A diamond can cut glass, table salt shatters when struck, copper wire bends without breaking, and sugar melts and burns in a kitchen pan. These materials look very different at the bulk scale, but the deeper reason is even more interesting: their particles are held together by different kinds of electrical attraction. Chemists cannot see these forces directly with the naked eye, so they rely on evidence from observable properties. When you test a substance's hardness, melting behavior, conductivity, or solubility, you are doing more than describing matter—you are gathering clues about how strongly its particles attract one another.
When scientists study matter, they often connect what they can measure to what they cannot directly observe. A bulk-scale property is a characteristic of a material that can be observed or measured for a sample of matter, such as hardness, melting point, boiling point, electrical conductivity, brittleness, or solubility. These properties arise from the way particles are arranged and from the electrical attractions and repulsions between them.
If particles are held together by very strong attractions, more energy is usually required to separate them. That often leads to a high melting point or high boiling point. If particles can shift while still staying attracted, a material may be malleable. If charged particles or electrons can move through a substance, it may conduct electricity. These are powerful patterns, but they are not random guesses; they are evidence-based inferences about structure.
Recall that all matter is made of atoms, and atoms contain positively charged protons and negatively charged electrons. Opposite charges attract and like charges repel. Those simple electrical ideas help explain bonding and the large-scale properties of substances.
At the atomic scale, the relevant forces are electrical. Positive and negative charges attract each other, and this attraction helps hold matter together. Even in substances made of neutral molecules, uneven distributions of charge can produce attractions between particles. The strength and arrangement of these interactions determine many of the properties you can test in a lab.
Electrical force is the attraction or repulsion between charged particles. In matter, electrical forces act between protons, electrons, ions, and regions of partial charge in molecules.
Bonding refers to the attractions that hold atoms or ions together in a substance.
Intermolecular forces are attractions between separate molecules, not within a single molecule.
A particle model of matter treats substances as collections of tiny particles whose arrangement and motion explain what we observe. In solids, particles are close together. In liquids, they remain close but can move past one another. In gases, they are much farther apart. For this lesson, the most important question is not just where the particles are, but what kind of attraction holds them together.
A ionic solid forms when positive and negative ions attract in a repeating lattice. Sodium chloride, \(\mathrm{NaCl}\), is a classic example. A molecular substance contains separate molecules held to one another by intermolecular forces. Sugar and carbon dioxide, \(\mathrm{CO_2}\), fit this category. A metal contains positive metal ions surrounded by mobile electrons, producing metallic bonding. Copper and aluminum behave this way. Some substances, such as diamond or silicon dioxide, form giant continuous networks of covalent bonds; these are called covalent-network solid structures.
Because electrical forces vary in strength and geometry, substances with different structures behave differently. This is why one investigation can compare several materials and still reveal meaningful patterns.
Graphite and diamond are both made only of carbon, yet graphite is soft enough to leave marks on paper while diamond is one of the hardest natural materials. The difference comes from structure and the way electrical forces act within each material.
[Figure 1] The major classes of substances differ in particle arrangement and in the kinds of attractions that dominate. This is why comparing several bulk properties is more useful than relying on just one. A single property can suggest a structure, but a pattern of properties provides stronger evidence.
