at room temperature, a melting point of \(0^\textrm{C}\), and a boiling point of \(100^\textrm{C}\) under normal air pressure. If a liquid sample has very different values, it is probably not pure water.
Not every investigation will use all six properties. Sometimes one or two properties are enough to suggest a conclusion. However, scientists prefer several pieces of evidence because a single measurement can be affected by error or by mixing substances without actually creating a new substance.
| Property | What it tells you | How it can help |
|---|---|---|
| Density | How much mass is packed into a given volume | Helps identify whether a material is the same before and after |
| Melting point | Temperature where solid changes to liquid | A different value may suggest a new substance |
| Boiling point | Temperature where liquid changes to gas | Can reveal whether the liquid is the same substance |
| Solubility | How well a substance dissolves | Changes can show different particle interactions |
| Flammability | Ability to burn | If flammability changes, chemical identity may have changed |
| Odor | Characteristic smell | A new odor can be evidence of a new substance |
Table 1. The six properties used in this lesson and how each one helps scientists identify substances.
Scientists do not guess. They follow a process, and [Figure 3] lays out that process clearly. First, record the properties of the starting substances. Next, record the properties of the materials after the interaction. Then compare the two sets of data and ask whether the final material has the same identifying properties or different ones.
If the after-data match the before-data, the evidence points toward no new substance. If the after-data are clearly different, the evidence supports a chemical reaction. The stronger the pattern of change across several properties, the stronger the conclusion.
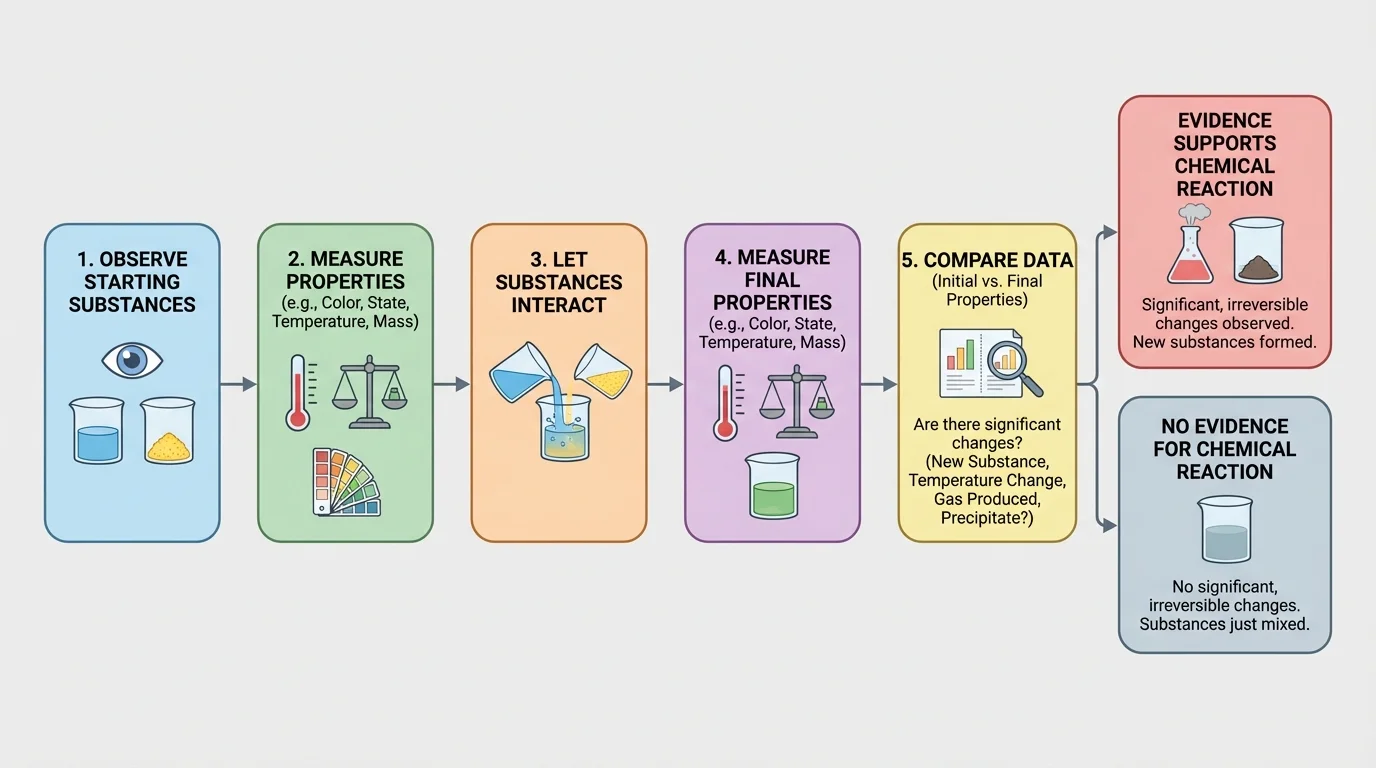
A useful habit is to separate observation from interpretation. An observation is what you measure or notice, such as "the liquid has an odor" or "the density changed from \(0.8 \textrm{ g/cm}^3\) to \(1.1 \textrm{ g/cm}^3\)." An interpretation is your conclusion, such as "a new substance probably formed." Good science keeps those two steps connected but distinct.
