The periodic table is one of the most powerful predictive tools in science. From a single element's position, a chemist can make good predictions about whether it is shiny or dull, reactive or mostly unreactive, likely to lose electrons or gain them, and what kinds of compounds it tends to form. That is remarkable because the table is not just a list of substances. It is a model of atomic structure, built around repeating patterns in the electrons found in the outermost energy level of atoms.
The periodic table organizes elements by atomic number, which is the number of protons in the nucleus. As atomic number increases, electrons are added in patterns. Those patterns are not random. Because chemical behavior depends strongly on the electrons farthest from the nucleus, the arrangement of the table lets us predict how elements behave without testing every single one from scratch.
Elements in the same vertical column often act similarly because they have the same number of outer electrons. Elements across a horizontal row change in more gradual ways because the number of outer electrons increases step by step. This is why the table is called periodic: properties repeat in a regular pattern.
Atoms contain protons, neutrons, and electrons. Protons are positively charged, electrons are negatively charged, and neutrons have no charge. In a neutral atom, the number of protons equals the number of electrons.
At the atomic scale, electric attraction between the positive nucleus and negative electrons shapes matter. The outer electrons are the ones most involved when atoms interact, form bonds, or become ions. That is why predicting properties begins with understanding electron arrangement.
An atom's electrons occupy different energy levels, sometimes called shells. The electrons in the outermost occupied energy level are called valence electrons. These are especially important because they are the electrons that can be lost, gained, or shared during chemical changes. As [Figure 1] shows, very different elements can still be compared by looking specifically at their outermost electrons.
