A skateboarder racing down a ramp, a stretched rubber band ready to snap forward, and a cup of soup cooling on a table may seem unrelated. But each one involves energy. Energy is one of the biggest ideas in science because it helps explain motion, change, heating, cooling, and many everyday events. When scientists study energy, they ask not only how much energy there is, but also what form it is in and where it is stored.
Energy is the ability to cause change. It can make things move, warm up, light up, stretch, bend, or change state. Some energy is easy to notice, like a speeding soccer ball. Other energy is stored and less obvious, like the energy in a book held high above the floor or in particles moving inside hot tea.
One reason energy is so useful in science is that it connects very different situations. The same ideas help explain why a roller coaster speeds up on the way down, why a hammer hits harder when swung faster, and why steam can burn more severely than warm water. To understand these situations, we need to look carefully at motion energy, stored energy, and thermal ideas related to temperature.
Kinetic energy is the energy an object has because it is moving.
Potential energy is stored energy in a system, often due to the relative positions of objects or the arrangement of parts in the system.
Temperature is a measure of the average kinetic energy of the particles in matter.
These definitions are connected. A moving bicycle has kinetic energy. A bicycle at the top of a hill has stored energy because of its position relative to Earth. And the metal frame of the bicycle contains particles that are always moving, with their average motion related to temperature.
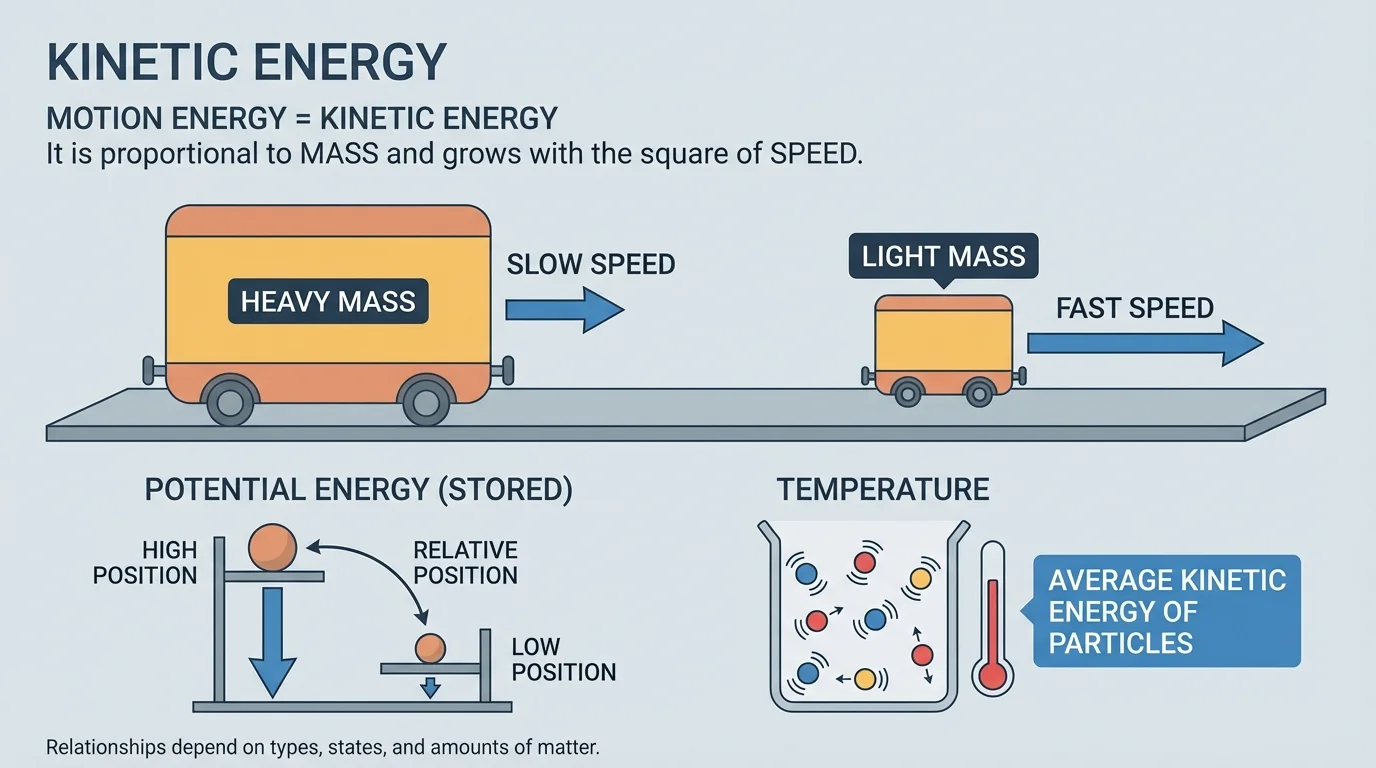
The kinetic energy of an object depends on how much matter it has and how fast it is moving, as [Figure 1] illustrates. In science, the amount of matter in an object is measured by its mass. A bowling ball rolling across the floor and a tennis ball rolling at the same speed do not have the same kinetic energy. The bowling ball has more because it has more mass.
