Your shoes, backpack, water bottle, phone case, and even parts of a hospital syringe may seem completely different, but many of them share a surprising origin: they began as natural resources taken from Earth. People often hear the word synthetic and think it means "not natural at all." In science, though, synthetic materials are usually made by humans from substances that came from nature first. Understanding that connection helps us answer a bigger question: how can one set of natural resources lead to so many different materials that change how people live?
Natural resources are useful materials found in nature, such as water, air, trees, crude oil, natural gas, cotton, and minerals. People use these resources directly or change them into other materials. Wood from trees can become paper. Sand and minerals can become glass. Crude oil can be changed into many chemicals that are used to make plastics.
A synthetic material is a material that people design and make through chemical and physical processes. It is not usually found in the same form in nature. Nylon, polyester, many plastics, and some medicines are examples. These materials are made because scientists and engineers want certain properties such as strength, stretchiness, water resistance, or low cost.
Natural resource means a substance or feature from nature that people can use.
Synthetic material means a human-made material produced from natural substances through processing.
Property means a characteristic of a material, such as hardness, flexibility, absorbency, or transparency.
Not every human-used material is synthetic. Cotton cloth is made from cotton plants, so it comes from a natural source and remains mostly natural even after processing. Polyester cloth, however, is made by people through chemical reactions using substances that often come from petroleum. Both can become shirts, but their origins and properties differ.
That difference matters because the source of a material often affects how it is made, how it behaves, and what happens when people throw it away. A paper bag and a plastic bag may both carry groceries, but they come from different natural resources and can have different effects on forests, fossil fuel use, trash, and pollution.
All materials are made of matter, and matter is made of tiny particles called atoms. Atoms can join together to form molecules. The way atoms connect, the kinds of atoms present, and the shape of the molecules all help determine a material's properties, as shown in [Figure 1]. This idea helps explain why some materials are stiff, some are soft, some dissolve in water, and some do not.
