A glow stick fades faster in a hot car than in a cold room. Food spoils more slowly in a refrigerator. A small spark can trigger rapid combustion in an engine under suitable conditions. These are not random facts—they all connect to one important concept in chemistry: the speed of a reaction depends on what reacting particles are doing. When scientists talk about the reaction rate, they are asking how quickly reactants turn into products, and that speed can change dramatically when temperature or concentration changes.
Reaction rate is important because chemistry is not just about whether a reaction can happen. It is also about how fast it happens. A reaction that takes years is very different from one that finishes in seconds, even if both produce the same products. In real life, reaction rates affect airbag deployment, rusting, digestion, battery performance, industrial manufacturing, and the shelf life of medicine and food.
In high school chemistry, rate is often studied using simple reactions with two reactants. You might observe how quickly bubbles form when an acid reacts with a solid, how fast a cloudy precipitate appears when two solutions are mixed, or how quickly a colored reactant disappears. These observations become evidence that helps explain what is happening at the particle level.
Matter is made of tiny particles that are always moving. In gases and liquids, particles move freely and collide often. Chemical reactions happen when atoms are rearranged to form new substances.
To understand why rate changes, we need to connect what we can see in the lab to what is happening among atoms, ions, or molecules too small to see directly.
The rate of a reaction indicates how quickly reactants are used up or how quickly products are formed. If a gas-producing reaction releases bubbles very quickly, the rate is high. If the same reaction produces only a few bubbles each minute, the rate is lower. In simple terms, faster visible change usually means a faster reaction.
Scientists sometimes compare rates by measuring how much product forms in a certain time or how long it takes for a specific change to happen. For example, if one trial produces enough gas in \(20\ \textrm{s}\) and another takes \(60\ \textrm{s}\), the first trial is faster. If one mixture turns cloudy in \(15\ \textrm{s}\) and another in \(45\ \textrm{s}\), the first has a higher reaction rate.
Reaction rate is the speed at which reactants are converted into products.
Temperature is a measure related to the average kinetic energy of particles in a substance.
Concentration describes how much of a substance is present in a given volume.
Rate is not something separate from the particles. It is the large-scale result of countless microscopic collisions happening every second.
Collision theory explains reaction rate by focusing on particle collisions, as [Figure 1] illustrates. According to this idea, particles must collide for a reaction to occur. But not every collision leads to products. Some collisions are too weak, and the particles simply separate again.
For a collision to produce a reaction, it must be a successful collision. That means the colliding particles need enough energy to begin breaking old bonds and forming new ones. The minimum energy needed is called activation energy. If the particles collide with less energy than this minimum, no reaction occurs, even though a collision happened.
This is why two substances can be mixed and still react slowly. The particles may be colliding often, but if too few collisions have enough energy, the overall rate remains low. In simple reactions with two reactants, the rate depends strongly on how often particles collide and how many of those collisions are successful.