Speed also matters, and it matters a lot. In fact, kinetic energy grows with the square of speed. That means if speed doubles, kinetic energy becomes four times as great. If speed triples, kinetic energy becomes nine times as great. This is why fast-moving objects can be much more dangerous than slow-moving ones, even if their mass stays the same.
The relationship is written as
\[KE = \frac{1}{2}mv^2\]
Here, \(KE\) is kinetic energy, \(m\) is mass, and \(v\) is speed. You do not need to memorize every detail of the formula to understand the big idea: more mass means more kinetic energy, and more speed means much more kinetic energy.
Example: How speed changes kinetic energy
Suppose a toy car has a mass of \(2 \textrm{ kg}\).
Step 1: Find the kinetic energy when the speed is \(3 \textrm{ m/s}\).
Substitute into the formula: \(KE = \dfrac{1}{2}(2)(3^2)\).
Since \(3^2 = 9\), we get \(KE = 1 \cdot 9 = 9 \textrm{ J}\).
Step 2: Find the kinetic energy when the speed is \(6 \textrm{ m/s}\).
Now use \(KE = \dfrac{1}{2}(2)(6^2)\).
Since \(6^2 = 36\), we get \(KE = 1 \cdot 36 = 36 \textrm{ J}\).
When the speed doubles from \(3 \textrm{ m/s}\) to \(6 \textrm{ m/s}\), the kinetic energy increases from \(9 \textrm{ J}\) to \(36 \textrm{ J}\), which is four times as much.
This squared relationship helps explain many real events. A car traveling twice as fast does not just have twice the motion energy. It has four times the kinetic energy, which means brakes must do much more work to stop it safely.
It is tempting to think mass and speed affect kinetic energy in the same way, but they do not. If mass doubles while speed stays the same, kinetic energy doubles. If speed doubles while mass stays the same, kinetic energy quadruples. Speed has the stronger effect.
Think about sports. A baseball and a shot put may move at similar speeds, but the shot put has far more kinetic energy because its mass is much larger. On the other hand, a small object moving extremely fast can also carry a lot of kinetic energy because speed is squared.
Why the square of speed matters
Squaring a number means multiplying it by itself. So if speed changes from \(2\) to \(4\), the squared part changes from \(2^2 = 4\) to \(4^2 = 16\). That is why increases in speed produce such large increases in kinetic energy.
Later, when we think about collisions, stopping distance, or why helmets and airbags matter, the idea from [Figure 1] stays important: kinetic energy depends on both mass and speed, but speed changes can have especially powerful effects.
Objects can also be part of a system that stores energy because of the relative positions of its parts, as [Figure 2] shows. This stored energy is called potential energy. A key idea is that potential energy is not just "inside one object." It belongs to the system of objects interacting with one another.
For example, a book on a shelf has gravitational potential energy because of its position relative to Earth. If the book falls, that stored energy can change into kinetic energy. The higher the book is above the floor, the more gravitational potential energy the Earth-book system has.
Another example is a stretched rubber band. When you stretch it, you do work on the rubber band system and store energy in it. A compressed spring also stores energy. In both cases, the arrangement of the system changes, and that change in position or shape stores energy.
Gravitational potential energy near Earth can often be described by
\(PE = mgh\)
Here, \(PE\) is gravitational potential energy, \(m\) is mass, \(g\) is the strength of gravity near Earth, and \(h\) is height. For middle school science, the most important pattern is simple: more mass and more height mean more gravitational potential energy.
Example: Height and stored energy
A \(2 \textrm{ kg}\) backpack is lifted to a shelf \(3 \textrm{ m}\) high. Using \(g \approx 9.8 \textrm{ m/s}^2\):
Step 1: Write the formula.
\(PE = mgh\)
Step 2: Substitute the values.
\(PE = (2)(9.8)(3)\)
Step 3: Calculate.