For example, lithium has the electron arrangement \(2,1\), carbon has \(2,4\), oxygen has \(2,6\), sodium has \(2,8,1\), and chlorine has \(2,8,7\). Even without memorizing full electron configurations, noticing the final number is enough to see the outer-electron pattern. Lithium and sodium each have one outer electron, so they belong to the same family and share important chemical traits.
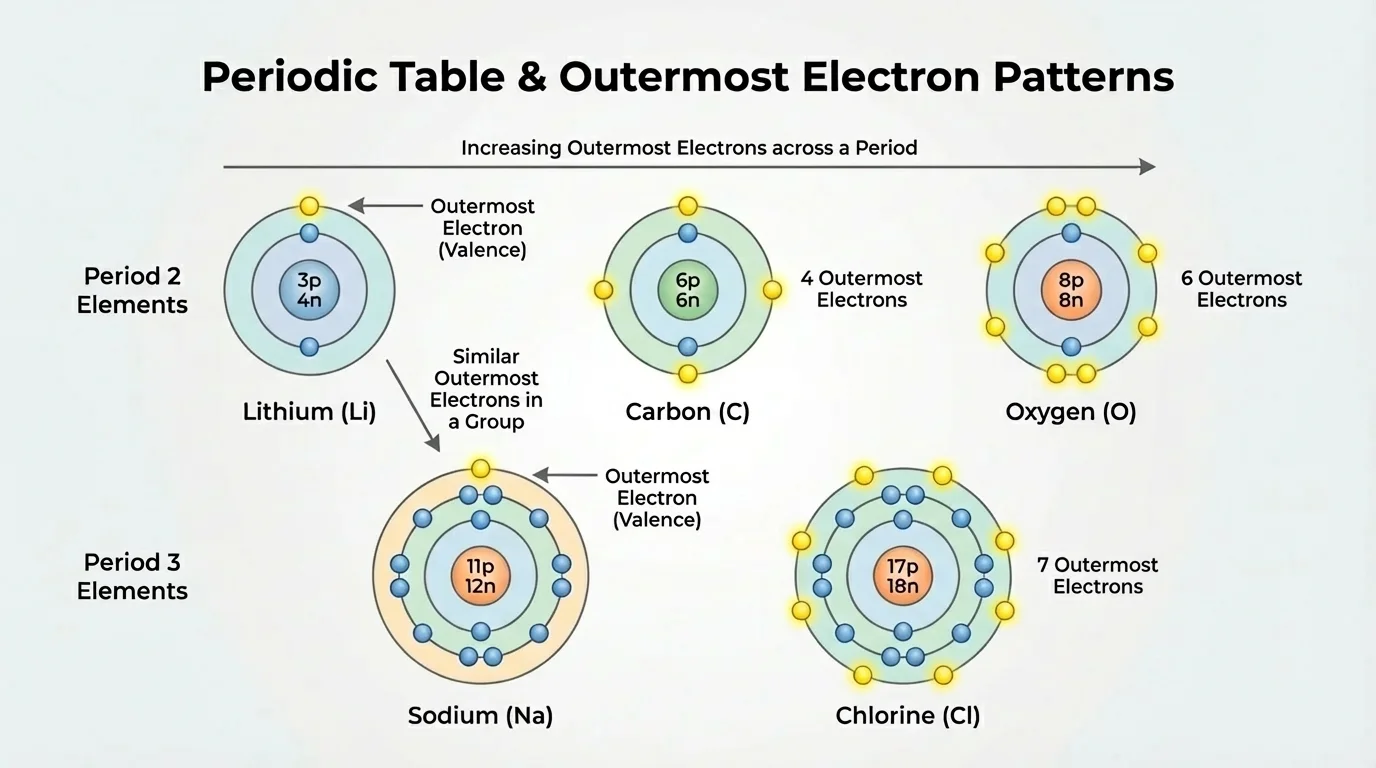
Atoms tend to become more stable when their outer energy level is filled. For many main group elements, a filled outer level means having eight valence electrons, often called an octet. There are simple exceptions in the smallest atoms, but the octet idea is a useful model for predicting the behavior of main group elements.
Valence electrons are the electrons in the outermost occupied energy level of an atom. They largely determine how an element reacts, what ions it forms, and what kinds of chemical bonds it makes.
Periodic trend is a predictable pattern in element properties across rows or down columns of the periodic table.
Because outer electrons are farther from the nucleus than inner electrons, they are held less tightly. That makes them the most available for interaction with other atoms. This simple idea explains a huge amount of chemistry.
A vertical column in the periodic table is called a group. For main group elements, members of the same group have the same number of valence electrons. This is why they show similar chemical behavior, as [Figure 2] illustrates with several important families.
Group 1 elements, including lithium, sodium, and potassium, each have one valence electron. They are soft, reactive metals that tend to lose that one electron and form ions with a charge of \(1+\). Group 2 elements, such as magnesium and calcium, have two valence electrons and commonly form \(2+\) ions.
Group 17 elements, the halogens, have seven valence electrons. Because they are just one electron short of a full outer level, they often gain one electron and form \(1-\) ions. Chlorine and bromine are classic examples. Group 18 elements, the noble gases, already have full outer energy levels, so they are much less reactive than most other elements.