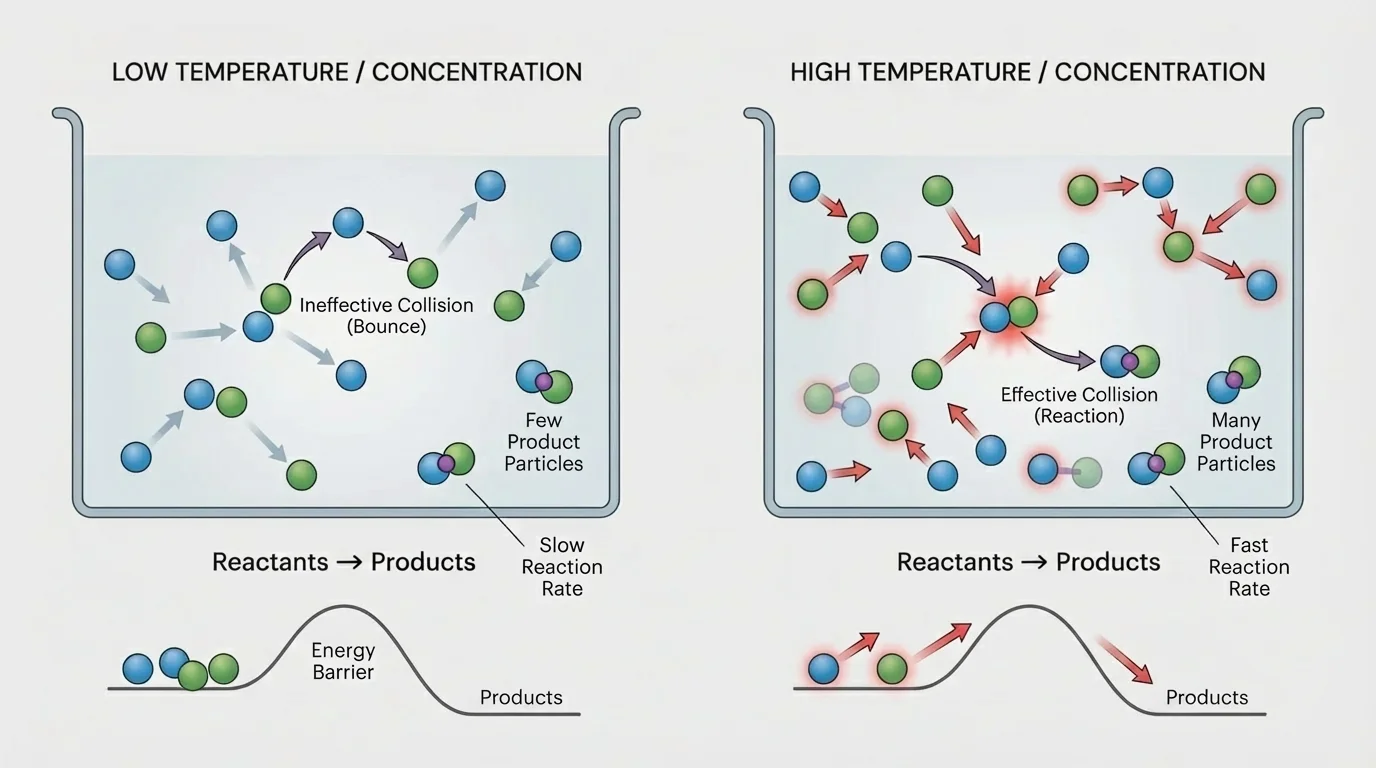
You can think of activation energy as a kind of energy hurdle. The particles do not need to be "trying" to react; they simply need to collide under the right conditions. Later, when we look at temperature and concentration, we will see that each factor changes collision patterns in a different way.
How a chemical reaction starts
In a simple two-reactant system, particles move, collide, and sometimes react. The overall rate depends on two linked ideas: collision frequency and the fraction of collisions that have enough energy to overcome activation energy. Temperature affects both strongly, while concentration mainly affects collision frequency.
The diagram in [Figure 1] helps explain an important point: a reaction does not speed up just because particles exist in the same container. What matters is the number of effective collisions over time.
When kinetic energy increases, particles move faster, and that is the key reason temperature changes reaction rate, as shown in [Figure 2]. At a higher temperature, particles in a gas or liquid have greater average kinetic energy. Faster-moving particles collide more often and with greater energy.
This produces two important effects. First, the number of collisions per second increases. Second, a larger fraction of those collisions have enough energy to overcome activation energy. Because of both effects, the reaction rate increases.
At lower temperature, the opposite happens. Particles move more slowly, collisions happen less often, and fewer collisions are energetic enough to cause a reaction. So the rate decreases. This is why refrigerating food slows many chemical reactions and biological processes that lead to spoilage.