\(2 \cdot 9.8 = 19.6\), and \(19.6 \cdot 3 = 58.8\).
\[PE = 58.8 \textrm{ J}\]
The lifted backpack has \(58.8 \textrm{ J}\) of gravitational potential energy relative to the lower level.
Potential energy is easier to understand when you focus on relationships. A climber high on a mountain has more gravitational potential energy relative to the ground below. A charged battery stores energy in a different way, but the same general idea applies: energy can be stored and later transferred or transformed.
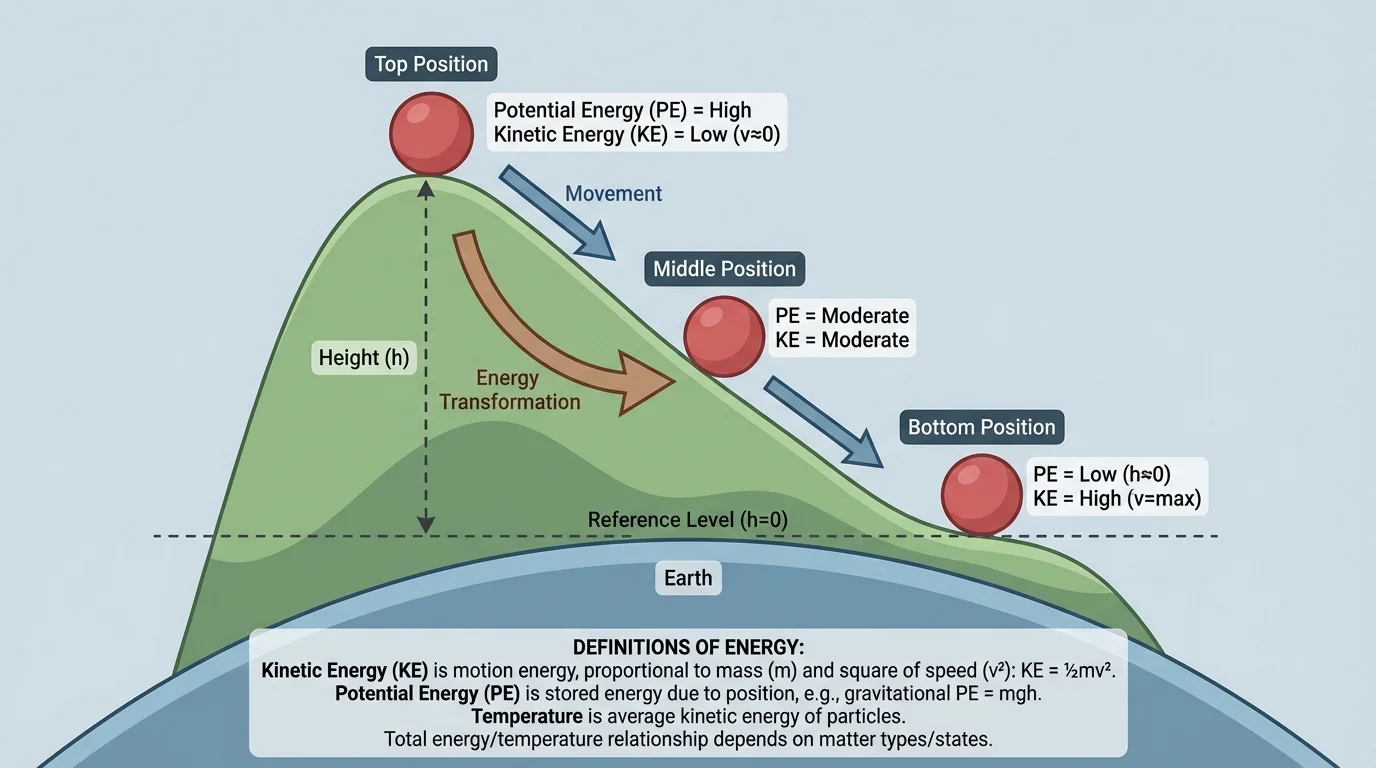
In many situations, kinetic and potential energy change back and forth. A roller coaster is a classic example. At the top of a hill, the coaster has a lot of gravitational potential energy. As it moves downward, that stored energy changes into kinetic energy, so the coaster speeds up. On the next climb, some kinetic energy changes back into potential energy.
A swinging pendulum works in a similar way. At the highest point of its swing, it has more gravitational potential energy and less kinetic energy. At the lowest point, it has more kinetic energy and less gravitational potential energy. Energy changes form within the system as the motion continues.
Real systems are not perfectly simple. Friction and air resistance often change some of the energy into thermal energy. That is why a rolling ball slows down and why brakes get hot. Energy is still conserved, but it may spread into forms that are less obvious than motion or height.