For example, water is made of \(\textrm{H}_2\textrm{O}\), and table salt is made of \(\textrm{NaCl}\). Both are matter, but they have very different properties because their particles are arranged differently. In synthetic materials, scientists often build large molecules called polymers. A polymer is like a very long chain made from many smaller repeating parts. Some polymers slide past each other easily, making a material flexible. Others are packed or linked in ways that make a material stronger or more rigid.
When scientists create a new material, they are not making matter from nothing. They are rearranging atoms from natural resources into new combinations. That is a key science idea: the same kinds of atoms can be organized in different ways to make materials with very different properties.
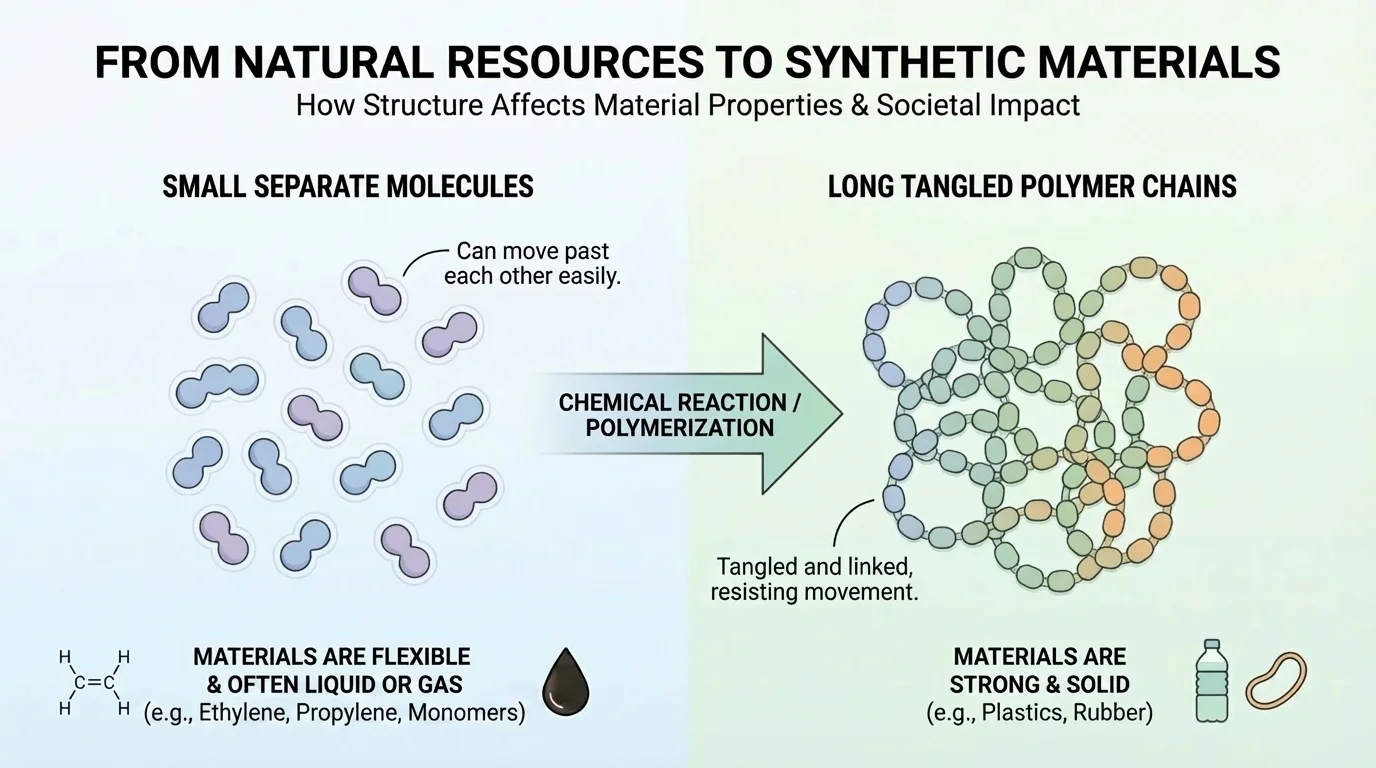
Think about plastic wrap and a hard plastic chair. Both are made from polymers, yet they feel very different. The difference comes from the kinds of molecules used and how those molecules are arranged. In the comparison in [Figure 1], long chains can tangle or line up in different ways, which helps explain why one polymer may stretch while another keeps its shape.
This is why material science is so powerful. By studying structure at the tiny scale of atoms and molecules, scientists can predict useful properties at the large scale we can see and touch. Middle school science often focuses on a simple but important idea: structure affects properties.
How structure changes what a material can do
If atoms are connected in one pattern, a substance may be brittle. If they are connected in long flexible chains, it may bend. If a material has particles that do not mix well with water, it may be water-resistant. Engineers use this relationship between structure and properties to select or invent materials for specific jobs.
Many synthetic materials can be traced back to a few major sources from nature, as [Figure 2] illustrates. One major source is petroleum, also called crude oil. Another is natural gas. These are fossil fuels formed over long periods from ancient living things. They contain many compounds rich in carbon and hydrogen, which chemists can separate and rebuild into useful substances.
Plants are also important sources. Corn, sugarcane, wood pulp, and vegetable oils can be used to make synthetic or semi-synthetic products. For instance, rayon is made from plant cellulose that has been chemically processed. Some newer plastics are made partly from plant materials instead of only from petroleum.
Minerals and salts from Earth also play roles in making synthetic materials. Ingredients for glass, cleaning products, and some medicines may come from mined minerals. Synthetic fertilizers use elements such as nitrogen, phosphorus, and potassium, but these materials are gathered, processed, and combined by people to support agriculture.