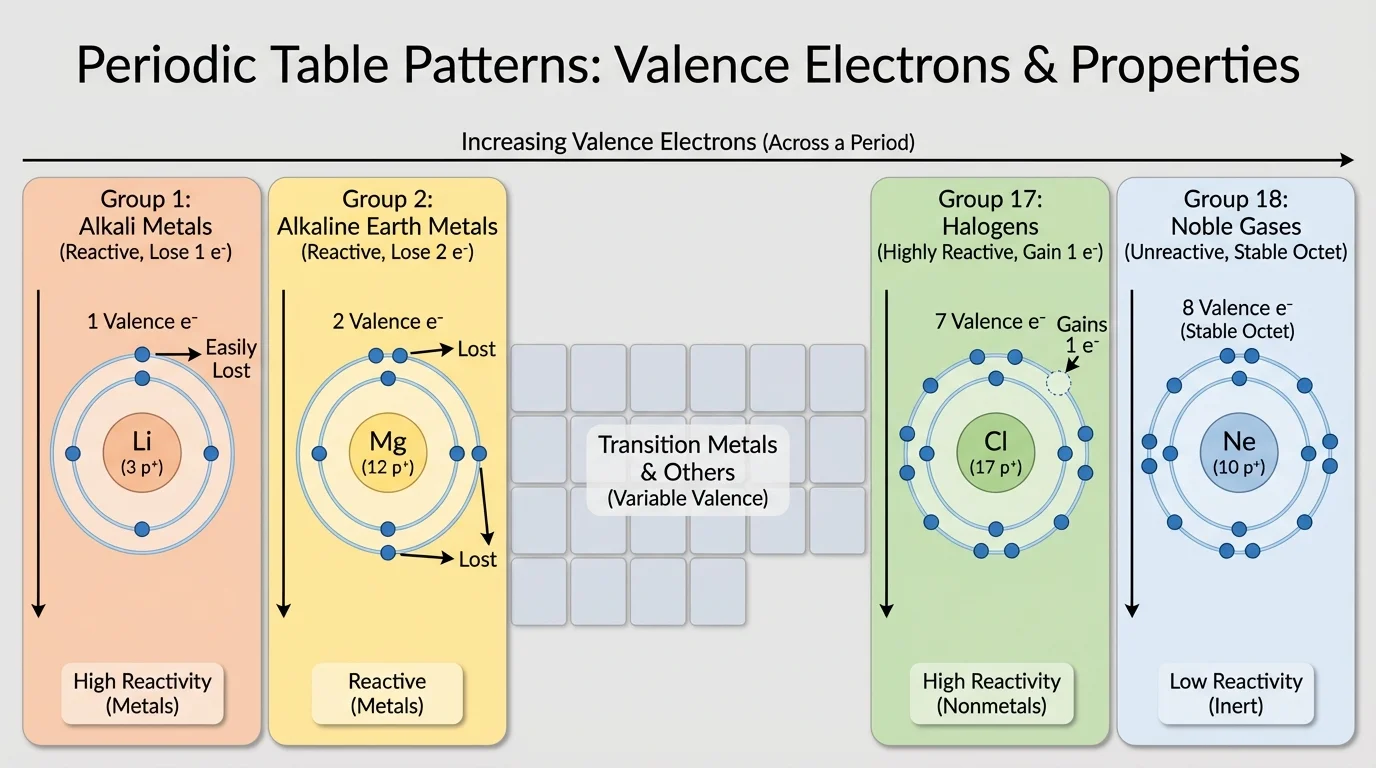
Going down a group, atoms have more energy levels. That means the outer electrons are farther from the nucleus. As a result, some properties change in a regular way down the column even though the elements still remain in the same family. For example, alkali metals generally become more reactive down the group because the single outer electron is easier to remove.
The same family pattern explains why sodium and potassium react similarly with water, producing hydrogen gas and a basic solution, while chlorine and bromine both form salts with metals. Their exact speed or intensity may differ, but the overall behavior follows the same outer-electron pattern.
Table salt, \(\textrm{NaCl}\), forms from two elements that are very different on their own: sodium is a highly reactive metal, and chlorine is a reactive nonmetal gas. Their positions in Groups 1 and 17 help predict that they will combine strongly.
Later, when you see unfamiliar main group elements, the group number gives an immediate clue about likely behavior. If an element sits with the halogens, expect strong attraction for an extra electron. If it sits with the noble gases, expect low reactivity.
A horizontal row in the periodic table is called a period. Across a period, atoms gain protons in the nucleus and also gain electrons in the same main energy level. This creates important trends. As [Figure 3] shows for a typical row, properties do not stay constant from left to right.
One major trend is atomic radius, the relative size of an atom. Across a period from left to right, atomic radius generally decreases. Even though electrons are being added, the increasing positive charge in the nucleus pulls the electron cloud closer.