Engineers who design roads, vehicles, and protective gear pay close attention to kinetic energy. A small change in speed can produce a much bigger change in the energy involved in a crash.
The hill diagram in [Figure 2] helps explain why downhill motion speeds up: stored energy related to height can become motion energy as the object descends.
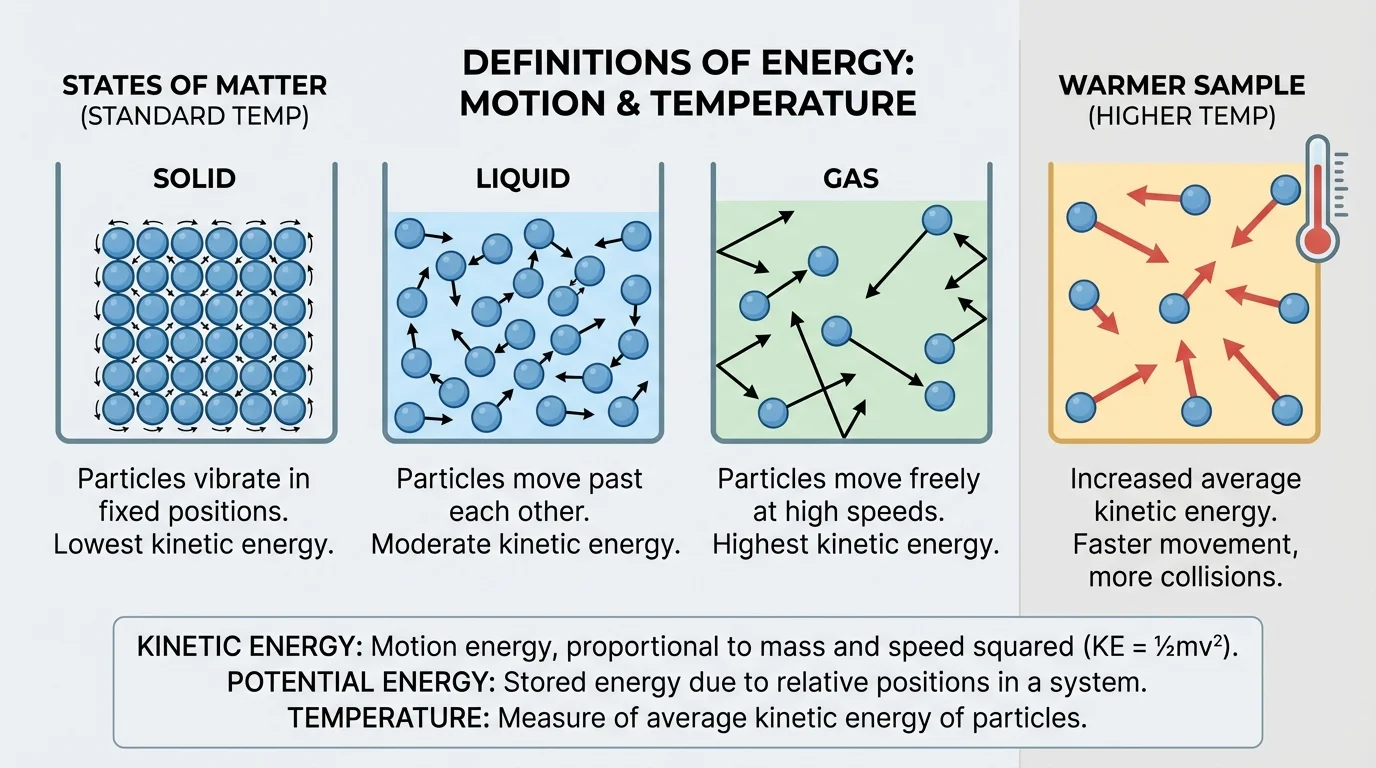
Temperature is not just a number on a thermometer. It is a measure of the average kinetic energy of the particles in matter, as [Figure 3] illustrates. Matter is made of tiny particles such as atoms and molecules, and these particles are always moving. In a solid, particles vibrate in place. In a liquid, they slide past one another. In a gas, they move more freely and rapidly.
When a substance gets hotter, its particles usually move faster on average. That means their average kinetic energy increases. When a substance cools, the average kinetic energy of its particles decreases.
This idea explains why heat can flow from a warmer object to a cooler one. If you place a metal spoon in hot soup, energy transfers from the faster-moving particles in the soup to particles in the spoon. As a result, the spoon becomes warmer.