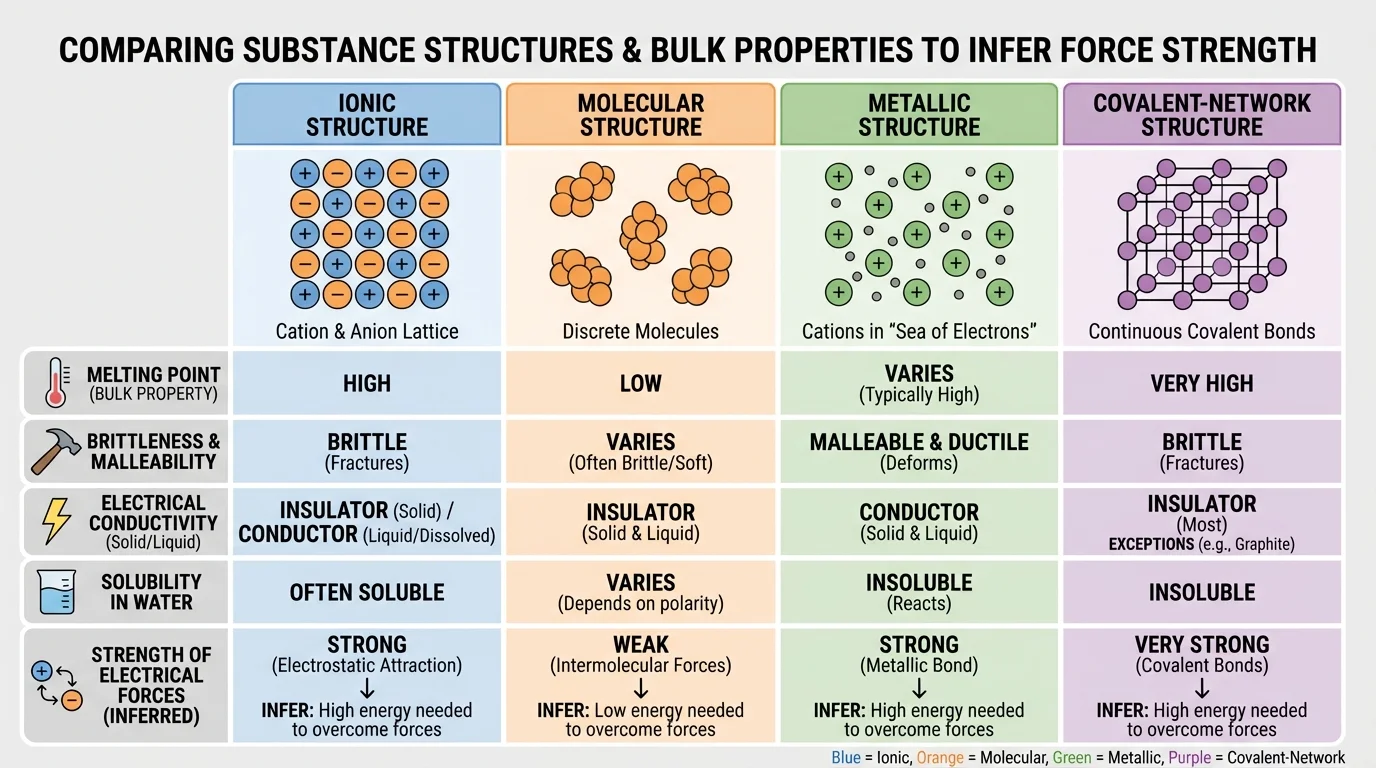
Ionic solids usually have high melting points because the attractions between oppositely charged ions are strong. They are often hard but brittle. When struck, layers can shift so that like charges become adjacent, causing repulsion and fracture. They do not conduct electricity well as solids because the ions are fixed in place, but they often conduct when melted or dissolved in water because the ions can move.
Molecular substances often have lower melting and boiling points because the forces between separate molecules are usually weaker than ionic or network bonding. Many are soft or easily deformed. Most do not conduct electricity because they lack mobile charged particles. Some dissolve in water, while others do not, depending on molecular polarity.
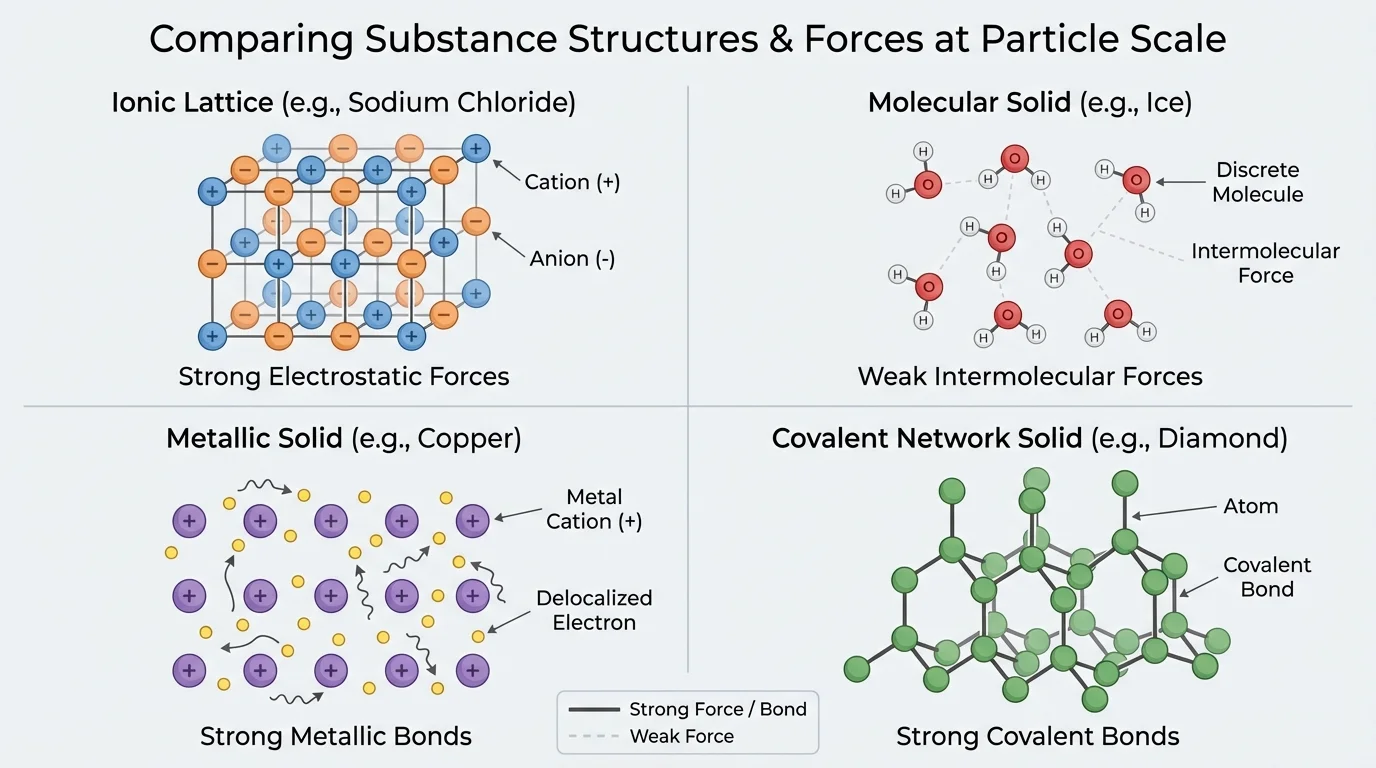
Metals often conduct electricity and heat well because electrons move through the structure. They are usually malleable and ductile because layers of atoms can shift without breaking the overall attraction. Their melting points vary, but many are relatively high.
Covalent-network solids, such as diamond and \(\mathrm{SiO_2}\), often have extremely high melting points because atoms are linked in an extended network by strong covalent bonds. They are usually very hard. Most do not conduct electricity, although graphite is an important exception because its electrons can move within layers. That exception reminds us to use evidence carefully rather than memorize oversimplified rules.