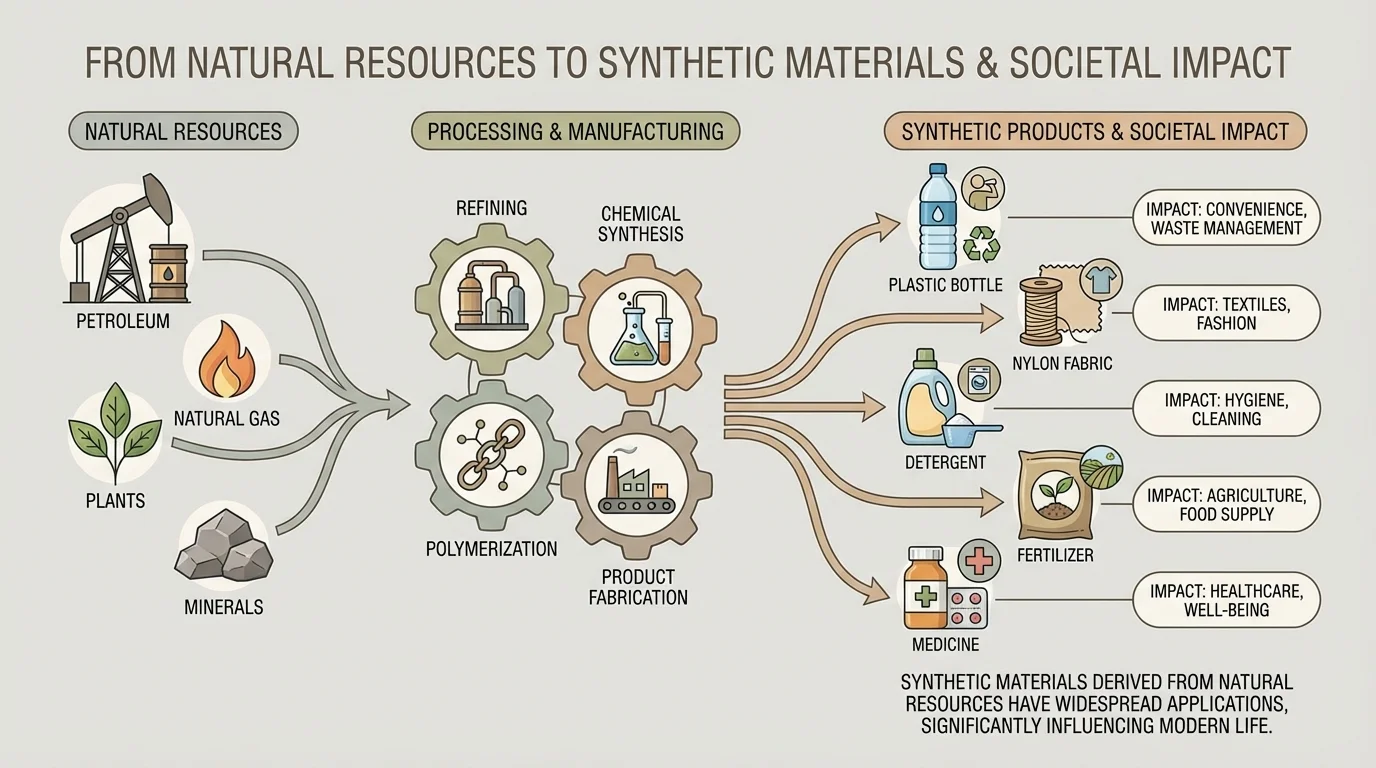
A useful way to make sense of this topic is to trace a product backward. A plastic water bottle may begin with petroleum or natural gas. A polyester jacket may begin with petroleum. A disposable diaper may include plastics from fossil fuels and absorbent materials from wood pulp. A toothbrush may combine plastic, synthetic bristles, and rubber-like materials from different natural resources.
This tracing process helps students gather evidence instead of guessing. If a package says "made from recycled plastic," that tells you something about its current ingredients, but the original plastic still came from natural resources at some earlier stage. If a label says "plant-based," it suggests that some part of the material came from crops rather than only fossil fuels.
Science is not only about memorizing facts. It is also about gathering information, checking sources, and deciding what the evidence supports. To understand synthetic materials, students can examine product labels, manufacturer descriptions, diagrams of production, recycling symbols, and reliable science articles.
Suppose you compare a cotton T-shirt, a polyester sports shirt, and a blended fabric shirt. The label might say "100% cotton," "100% polyester," or "60% cotton, 40% polyester." From that information, you can infer that the shirts come from different sources and likely have different properties. Cotton may absorb sweat well. Polyester may dry faster. A blended fabric combines properties from both.
Case study: tracing a running shoe
A running shoe is a good example because it contains several materials with different origins.
Step 1: Identify visible parts
The outer fabric, foam sole, rubber tread, laces, and glue are different materials.
Step 2: Match materials to likely sources
The fabric may be polyester, the foam may be a synthetic polymer, the tread may be synthetic rubber, and the glue may contain synthetic chemicals. Many of these come from petroleum or natural gas.
Step 3: Infer why each material was chosen
Polyester can be light, foam can cushion impact, and rubber can improve grip.
Step 4: Consider impact
The shoe is useful and durable, but mixed materials can make recycling difficult.
This example shows how evidence about structure, source, and use can be connected to explain a real object.
When gathering information, it is important to ask good questions: What is this made of? Where did those ingredients come from? Why was this material chosen? What happens after people are done using it? These questions help turn simple observations into scientific explanations.
Reliable information matters. An advertisement may say a product is "green" or "eco-friendly," but scientists look for evidence. Does the material come from renewable plants? Does it break down easily? Can it be recycled? Does making it use less energy or produce less pollution? Good science depends on comparing claims with evidence.
