Your phone screen resists scratches, a bike helmet absorbs impact, a rain jacket keeps water out while remaining flexible, and a medical contact lens stays soft and wet on your eye. These materials do very different jobs, yet their success depends on the same deep idea: what a material does depends on how its particles are arranged. Engineers do not choose materials only by looking at color, weight, or cost. They also care about the invisible level of atoms and molecules, because that level controls strength, flexibility, conductivity, transparency, and even how a material responds to electric fields.
When scientists and engineers communicate about materials, they often connect three linked ideas: structure, property, and function. A material's structure includes the kinds of atoms present, how they are bonded, the shape of the molecules, and how those molecules are arranged relative to one another. Its properties include traits such as hardness, elasticity, melting behavior, water resistance, or electrical insulation. Its function is the job it performs in a design. Good scientific communication makes clear how one leads to the next: structure influences property, and property determines function.
A designed material is a material selected or engineered for a specific purpose. Some designed materials occur naturally and are modified for use, but many are created by controlling composition and molecular arrangement. For example, packaging film needs to bend without breaking, while a bullet-resistant vest needs high tensile strength. Those requirements are different, so the molecular structures must also be different.
This idea may seem surprising because two materials can contain some of the same elements and still behave very differently. Carbon provides a famous example in nature: one arrangement forms soft graphite, while another forms extremely hard diamond. In designed materials, a similar principle applies. If polymer chains are packed loosely, they may slide past each other easily, leading to flexibility. If they are tightly linked or strongly attracted, the material may become stiff, strong, or heat-resistant.
Molecular-level structure means the arrangement of atoms within molecules and the arrangement of molecules relative to each other. It includes bonding patterns, chain length, branching, polarity, and cross-linking.
Property is a measurable or observable characteristic of a material, such as stiffness, conductivity, transparency, or water resistance.
Function is the role a material performs in a device or product.
Because the topic focuses on provided molecular structures of specific designed materials, the most important skill is not inventing a brand-new molecule. Instead, it is learning to read a structure and explain what that structure suggests about material behavior.