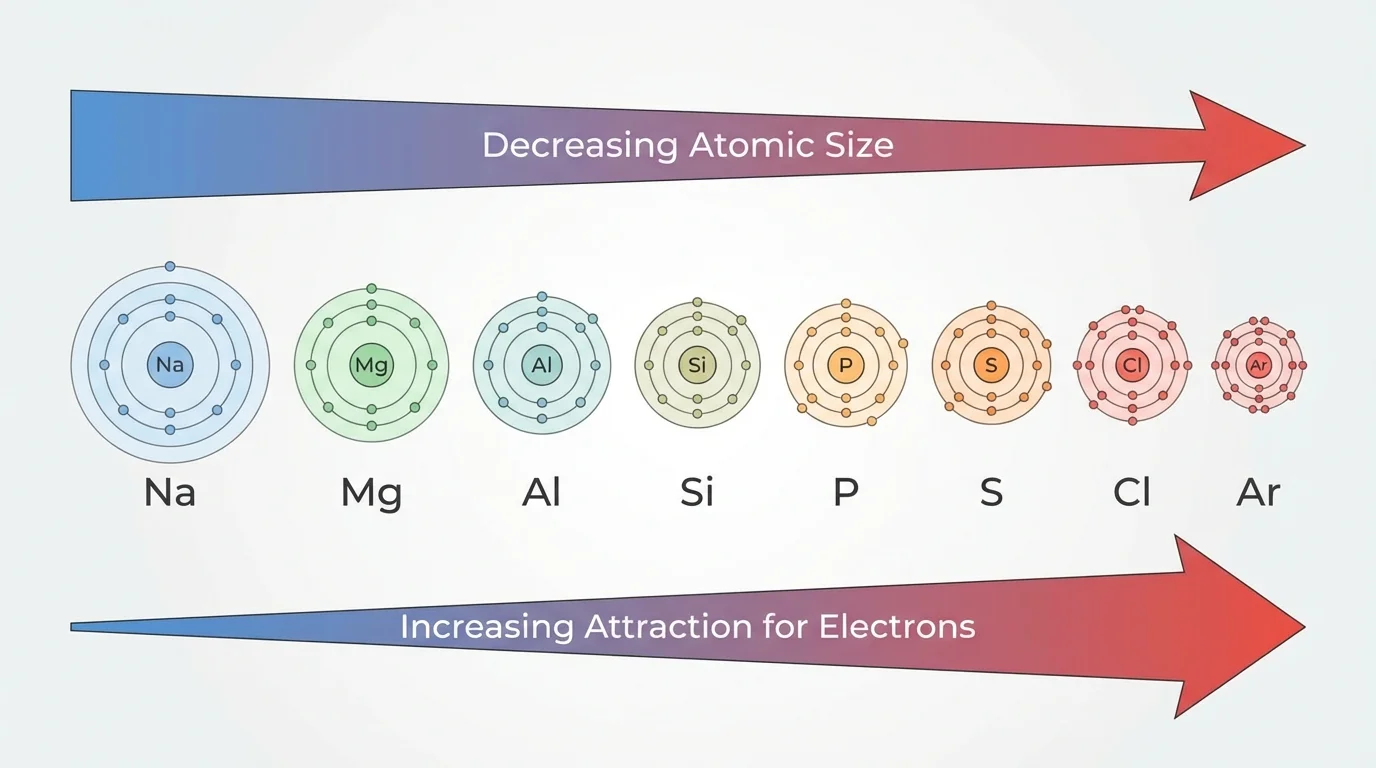
Another trend is the change from metallic behavior to nonmetallic behavior. Elements on the left side of a period are more likely to be metals. They tend to lose electrons, conduct electricity well, and form positive ions. As you move right, elements become less metallic and more likely to gain or share electrons.
Across a period, the attraction between the nucleus and outer electrons generally increases. Because of that, it becomes harder to remove an electron. This trend relates to ionization energy, which is the energy needed to remove an electron from an atom. For this topic, it is enough to understand the relative trend: ionization energy usually increases across a period and usually decreases down a group for main group elements.