Why comparing data works
Each pure substance has a characteristic combination of properties because of the way its atoms and molecules are arranged. When a chemical reaction forms a new substance, the arrangement changes, so the set of properties can change too. That is why before-and-after comparisons are powerful evidence.
For example, suppose a liquid has a boiling point of \(78^\textrm{C}\) before an interaction and the final liquid boils at \(100^\textrm{C}\). That difference matters. It does not prove the exact identity by itself, but it shows the final substance is not the same liquid as the starting one.
Density: If the density after the interaction is different from the starting substance, that suggests the particles are arranged differently in the new material. For instance, a solid with density \(7.8 \textrm{ g/cm}^3\) that changes into a flaky solid with density \(5.2 \textrm{ g/cm}^3\) is probably not the same pure substance.
Melting point and boiling point: These temperatures act like identity checks. If a solid used to melt at \(232^\textrm{C}\) but the product melts at a much higher temperature, the product is likely a different substance. The same reasoning works for boiling point.
Solubility: A substance may dissolve well in water, poorly, or not at all. If the material after interaction behaves differently in water than the starting substance did, that is evidence of changed identity. But be careful: simply dissolving something does not always mean a chemical reaction happened. Salt dissolving in water changes the mixture, not the salt itself.
Flammability: Some substances burn easily, some do not burn, and some support burning differently. If a material that was flammable becomes nonflammable after interaction, or vice versa, that is strong evidence of a new substance. A piece of paper burns, but the ash left after burning is not flammable in the same way.
Odor: A new smell can be a clue that a new substance formed. For example, food batter and baked bread do not smell the same. The heating causes chemical reactions that create new substances with new odors. Odor alone is not enough for a full conclusion, but it is useful when combined with other data.
These ideas connect back to the particle model in [Figure 1]. If the particle arrangement changes enough to create a new substance, the measurable properties can change as well.
When scientists organize evidence in tables, patterns become easier to see, as [Figure 4] demonstrates. The goal is not to memorize every value but to compare the properties of the starting materials with the properties of the final material.
Example 1: Dissolving salt in water
Step 1: Look at the starting substances.
Salt, \(\textrm{NaCl}\), is a solid with no characteristic odor, and it is soluble in water. Water, \(\textrm{H}_2\textrm{O}\), is a liquid with its own density, melting point, and boiling point.
Step 2: Observe what happens after mixing.
The salt seems to disappear into the water, forming a solution. No new odor appears, and the salt can be recovered by evaporating the water.
Step 3: Interpret the data.
The evidence does not strongly support a chemical reaction. Dissolving changed the mixture, but it did not create a new substance from the salt.
This example is important because students often confuse dissolving with a chemical reaction. A solution can look very different from the starting materials, but appearance alone is not enough.
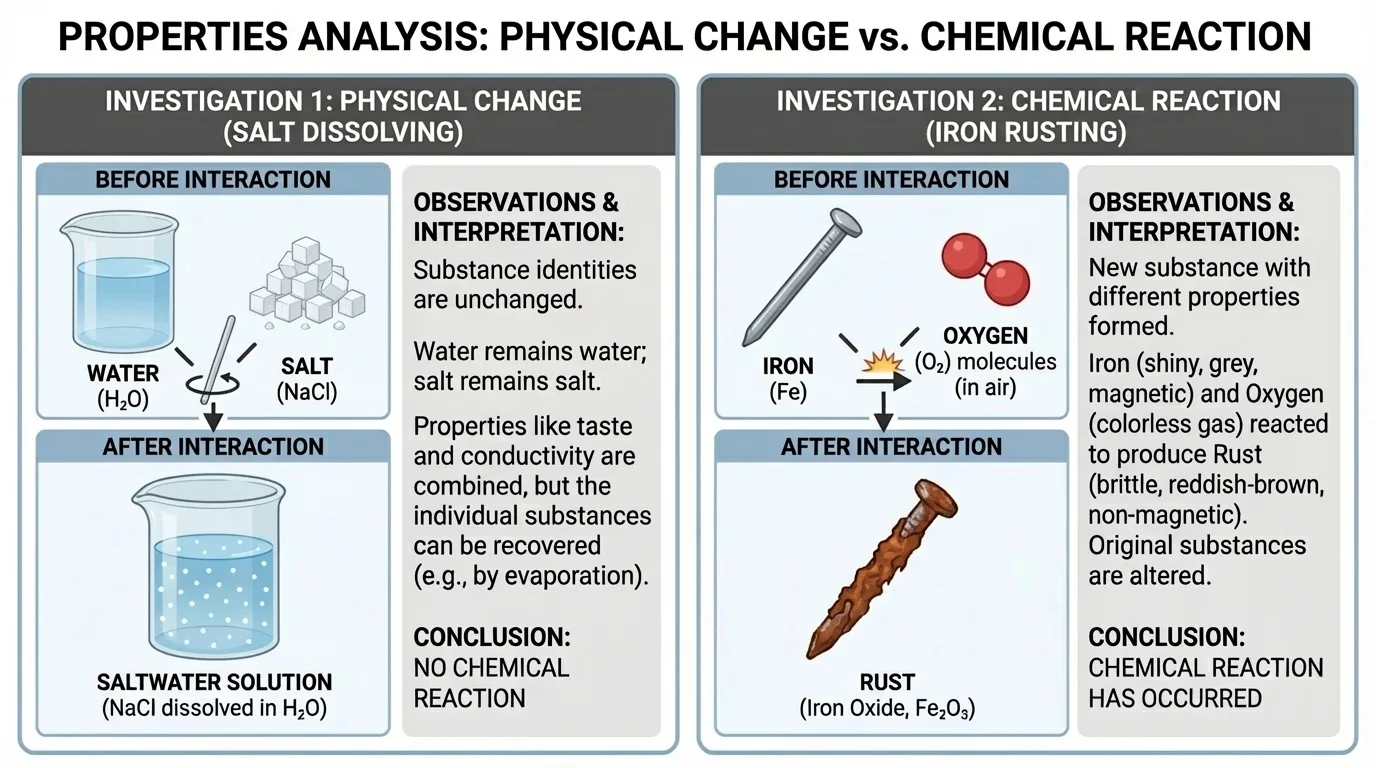
Example 2: Iron forming rust
Step 1: Record the starting substance.
Iron is a metal with its own density, odor, and other characteristic properties.
Step 2: Observe the final material after exposure to oxygen and moisture.