The first step is understanding that not all forces in materials are the same. Some atoms are joined by strong chemical bonds within a molecule or chain, while molecules or chains may interact through weaker attractions between them. These different interactions matter enormously, as [Figure 1] illustrates in polymer materials where strong bonds hold each chain together but weaker attractions often control how chains move past one another.
A polymer is a large molecule built from repeating smaller units. In many polymers, atoms are connected along long chains by strong covalent bonds. However, the chains do not exist in isolation. They may attract each other through intermolecular forces such as dispersion forces, dipole-dipole attractions, or hydrogen bonding. If these attractions are weak, the chains can shift more easily and the material may be softer or more flexible. If the attractions are stronger, the chains resist movement and the material may be tougher or stiffer.
Intermolecular forces are weaker than covalent bonds, but they still matter because a material contains huge numbers of molecules. When many weak attractions act together, their total effect can strongly influence melting point, elasticity, and strength.
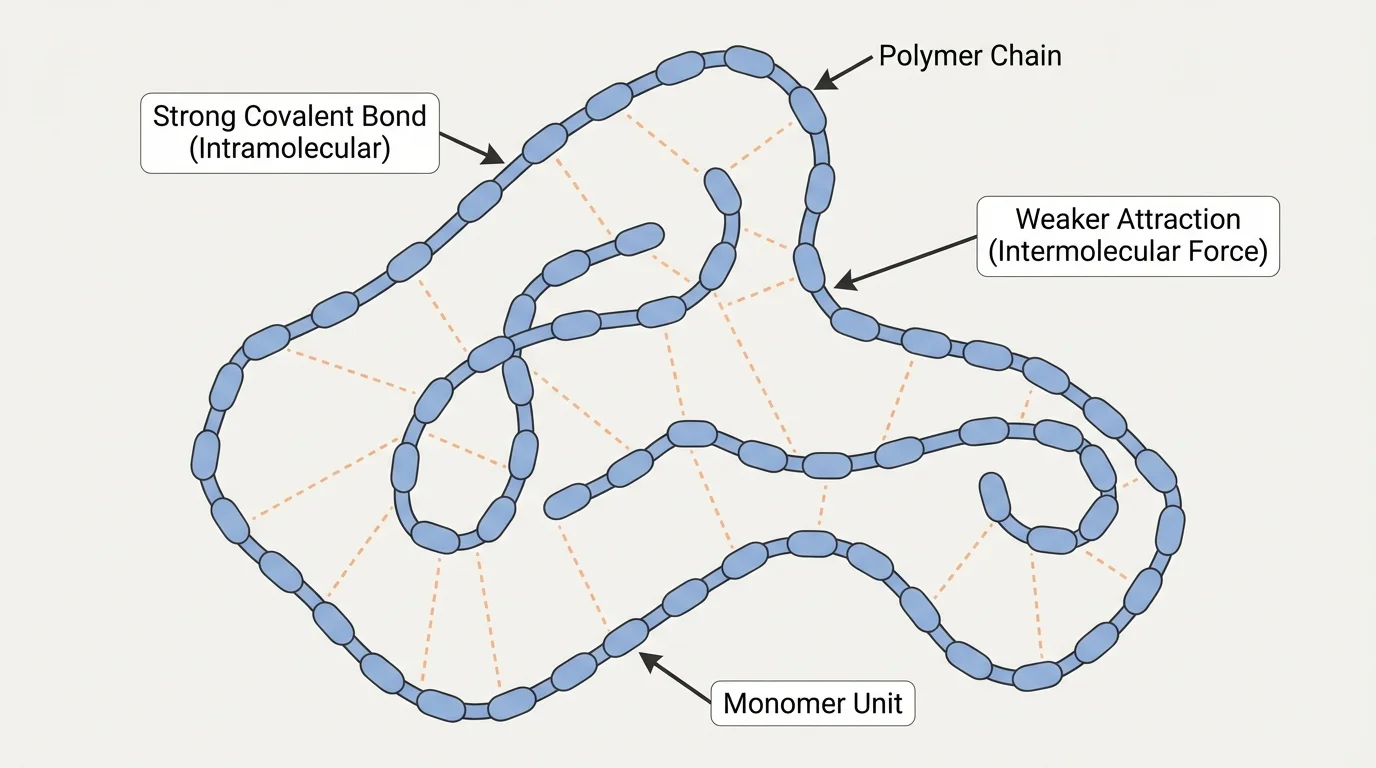
Molecular shape also matters. Long straight chains may pack close together, increasing attraction between chains. Bent or irregular molecules may not pack as neatly, which can lower density or make a material softer. This is one reason molecular-level structure is so important: tiny changes in shape can produce large changes in behavior that we can see and feel.
One useful relationship involving electric interactions is the force between charges. Opposite charges attract and like charges repel, and the magnitude of that electric force can be described by Coulomb's law:
\[F = k\frac{|q_1q_2|}{r^2}\]
Here, \(F\) is electric force, \(q_1\) and \(q_2\) are charges, \(r\) is the distance between them, and \(k\) is a constant. If two charged parts of molecules are closer together, the force becomes stronger because \(F\) increases as \(r\) decreases. For example, if the distance changes from \(r = 2\) units to \(r = 1\) unit while the charges stay the same, then the force changes by a factor of \(\dfrac{1}{1^2} \div \dfrac{1}{2^2} = 4\). The attraction becomes four times stronger. This helps explain why closely packed polar regions of molecules can interact strongly.
Many high-performance materials are powerful not because they contain rare atoms, but because ordinary atoms are arranged in unusually effective ways. Structure often matters as much as composition.
As later examples show, changing how molecules line up can transform a material from ordinary to exceptionally strong.