A similar idea applies to electronegativity, which describes how strongly an atom attracts shared electrons in a bond. Main group elements toward the upper right of the table, excluding noble gases, generally have higher electronegativity than elements toward the lower left.
Why trends exist across a period
When moving left to right in a period, the nucleus gains more protons while the added electrons go into the same general outer energy level. The stronger positive pull of the nucleus increases attraction to electrons, so atoms become smaller, less metallic, and more likely to gain or share electrons rather than lose them.
The pattern in [Figure 3] helps explain why sodium in Period 3 is a reactive metal, silicon is a metalloid, chlorine is a reactive nonmetal, and argon is a nearly unreactive noble gas. Their positions reflect a step-by-step change in outer-electron behavior across the row.
The periodic table also helps classify elements as metals, nonmetals, or metalloids. Most elements are metals, found on the left and center of the table. Metals are typically shiny, malleable, and good conductors of heat and electricity. Because they tend to lose electrons, they often form positive ions.
Nonmetals are found mostly on the right side. They are often dull in appearance, poor conductors, and more likely to gain or share electrons. Many important biological elements are nonmetals, including carbon, nitrogen, oxygen, phosphorus, and sulfur.
Metalloids lie along the staircase-like boundary between metals and nonmetals. They have mixed properties. Silicon, for example, does not conduct electricity as well as a metal, but it can conduct under certain conditions. That makes silicon useful in electronics.
| Category | Typical location | Electron tendency | Common physical traits |
|---|---|---|---|
| Metals | Left side | Lose electrons | Conductive, malleable, lustrous |
| Metalloids | Stair-step region | Can lose or share depending on context | Intermediate, semiconducting |
| Nonmetals | Right side | Gain or share electrons | Poor conductors, often brittle if solid |
Table 1. General comparison of metals, metalloids, and nonmetals based on periodic table location and electron behavior.
This classification is not separate from electron structure; it comes from it. Metallic character reflects how easily an atom gives up outer electrons. Nonmetallic character reflects stronger attraction for those electrons.