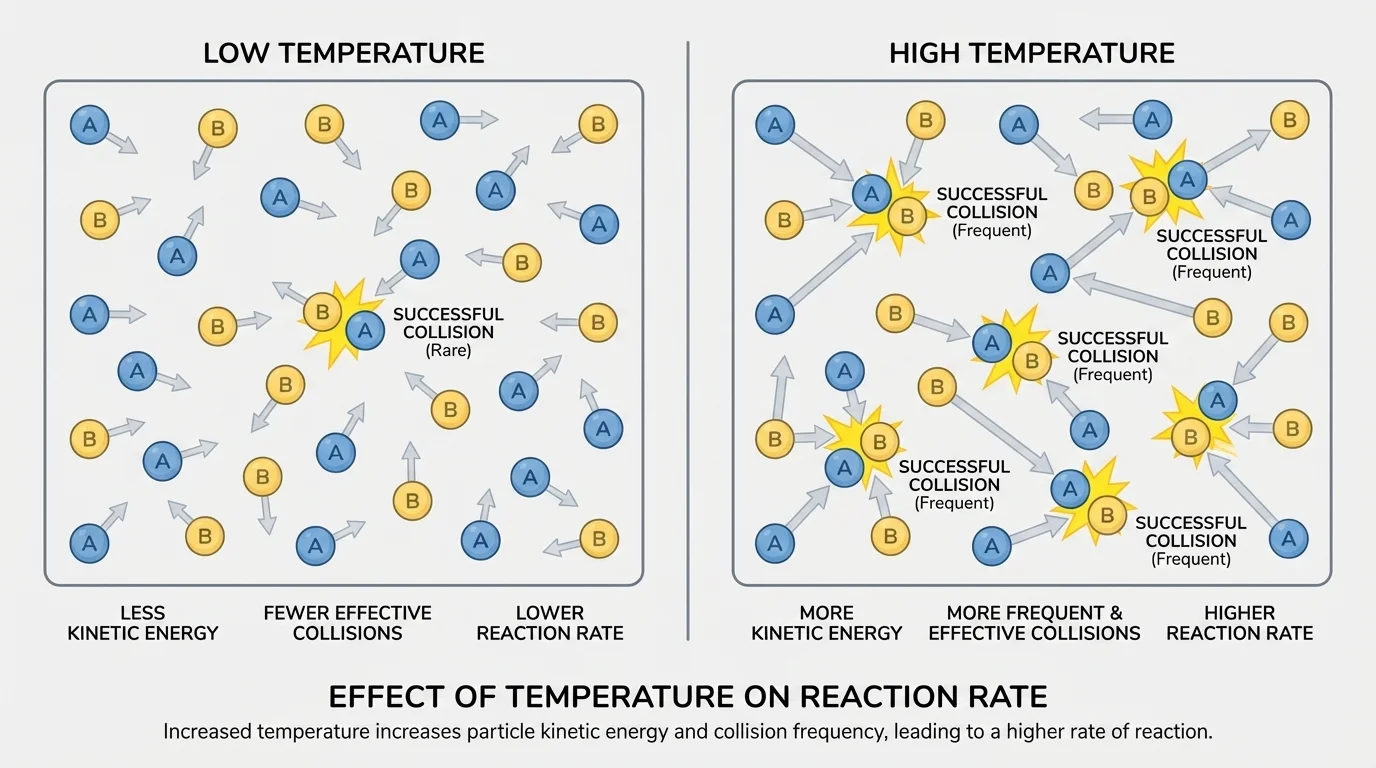
The relationship between temperature and rate is often very noticeable. Even a moderate temperature increase can make a reaction occur much faster. In this lesson, the important idea is qualitative: higher temperature usually means faster reaction rate because particles have more energy and more successful collisions occur.
Consider a simple reaction between hydrochloric acid and magnesium: \[\textrm{Mg} + 2\textrm{HCl} \rightarrow \textrm{MgCl}_2 + \textrm{H}_2\]
If the acid and magnesium are warmed, hydrogen gas forms more quickly. The faster bubbling is evidence that the reaction rate has increased. The atoms involved are the same, and the products are the same, but the particles collide under more energetic conditions.