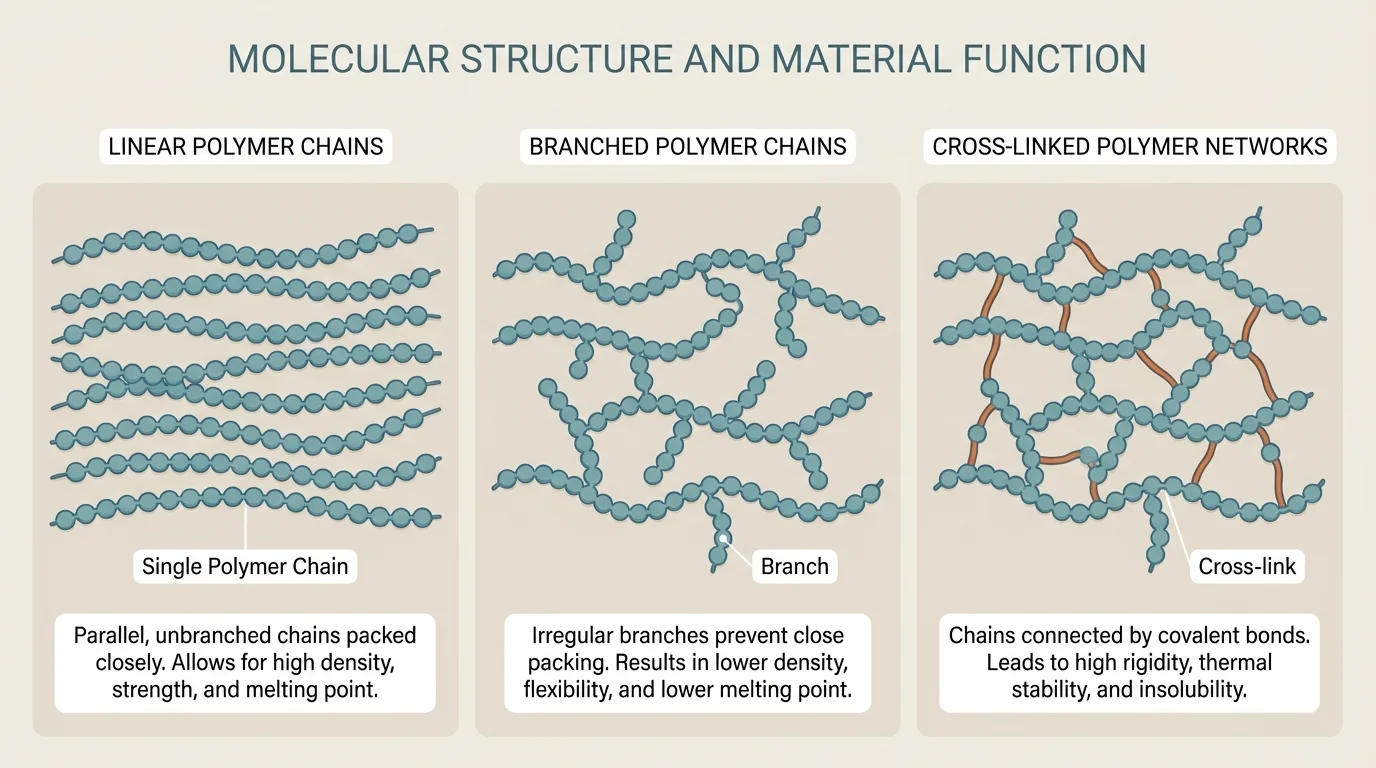
Not all polymers have the same internal arrangement. The arrangement of chains changes flexibility, strength, and thermal behavior, as [Figure 2] shows by comparing linear, branched, and cross-linked structures.
Linear polymers have long chains with little branching. Because the chains can often pack more closely, they may have stronger attractions between chains and greater strength or density. Branched polymers include side chains that interfere with close packing, often making the material less dense and more flexible. Cross-linked polymers contain connections between chains, creating a network that limits movement. This can increase stiffness, shape retention, and heat resistance.
A related structure is a network solid, in which atoms are linked in a broad, continuous structure rather than existing as separate small molecules. In designed materials such structures often lead to hardness and high melting temperature. Glasses and some ceramics are examples of materials where extensive bonding patterns strongly affect performance, especially in heat resistance and rigidity.
Another key idea is crystalline region. In some materials, parts of the structure are ordered and regular, while other parts are more disordered. More ordered regions usually let particles pack tightly, which can increase stiffness and strength. More disordered regions often increase flexibility. Many useful plastics combine both kinds of regions, which helps them balance toughness and bendability.
When scientists describe a material, they often explain whether it has long chains, branching, cross-links, or ordered packing. These details are not decoration. They are evidence for why the material behaves the way it does.
Structure-property-function reasoning is a cause-and-effect pattern used throughout materials science. A clear explanation follows this sequence: identify a structural feature, explain how that feature affects particle interactions or motion, then connect that effect to a useful property and finally to the material's job in a design.
For example, if a provided structure shows many cross-links between chains, a student can reasonably argue that the chains cannot slide easily, so the material is more rigid and keeps its shape well. That property makes it useful for products that must resist deformation.