When atoms interact, they often do so in ways that make their outer energy levels more stable. The periodic table allows us to predict common ion charges and common bonding patterns for main group elements. As [Figure 4] illustrates, some atoms transfer electrons while others share them.
An ion is an atom that has gained or lost electrons and therefore has a net electric charge. Main group metals on the left side of the table often lose electrons. Main group nonmetals on the right side often gain electrons. This electron transfer can produce an ionic bond, the attraction between oppositely charged ions.
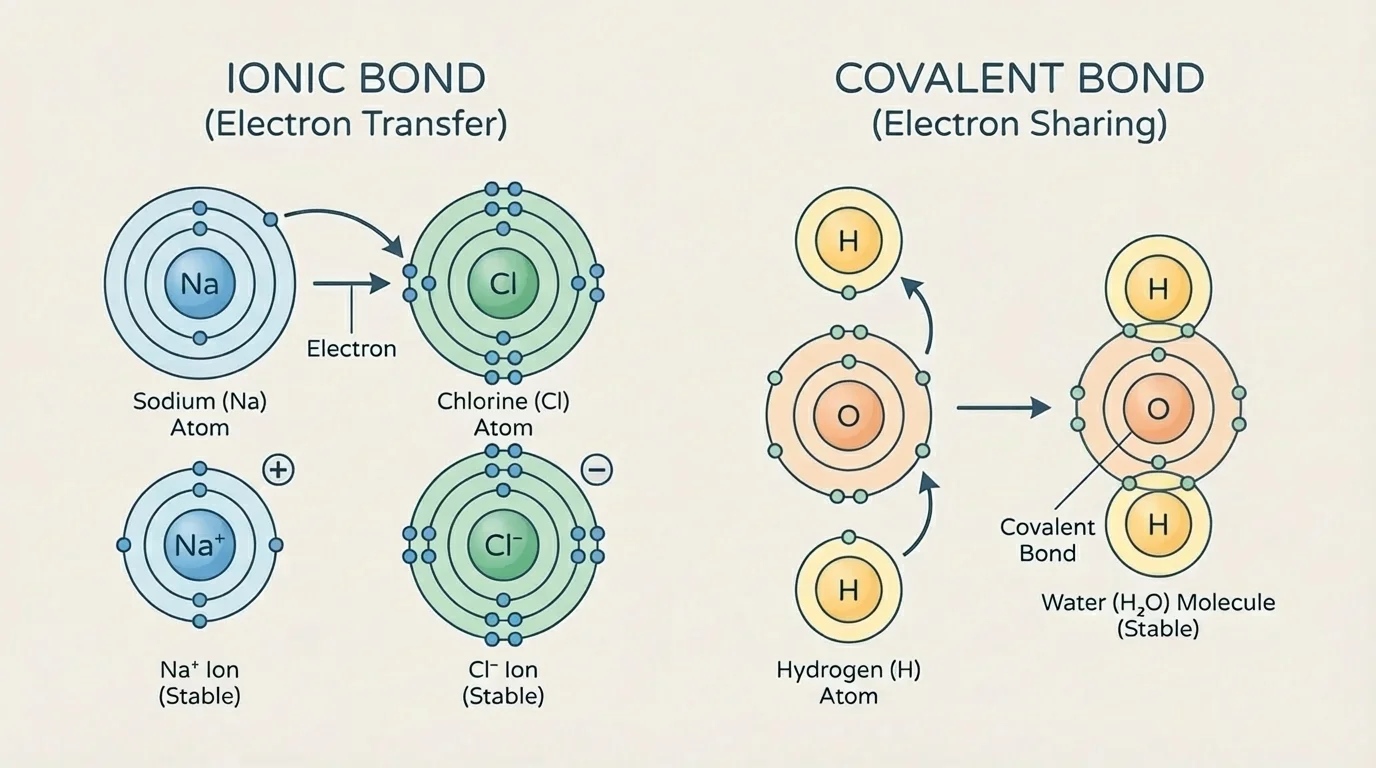
For example, sodium has one valence electron and chlorine has seven. Sodium tends to lose one electron to become \(\textrm{Na}^+\), and chlorine tends to gain one electron to become \(\textrm{Cl}^-\). The result is the ionic compound \(\textrm{NaCl}\).
By contrast, nonmetals often share electrons. Shared electrons create covalent bonds. Oxygen has six valence electrons and commonly forms two covalent bonds to complete an octet. That is why water has the formula \(\textrm{H}_2\textrm{O}\): oxygen shares electrons with two hydrogen atoms.
Using valence electrons to predict a compound
Predict the likely formula formed by magnesium and chlorine.
Step 1: Identify valence-electron behavior.
Magnesium is in Group 2, so it tends to lose two electrons and form \(\textrm{Mg}^{2+}\). Chlorine is in Group 17, so it tends to gain one electron and form \(\textrm{Cl}^-\).
Step 2: Balance charges.
One \(\textrm{Mg}^{2+}\) needs two \(\textrm{Cl}^-\) ions so the total charge is zero: \(2(-1) + (+2) = 0\).
Step 3: Write the formula.
The compound is \(\textrm{MgCl}_2\).
The periodic table makes this prediction possible even before seeing the substance.
The same reasoning predicts \(\textrm{CaO}\), because calcium tends to form \(\textrm{Ca}^{2+}\) and oxygen tends to form \(\textrm{O}^{2-}\). It also helps explain why carbon forms many covalent compounds: with four valence electrons, carbon often shares electrons rather than fully gaining or losing four.
We can return to [Figure 4] here: the diagram contrasts electron transfer and electron sharing, which are both direct consequences of outer-electron patterns. The periodic table does not just classify elements; it helps explain why compounds form in recognizable ways.
Some groups are especially useful to know because their trends are so clear.