The reddish-brown rust has different properties from iron. Its density is different, it is more brittle, and it does not behave like the original metal.
Step 3: Interpret the data.
The changed properties support the conclusion that a chemical reaction occurred and a new substance formed.
Even though you may not know the exact formula of rust at this level, the property changes are enough to identify chemical change. This is why evidence matters more than guesswork.
| Investigation | Before interaction | After interaction | Conclusion |
|---|---|---|---|
| Salt + water | Salt is soluble; water remains water | Solution forms; salt can be recovered | Evidence does not strongly support a chemical reaction |
| Iron + oxygen + moisture | Iron has metal properties | Rust has different properties | Evidence supports a chemical reaction |
| Wax melts | Solid wax | Liquid wax with same substance identity | Physical change |
| Wax burns | Flammable wax | New gases and residue with different properties | Chemical reaction |
Table 2. Sample before-and-after investigations showing how property data help classify changes.
Science is strongest when conclusions match the evidence. Sometimes one property changes because of temperature, pressure, or mixing, not because a new substance formed. For example, melting changes state but not identity. That is why scientists look for patterns across multiple properties whenever possible.
Suppose a substance has a stronger odor after heating. That may mean new substances formed, but it could also mean the same substance evaporated more quickly and became easier to smell. If the odor changes and the flammability, melting point, or solubility also change, the evidence becomes much stronger.
Example 3: Using density data carefully
Step 1: Calculate the density of a starting sample.
A sample has mass \(30 \textrm{ g}\) and volume \(15 \textrm{ cm}^3\). Its density is \(\rho = \dfrac{30}{15} = 2 \textrm{ g/cm}^3\).
Step 2: Calculate the density of the final sample.
The final sample has mass \(33 \textrm{ g}\) and volume \(15 \textrm{ cm}^3\). Its density is \(\rho = \dfrac{33}{15} = 2.2 \textrm{ g/cm}^3\).
Step 3: Interpret carefully.
The density changed from \(2\) to \(2.2 \textrm{ g/cm}^3\). That suggests the material may be different, but a scientist would still want more evidence from other properties before making a strong conclusion.
The comparison chart in [Figure 4] helps show this idea: strong conclusions come from matching several observations, not from a single clue used by itself.
Food scientists study chemical reactions when bread bakes, onions cook, or milk spoils. The odor changes because new substances form. Engineers monitor materials for rust because new substances can weaken bridges, tools, and vehicles. Safety experts test flammability when designing clothing, furniture, and building materials.
Environmental scientists also analyze substances before and after interactions. For example, if a liquid spilled into water changes odor or flammability after reacting with air or sunlight, those property changes can help identify what happened. In industry, technicians compare boiling point and density to check whether a liquid is still pure or has changed into something else.
"Good conclusions come from good evidence."
— A core idea of scientific investigation
The big idea is that substances are made of atoms and molecules, and their properties come from how those particles are arranged. When substances interact chemically, atoms rearrange into new substances, and the new substances may have different density, melting point, boiling point, solubility, flammability, or odor. Those measurable changes are the clues scientists use to decide what happened.