| Type of substance | Typical particle arrangement | Common bulk properties | What the properties suggest |
|---|---|---|---|
| Ionic solid | Positive and negative ions in a lattice | High melting point, brittle, conducts when melted or dissolved | Strong attractions between oppositely charged ions |
| Molecular substance | Separate molecules | Lower melting point, often soft, poor conductor | Weaker attractions between particles |
| Metal | Positive metal ions with mobile electrons | Conductive, malleable, often shiny | Attraction involving delocalized electrons allows movement and conductivity |
| Covalent-network solid | Atoms joined in a continuous covalent network | Very hard, very high melting point, usually poor conductor | Strong bonding throughout the entire structure |
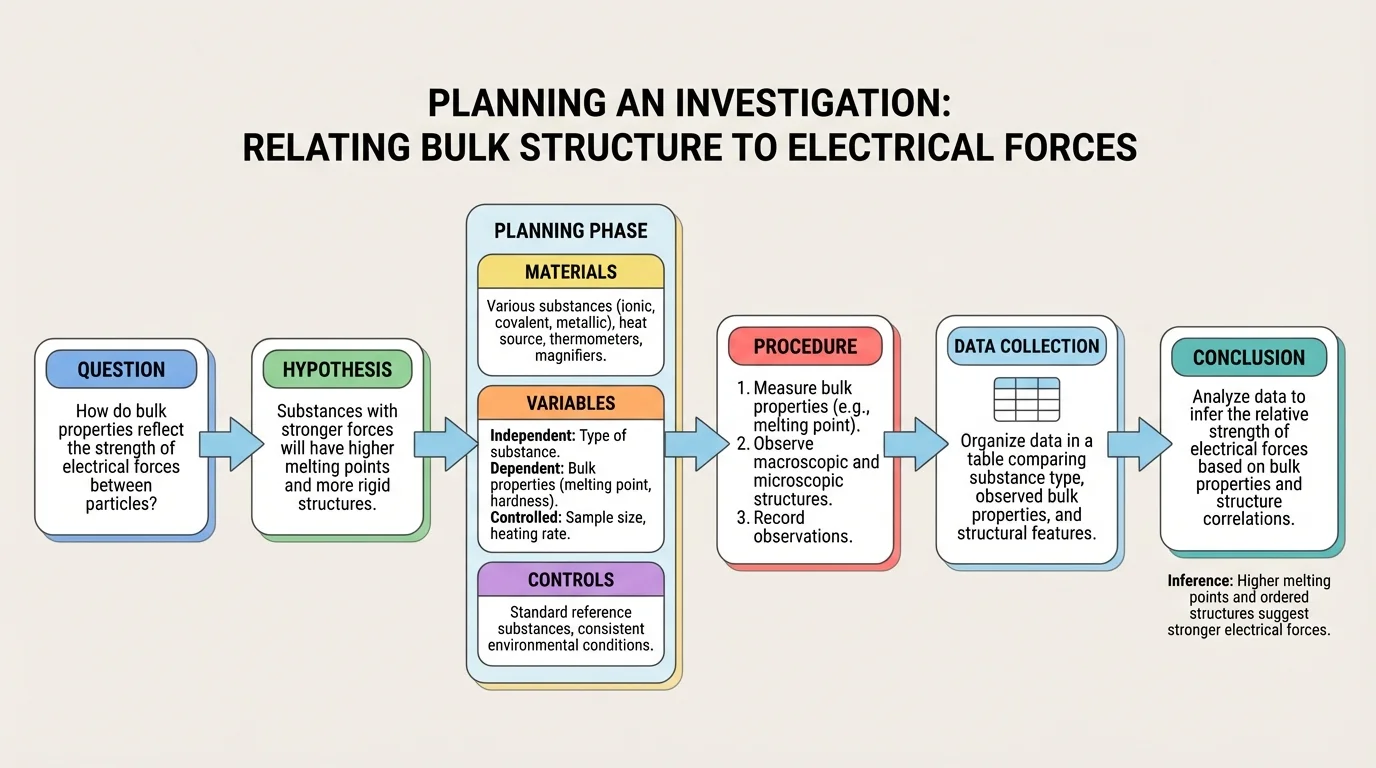
[Figure 2] A good investigation begins with a focused question. For this topic, a strong question might be: How do conductivity, hardness, solubility, and melting behavior provide evidence about the structure of different substances? A fair investigation compares several samples using the same procedures.
You might choose samples such as \(\mathrm{NaCl}\) for an ionic solid, sugar for a molecular substance, paraffin wax for another molecular substance, copper for a metal, and graphite or sand as an example of a covalent-network solid. Not every school lab can test all materials under the same conditions, so sample choice should match available equipment and safety limits.
The independent variable is the substance being tested. The dependent variable is the property measured, such as conductivity or time to melt under a fixed heat source. controlled variables include sample size, heating method, amount of water used for dissolving, voltage in a conductivity tester, and testing time.
For safety, students should wear goggles, handle hot equipment carefully, avoid tasting any substance, and follow teacher instructions for heating and disposal. Some substances can release fumes or behave unpredictably when heated, so direct flame testing should be limited to approved materials and procedures.
Why multiple tests matter
One property alone rarely proves a structure. For example, a high melting point may suggest strong attractions, but both ionic solids and covalent-network solids can have high melting points. Conductivity, brittleness, solubility, and melting behavior together provide a more reliable pattern of evidence.
A useful data table should be prepared before testing begins. This helps ensure that observations are recorded consistently rather than from memory. Qualitative data, such as "brittle" or "did not dissolve," and quantitative data, such as conductivity reading or approximate melting temperature, can both contribute to valid conclusions.