Alkali metals in Group 1 are among the most reactive metals. Because they have one valence electron, they readily lose it. Lithium reacts with water less vigorously than sodium, and sodium less vigorously than potassium, showing a down-the-group increase in reactivity.
Alkaline earth metals in Group 2 are reactive metals as well, though usually less reactive than Group 1. They have two valence electrons and commonly form \(2+\) ions. Magnesium in fireworks and calcium in bones are familiar examples of elements from this family appearing in different contexts.
Halogens in Group 17 are very reactive nonmetals. They are often one electron short of a full outer shell, so they strongly tend to gain one electron or share electrons in covalent bonds. Fluorine is extremely reactive, and chlorine is widely used in water treatment because of its reactivity.
Noble gases in Group 18 are much less reactive because their outer shells are full. Neon signs, argon-filled light bulbs, and helium balloons all rely on this low reactivity. Their electron arrangement helps make them stable as individual atoms.
Other main group families also show patterns. Group 14 elements like carbon and silicon often form four bonds. Group 15 elements like nitrogen and phosphorus often form three bonds in covalent compounds. Group 16 elements like oxygen and sulfur often form two bonds. These are not random facts; they come from the number of valence electrons.
"The properties of the elements are a periodic function of their atomic numbers."
— A modern statement of the periodic law
This periodic law is what turns the table into a scientific model. It links invisible atomic structure to visible material behavior.
Periodic trends help engineers, chemists, doctors, and environmental scientists choose materials and predict behavior in practical situations. This is not just classroom chemistry.
In rechargeable batteries, metals such as lithium are useful because they lose electrons relatively easily. That behavior connects directly to their Group 1 position and single valence electron. Understanding this pattern helps scientists compare possible battery materials.
In computer chips and solar cells, silicon is valuable because it is a metalloid. Its intermediate electrical behavior makes it a semiconductor. That property fits its place near the border between metals and nonmetals and reflects how its outer electrons behave.
In agriculture, fertilizers often contain nitrogen, phosphorus, and potassium. These elements come from different families and play different chemical roles. Their behavior in compounds can be understood from their valence electrons and their tendency to form certain ions or covalent structures.
In medicine and biology, ions such as \(\textrm{Na}^+\), \(\textrm{K}^+\), \(\textrm{Ca}^{2+}\), and \(\textrm{Cl}^-\) are essential in nerve signaling, muscle contraction, and fluid balance. Their charges are predictable from group patterns, which is one reason the periodic table remains so useful in life sciences.
Real-world case study: choosing a metal for conductivity
Suppose an engineer needs a material that conducts electricity well and can give up electrons relatively easily.
Step 1: Look to the metallic region of the periodic table.
Metals on the left side tend to have fewer valence electrons and hold them less tightly than nonmetals on the right.
Step 2: Compare likely candidates.
Copper, aluminum, and sodium are all metals, but sodium is too reactive for many structural uses, while copper and aluminum combine conductivity with better practical stability.
Step 3: Use periodic reasoning.
The table helps predict that nonmetals like sulfur or chlorine would not be chosen for wire because they do not behave like conductive metals.
The periodic table guides material selection before a device is ever built.
The family patterns in [Figure 2] and the left-to-right changes in [Figure 3] support these real-world decisions. Scientists use both kinds of patterns at once: family similarity and row-by-row change.
Like any scientific model, the periodic table gives patterns rather than perfect shortcuts for every detail. The trends are most reliable when used comparatively: larger or smaller, more or less reactive, more metallic or less metallic. That is exactly how scientists often reason before doing experiments.
For this topic, the focus stays on main group elements. Transition metals can show more complicated behavior, including multiple common charges, so they are outside the main assessment boundary here. Also, while ionization energy is a useful idea, the important point is the relative trend, not detailed numerical calculations.
Still, within those limits, the periodic table is extraordinarily powerful. If you know where an element sits, you can often predict its outer electrons, likely ion charge, whether it behaves more like a metal or nonmetal, how strongly it holds electrons compared with nearby elements, and what kinds of compounds it tends to form.