Plastic is one of the most common groups of synthetic materials. Plastics are used in bottles, containers, toys, car parts, keyboards, and medical devices. They are popular because they can be lightweight, moldable, strong, and often inexpensive. But "plastic" is a broad category, not just one material. Different plastics have different properties and uses.
Synthetic fibers include nylon, polyester, acrylic, and spandex. These are used in clothing, ropes, backpacks, carpeting, and sports equipment. Athletes often wear polyester because it can dry quickly. Spandex can stretch and return to shape, which is useful in sportswear and dance clothing.
Synthetic rubber is used in tires, seals, shoe soles, and gloves. It can be designed to resist heat, wear, or chemicals better than some natural rubber in certain situations. That makes it valuable in transportation and industry.
Other synthetic products include detergents, some dyes, many medicines, paints, fertilizers, and adhesives. Even when a final product is not entirely synthetic, synthetic ingredients can still play important roles. A smartphone, for example, contains glass, metals, ceramics, and many polymers in wiring, coatings, and protective parts.
| Material | Main natural source | Common use | Useful property |
|---|---|---|---|
| Polyethylene plastic | Petroleum or natural gas | Bags, bottles, containers | Lightweight and water-resistant |
| Polyester | Usually petroleum | Clothing, backpacks | Durable and quick-drying |
| Nylon | Usually petroleum | Ropes, fabric, gears | Strong and flexible |
| Rayon | Wood pulp | Clothing, fabric | Soft and absorbent |
| Synthetic rubber | Petroleum | Tires, soles, seals | Elastic and wear-resistant |
Table 1. Examples of synthetic materials, their major natural-resource sources, common uses, and useful properties.
Some synthetic materials are designed for very specialized jobs. The same basic idea of arranging atoms into useful structures can lead to a soft contact lens, a bullet-resistant fiber, or a lightweight part for a spacecraft.
Synthetic materials have changed society in major ways. In medicine, sterile plastic syringes, IV bags, artificial joints, sutures, and protective gloves help doctors and nurses care for patients safely. Many medicines are synthetic or partly synthetic, allowing scientists to design substances that fight disease or relieve pain.
In transportation, synthetic materials help make cars, bikes, and airplanes lighter. Lighter vehicles often use less fuel. In communication and technology, polymers insulate wires, protect screens, and form parts inside computers and phones. In sports, synthetic materials improve helmets, running shoes, rackets, and weather-resistant clothing.
Food storage is another important example. Plastic wraps and containers can help keep food clean and reduce spoilage. Fertilizers and packaging support farming and transport systems that feed large populations. Society benefits when materials are matched carefully to human needs.
Properties help explain uses. A material chosen for a raincoat should resist water. A material chosen for a window should let light through. A material chosen for a bicycle helmet should absorb impact. Engineers always connect a material's properties to its function.
These benefits are part of why synthetic materials became so widespread. They can often be produced in large amounts, shaped into many forms, and tailored for specific tasks. That flexibility has helped modern societies grow, build, and solve problems.
A material's impact includes its whole life cycle, from resource extraction to manufacture, use, and disposal, as [Figure 3] shows. Synthetic materials improve lives, but they can also create serious problems. Extracting petroleum and natural gas can disturb habitats and contribute to pollution. Factories that produce materials may use large amounts of energy and create waste.
Disposal is a major issue. Some synthetic materials last a very long time before breaking down. That can be useful during use, because the item is durable, but it can become a problem after disposal. Plastic waste in landfills, rivers, and oceans can harm wildlife and damage ecosystems.