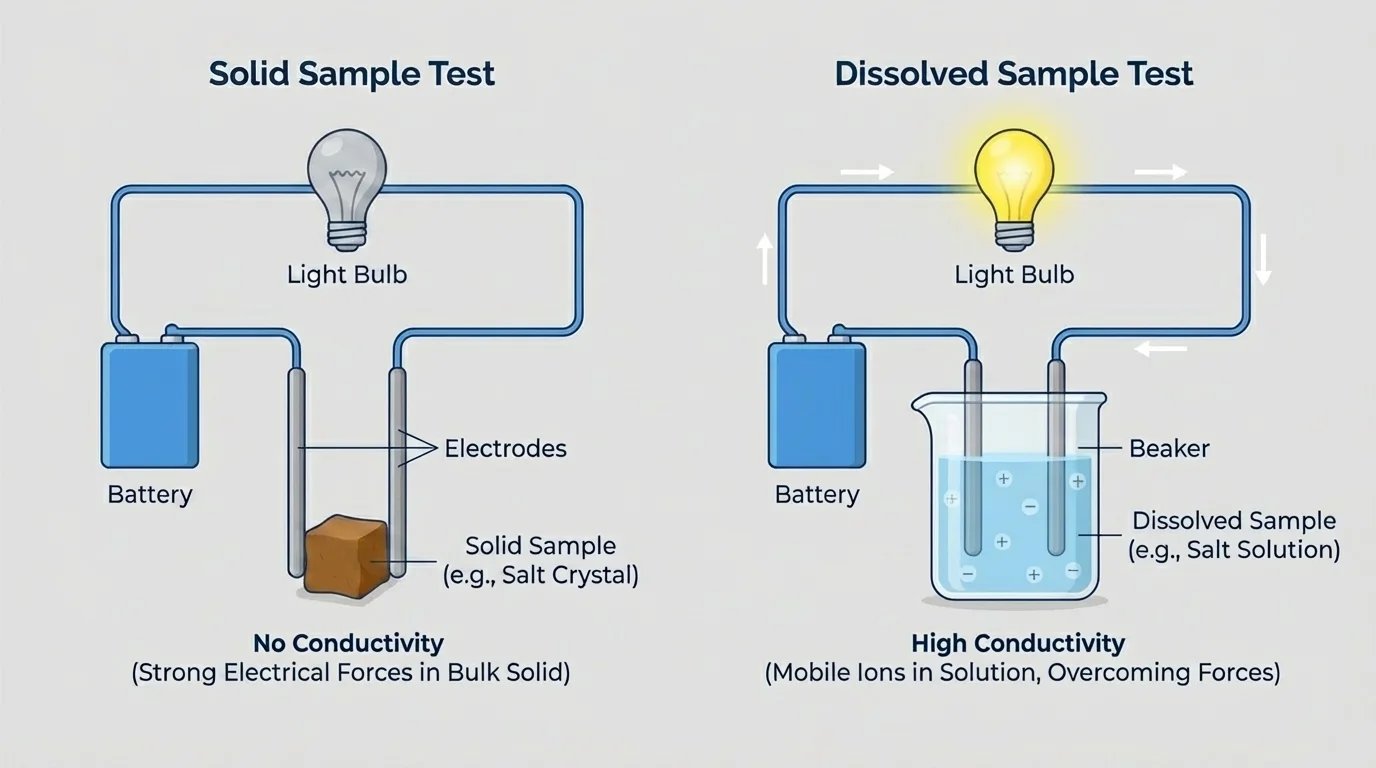
[Figure 3] One test often used in this kind of investigation is electrical conductivity. A simple circuit with a battery, wires, and a bulb or conductivity sensor can provide evidence. If a solid sample completes the circuit, it likely has mobile charged particles or electrons. If it does not, you may still test the substance after dissolving it in water, if safe and appropriate.
For example, solid \(\mathrm{NaCl}\) usually does not conduct, but an aqueous solution of \(\mathrm{NaCl}\) does because dissolved \(\mathrm{Na^+}\) and \(\mathrm{Cl^-}\) ions can move. Sugar dissolves in water but generally does not conduct because it forms neutral molecules in solution rather than ions. Copper conducts as a solid because electrons move through the metallic structure.
A melting-behavior test can be done by heating small, equal-sized samples using the same apparatus and time interval, while following strict safety rules. The goal is not always to measure an exact melting point. In many classroom investigations, the useful evidence is comparative: which sample melts easily, softens slightly, remains unchanged, or decomposes before melting.
Hardness and brittleness can be tested with care by applying controlled pressure or gentle tapping. A substance that shatters is brittle; a substance that bends is malleable. Solubility can be tested by adding equal masses of substances to equal volumes of water, stirring for the same amount of time, and observing whether the sample dissolves fully, partly, or not at all.
Sample investigation design
Suppose a class compares \(\mathrm{NaCl}\), sugar, copper, and sand.
Step 1: Test conductivity of each solid using the same circuit.
Expected pattern: copper conducts strongly; \(\mathrm{NaCl}\), sugar, and sand do not conduct as solids.
Step 2: Dissolve \(\mathrm{NaCl}\) and sugar in equal volumes of water and test again.
Expected pattern: the \(\mathrm{NaCl}\) solution conducts; the sugar solution does not.
Step 3: Compare behavior during careful heating.
Expected pattern: sugar changes at lower temperature than sand; sand remains unchanged under classroom heating because its structure is much stronger.