Using temperature data as evidence
A class compares the same two-reactant reaction at three temperatures. Each trial uses equal amounts of reactants, and students measure how long it takes to produce a fixed amount of gas.
Step 1: Examine the evidence
At \(10^\circ \textrm{C}\), the fixed amount of gas is reached in \(90\ \textrm{s}\). At \(25^\circ \textrm{C}\), it is reached in \(50\ \textrm{s}\). At \(40^\circ \textrm{C}\), it is reached in \(25\ \textrm{s}\).
Step 2: Compare the times
Shorter time means faster rate. Since \(25\ \textrm{s} < 50\ \textrm{s} < 90\ \textrm{s}\), the reaction is fastest at \(40^\circ \textrm{C}\) and slowest at \(10^\circ \textrm{C}\).
Step 3: Explain scientifically
Higher temperature gives particles more kinetic energy, so collisions happen more often and more collisions exceed activation energy.
The data support the explanation that increasing temperature increases reaction rate.
The particle picture in [Figure 2] also explains why temperature changes are often more powerful than students expect. The change is not just "particles moving a little faster." It changes how many collisions are energetic enough to produce new substances.
When interpreting evidence, it is important to focus on what was kept constant and what was changed. If the amount of reactants stays the same and only temperature changes, then differences in rate can be attributed to temperature. This is part of good scientific reasoning: use controlled evidence to support a claim.
Suppose two equal volumes of the same acid react with equal masses of the same metal. Trial A at \(20^\circ \textrm{C}\) produces steady bubbling and finishes in \(80\ \textrm{s}\). Trial B at \(35^\circ \textrm{C}\) bubbles much more vigorously and finishes in \(35\ \textrm{s}\). A strong explanation is not just "heat makes it faster." A better explanation is that higher temperature increases particle kinetic energy, which increases both collision frequency and the number of successful collisions.
Notice that temperature changes the rate, not the identity of the products. The same atoms are rearranged; they are just rearranged more quickly.
[Figure 3] Temperature changes particle energy, but concentration changes how crowded the reacting particles are in a given volume. In a solution, higher concentration means more reactant particles are present in the same amount of space.
When more particles occupy the same volume, collisions between reactant particles happen more often. More collisions per second means more chances for successful collisions, so the reaction rate increases. Lower concentration means fewer particles in the same space, fewer collisions, and a slower rate.

This explanation fits many simple reactions between two reactants. For example, a more concentrated acid reacting with the same amount of a metal often produces gas faster than a more dilute acid. The acid particles are more numerous in each milliliter of solution, so they collide with the metal surface more frequently.
Concentration does not mean each particle becomes larger or more energetic. The particles are not "stronger" by themselves. There are simply more of them in the same volume, so collisions happen more often.
Using concentration data as evidence
Students test a reaction between two aqueous reactants. They keep temperature and total volume the same, but they change the concentration of one reactant.
Step 1: Read the observations
At \(0.50\ \textrm{mol/L}\), the mixture becomes cloudy in \(48\ \textrm{s}\). At \(1.00\ \textrm{mol/L}\), it becomes cloudy in \(24\ \textrm{s}\). At \(1.50\ \textrm{mol/L}\), it becomes cloudy in \(16\ \textrm{s}\).
Step 2: Identify the pattern
As concentration increases, the time needed for the visible change decreases.
Step 3: Explain the cause
Higher concentration places more reactant particles in the same volume, increasing collision frequency and increasing the reaction rate.
The evidence supports the claim that increasing concentration increases reaction rate in this two-reactant system.
The side-by-side comparison in [Figure 3] makes the concentration idea easier to see: the crowded container does not change the type of particles, only how often they meet.
Scientific explanations should connect patterns in data to particle behavior. If a more concentrated solution causes a reaction to finish sooner, that shorter time is evidence of a faster rate. The explanation should mention the greater number of particles per volume and the increased collision frequency.