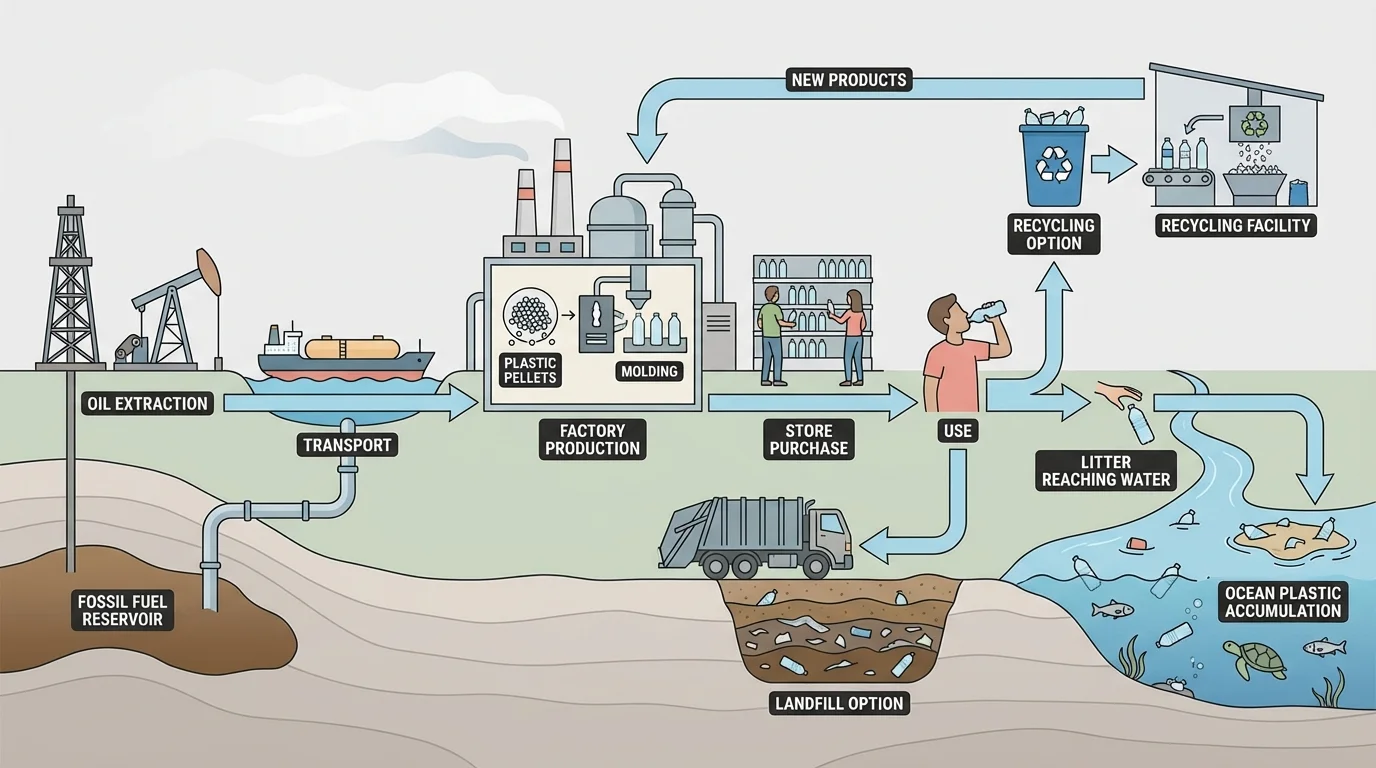
Very small pieces called microplastics can form when larger plastic items break into tiny fragments. These can be found in water, soil, and even the air. Scientists are still studying how microplastics affect living things and human health, but their presence shows that the story of a material does not end when someone throws it away.
There are also social impacts. Some communities live near extraction sites, factories, or landfills and may face more pollution than others. That means the benefits and burdens of synthetic materials are not always shared equally. Science helps identify these problems, and society must decide how to respond.
Looking back to [Figure 3], the same plastic bottle can represent convenience, sanitation, resource use, transportation, recycling challenges, and pollution risk. A single object can have both helpful and harmful effects depending on how it is made, used, and managed.
Because synthetic materials are so useful, the goal is not simply to reject them all. A better goal is to make smarter choices. People can reduce unnecessary waste, reuse products when possible, recycle materials that can be processed again, and support designs that use fewer resources.
Scientists and engineers are developing new materials that may be safer or easier to manage. Some bioplastics are made partly from plants. Some products are designed so their parts can be separated more easily for recycling. Researchers also look for ways to make materials strong and useful while producing less pollution.
When comparing old and new materials, it is important to stay evidence-based. A plant-based material is not automatically harmless, and a synthetic material from fossil fuels is not automatically the worst choice in every case. The best decision depends on the full picture: source, properties, use, durability, and disposal.
Why society needs both science and responsibility
Science explains where materials come from and why they act the way they do. Responsibility means using that knowledge to make choices that protect health, conserve resources, and reduce harm. Good decisions about materials depend on both accurate evidence and thoughtful values.
By gathering information and making sense of it, students can do what scientists do: connect small-scale ideas about atoms and molecules to large-scale questions about people and the planet. A synthetic material is never just "fake stuff." It is a human-made result of natural resources, scientific knowledge, engineering design, and social choices.