Step 4: Test mechanical behavior.
Expected pattern: copper bends; \(\mathrm{NaCl}\) crystals crush and fracture; sand grains remain hard but do not bend.
Together, these observations support different structural models for each substance.
Scientists value repeatability, so each test should be repeated when possible. If three conductivity trials produce values of 0.82, 0.80, and 0.84 in the same units, the average is \(\dfrac{0.82 + 0.80 + 0.84}{3} = 0.82\). Averaging helps reduce the effect of random variation. Even simple numerical comparisons can strengthen a claim.
[Figure 4] The most important scientific move happens after the measurements: turning observations into evidence-based reasoning. This step links a sample's behavior to its internal structure. The question is not just what happened? but what does that pattern suggest about the attractions between particles?
If a substance has a high melting point and is brittle, that supports the idea of strong forces holding particles in fixed positions, which is consistent with many ionic solids or covalent-network solids. If the same substance does not conduct as a solid but does conduct when dissolved, the ionic model becomes much stronger. That is because mobile ions in solution explain the new conductivity.
If a substance melts relatively easily, does not conduct in either solid or dissolved form, and consists of separate molecules, the evidence points toward weaker intermolecular attractions between neutral particles. This pattern is common for many molecular substances. If a substance conducts as a solid and bends instead of shattering, a metallic structure is a strong inference.
Notice that the conclusion is usually comparative rather than absolute. Students infer whether electrical forces are relatively stronger or weaker among the substances tested. For example, if wax melts under mild heating but sand does not, the attractions between particles in sand must be stronger overall than those between wax molecules. If copper conducts while sugar does not, copper must contain mobile charges that sugar lacks.
Later in the analysis, [Figure 1] remains useful because it connects these test results to particle arrangement. Likewise, the design logic from [Figure 2] helps explain why a claim is more convincing when variables were controlled and observations were collected systematically.
Using evidence to make a claim
A student group observes the following for an unknown substance: it does not conduct as a solid, dissolves in water, the solution conducts strongly, and the crystals are brittle.
Step 1: List the evidence.
Nonconductive solid, conductive solution, brittle crystal structure, water solubility.
Step 2: Compare with known patterns.
This matches the behavior of many ionic substances better than molecular, metallic, or network substances.
Step 3: Infer particle-level structure.
The substance likely contains oppositely charged ions arranged in a crystal lattice.
Step 4: Infer force strength.
The electrical attractions between particles are strong because ions in the lattice are held tightly, which helps explain the brittleness and likely high melting behavior.
This is an example of reasoning from evidence rather than guessing from appearance.
Engineers choose materials by thinking exactly this way. Electrical wires are made from metals such as copper because mobile electrons allow strong conductivity and metallic bonding allows the wire to be drawn into long, thin shapes. Ceramics used in tiles, furnace linings, and some electronics often have strong particle attractions and high thermal stability. Molecular substances are used where lower melting points or flexible behavior are useful, such as waxes, fuels, and many plastics.
Road salt works because ionic compounds dissolve in water and interact strongly with it, while semiconductor design depends on precise control of particle structure and electron movement. Battery technology also depends on ionic movement in electrolytes and electron flow in external circuits. These are not separate ideas from chemistry class; they are direct applications of structure-property relationships.
Materials scientists often begin with the question, "What property do we need?" and then work backward to the particle structure most likely to produce it. Strong lightweight materials, flexible electronics, and high-temperature ceramics all come from this kind of reasoning.
One common mistake is thinking that strong bonds within a molecule automatically mean a substance has a high melting point. That is not always true. In many molecular substances, the molecules themselves are strongly bonded internally, but the attractions between molecules are much weaker. Melting often separates molecules from one another without breaking the covalent bonds inside each molecule.
Another mistake is assuming conductivity always means ions are present. Metals conduct because electrons move freely, not because ions flow through the solid. A third mistake is assuming one test gives a final answer. A good investigation uses multiple sources of evidence, because several properties must be compared together.
There are also practical limits. Classroom heating may not reach the actual melting point of some substances, so students may only compare whether a material changes under the available conditions. That is still useful evidence. Solubility results can also depend on the solvent used. Water is common in school labs, but a substance insoluble in water might dissolve in another liquid. These limits do not make the investigation invalid; they define the scope of the conclusions.
This topic does not require vapor-pressure calculations such as Raoult's law. The emphasis is on planning and conducting investigations, gathering evidence from observable properties, and using that evidence to infer the relative strength of electrical forces between particles.