Here is a simple comparison:
| Trial | Condition Changed | Observation | What It Means |
|---|---|---|---|
| A | Lower concentration | Slower bubbling or longer time | Lower reaction rate |
| B | Higher concentration | Faster bubbling or shorter time | Higher reaction rate |
A complete explanation uses both observation and theory: the visible change happens faster, and collision theory explains why.
Both temperature and concentration can increase reaction rate, but they do so in different ways. Higher concentration mainly increases how often particles collide. Higher temperature also increases collision frequency, but it has an additional effect: it increases the fraction of collisions that have enough energy to overcome activation energy.
That difference matters. Two reactions might experience the same general outcome—faster rate—but the particle-level reason is not identical. Concentration packs more reactant particles into the same volume. Temperature gives particles more kinetic energy.
This comparison shows why a strong scientific explanation must be precise. Saying "the particles react more" is too vague. A stronger explanation identifies whether the main change is in collision frequency, collision energy, or both.
Glow sticks are a good example of temperature affecting reaction rate. The chemicals inside still produce light in the cold, but the reaction slows down, so the glow is dimmer and lasts longer.
That same idea applies in many technologies. Engineers and chemists often control reaction speed by carefully choosing temperature and concentration to get useful rates without causing dangerous conditions.
In refrigeration, lower temperature slows reactions that make food spoil. In medicine, some drugs must be stored at specific temperatures so that decomposition reactions occur more slowly. In industry, manufacturers may use higher temperatures or higher concentrations to increase production rate, but they must also manage safety because fast reactions can release energy quickly.
Combustion in engines is another useful example. Fuel and oxygen react far more rapidly when the conditions allow frequent, energetic collisions. In environmental chemistry, warmer conditions can speed some reactions in air or water. In all these cases, the same scientific principle applies: particle behavior determines rate.
Even cooking involves reaction rate. Browning reactions, protein changes, and flavor-producing chemical processes occur faster at higher temperatures. A refrigerator slows chemical change, while a hot pan speeds it up dramatically.
A straightforward way to study rate is to use a simple two-reactant reaction that gives a visible result, such as gas production or cloudiness. One trial can be run at room temperature and another at a higher temperature, with all other conditions kept the same. Students then compare the time needed for the same amount of visible change.
Another investigation uses different concentrations of one reactant while keeping temperature, total volume, and the amount of the second reactant constant. If the more concentrated trial changes faster, that evidence supports the idea that concentration affects collision frequency.
Designing a fair test
A fair test changes only one factor at a time.
Step 1: Choose the factor
Change either temperature or concentration, but not both in the same comparison.
Step 2: Keep other conditions constant
Use the same reactants, the same amounts, the same container size, and the same method of timing.
Step 3: Collect evidence
Record the time for a visible change, such as the formation of a fixed amount of gas or the appearance of cloudiness.
Step 4: Explain using particles
Use collision theory to connect the evidence to collision frequency and successful collisions.
This method turns observations into scientific evidence.
Careful control of variables is what makes an explanation trustworthy. Without that control, it becomes hard to know which factor actually caused the rate change.
One common mistake is saying that heat is a reactant. It is not. Temperature changes the energy of particles, which changes the rate, but it does not automatically become one of the substances in the chemical equation.
Another mistake is thinking that high concentration means each particle is more powerful. The individual particles are not bigger or stronger just because concentration is higher. There are simply more of them in a certain volume, so collisions are more frequent.
A third mistake is assuming every collision causes a reaction. As we saw with activation energy in [Figure 1], many collisions are unsuccessful. A reaction rate depends on successful collisions, not just total collisions.
Finally, students sometimes confuse a fast reaction with a larger amount of product. Rate tells how quickly products form, not necessarily how much product is formed in total. Two trials can make the same total amount of product, but one can reach that amount much faster.
"Chemistry happens one collision at a time, but the evidence appears where we can see it."
When you explain changes in reaction rate scientifically, always connect the visible evidence to the particle model: particles collide, energy matters, and changing temperature or concentration changes how often successful collisions occur.