Temperature is about an average. Not every particle moves at exactly the same speed. Some move faster and some slower, but temperature tells us about the average kinetic energy across huge numbers of particles.
Particles are too small to see directly with your eyes, but the particle model helps explain many observations, including melting, evaporation, and the way perfumes spread through air.
The particle picture from [Figure 3] also helps show why a hot gas and a hot liquid do not behave in exactly the same way. The particles are arranged differently and move differently depending on the state of matter.
Students often confuse temperature with total energy, but they are not the same thing. Temperature measures average kinetic energy of particles. Thermal energy, often used in middle school science to mean the total internal energy related to particle motion and interactions, depends on more than just temperature.
The total energy of a system depends on the types of matter present, the states of matter present, and the amounts of matter present. This is why a bathtub of warm water can contain more total thermal energy than a small cup of very hot water, even if the cup has the higher temperature.
Amount matters because more matter means more particles. If two samples of the same substance are at the same temperature, the larger sample usually has more total thermal energy because there are more particles contributing to the total.
Type matters because different substances respond differently when energy is added or removed. For example, water and metal can be at the same temperature, but they may store and transfer energy differently. This is one reason a metal bench can feel cooler to the touch than a wooden bench on the same winter day, even when both are at nearly the same temperature.
State matters because particles in solids, liquids, and gases are arranged and interacting in different ways. Changing state, such as melting ice or boiling water, involves energy changes that are not just about making particles move faster. Energy can also go into changing how particles are arranged relative to one another.
| Situation | Temperature | Total energy idea |
|---|---|---|
| Small cup of hot water | High | May have less total thermal energy because there is less water |
| Large bathtub of warm water | Lower than the cup | May have more total thermal energy because there is much more water |
| Hot metal pan | High | Transfers energy quickly to your hand |
| Ice at \(0^\circ\textrm{C}\) | Same as liquid water at \(0^\circ\textrm{C}\) | Has a different particle arrangement and energy distribution because the state is different |
Table 1. Comparisons showing that temperature and total energy are related but not identical.
Temperature versus total energy
A higher temperature means particles have greater average kinetic energy. It does not automatically mean the sample has more total energy than every cooler sample. Total energy depends on how many particles there are, what substance they make up, and whether the matter is solid, liquid, or gas.
This is why scientists must be careful with words. Saying something is "hotter" tells you about temperature. It does not fully tell you how much total energy the whole system contains.
These ideas appear everywhere. In transportation, seat belts and airbags reduce injury by helping manage changes in kinetic energy during a crash. In sports, coaches teach athletes to control speed and body position because both affect energy and motion. In construction, engineers think about gravitational potential energy when designing elevators, cranes, and tall buildings.
Cooking depends strongly on particle motion and energy transfer. Water on a stove heats up as energy is transferred into it, increasing the average kinetic energy of particles until boiling occurs. Baking, frying, and steaming all involve differences in temperature, total energy, and how substances transfer energy.
Weather also connects to these ideas. Warm air and cool air behave differently because the particles in the warmer air have greater average kinetic energy. This affects air movement, cloud formation, and storms.
Example: Same temperature, different total energy
Compare two containers of water that are both at \(40^\circ\textrm{C}\). One contains \(200 \textrm{ mL}\), and the other contains \(2{,}000 \textrm{ mL}\).
Step 1: Compare temperature.
Both samples have the same temperature, so their particles have the same average kinetic energy.
Step 2: Compare amount of matter.
The \(2{,}000 \textrm{ mL}\) sample has ten times as much water as the \(200 \textrm{ mL}\) sample.
Step 3: Draw the conclusion.
Because there are more particles in the larger sample, it has more total thermal energy even though the temperature is the same.
This example shows why temperature alone does not tell the whole energy story.
The particle ideas from [Figure 3] help explain these applications. Faster average particle motion means higher temperature, but the total energy still depends on how many particles there are and what kind of substance they form.
One common misunderstanding is thinking that kinetic energy is just "motion" without any pattern. In fact, kinetic energy follows a clear relationship with mass and speed. Another misunderstanding is believing that potential energy is stored in a single object all by itself. It is better to think of it as energy in a system of interacting objects.
A third misunderstanding is thinking that temperature tells you the total energy of a sample. It does not. A spark from a fire can be much hotter than a bathtub of warm water, but the bathtub contains far more total thermal energy because it has so much more matter.
Once these ideas are clear, many science topics make more sense: phase changes, heat transfer, collisions, motion, weather, and even why different materials feel different to the touch.