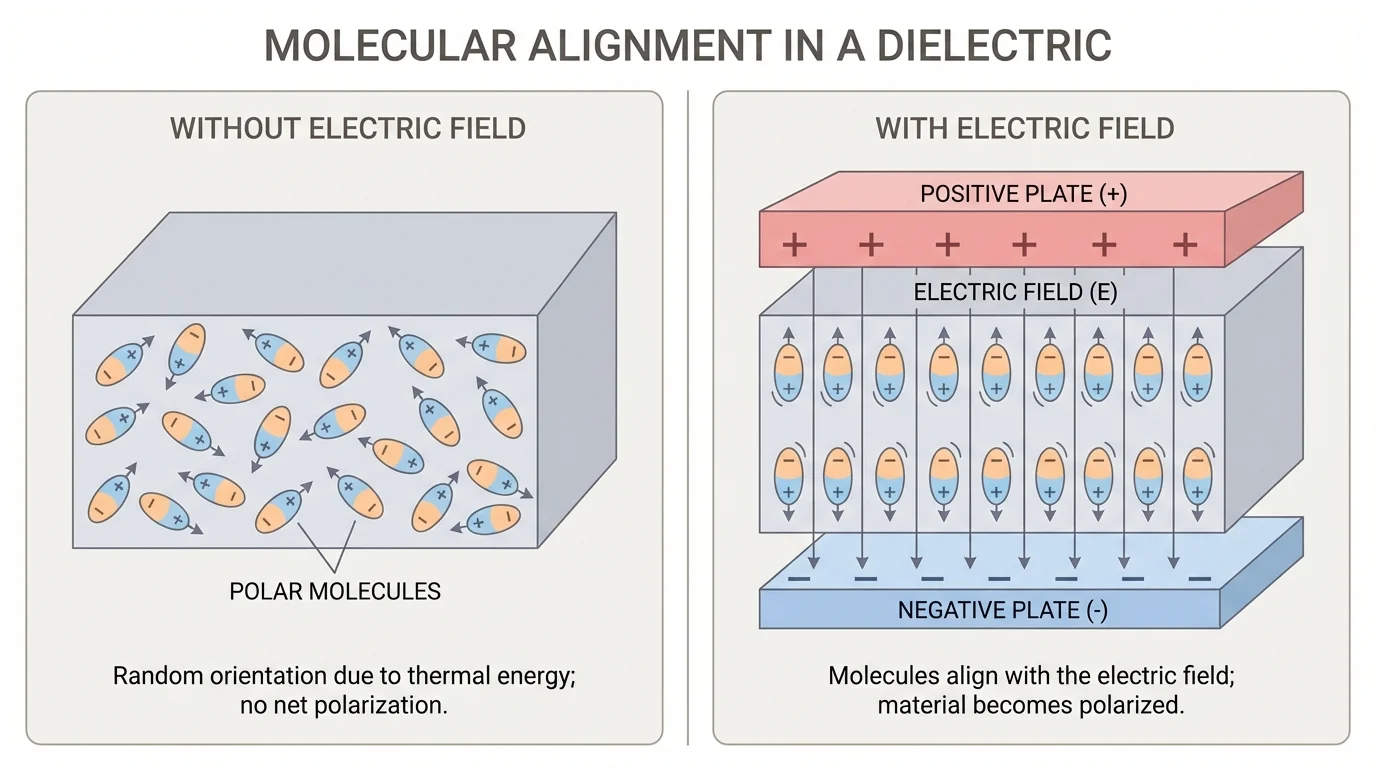
[Figure 3] Some materials function the way they do because of electric fields and other forces that act over distance. An electric field can interact with charged particles or with parts of molecules that have uneven charge distribution. In a dielectric material, for instance, the charges do not flow freely as they do in a metal, but the electron clouds and polar molecules can shift slightly in response to the field.
This shifting is called polarization. Polarization matters in capacitors, touchscreens, cable insulation, and many electronic components. A material with molecules that can reorient or shift charge in response to an electric field may store electrical energy effectively while still acting as an insulator.
The electrical energy stored in the electric field inside a capacitor depends partly on the material between the plates. A simple relationship is
\(E = qV\)
where \(E\) is electric energy, \(q\) is charge, and \(V\) is electric potential difference. If a capacitor holds \(q = 2 \textrm{ C}\) across \(V = 3 \textrm{ V}\), then \(E = 2 \times 3 = 6 \textrm{ J}\). Materials that polarize effectively can improve how such devices function by helping the system store energy.
Fields also help explain why water may spread on one surface but bead up on another. Surface interactions depend on charge distribution and attraction between molecules. If a coating has a structure that minimizes attraction to water, droplets remain more rounded and roll away more easily. That is useful in water-repellent fabrics and self-cleaning surfaces.
Later, when discussing nonstick and water-resistant materials, the alignment of charge and weak attraction patterns introduced in [Figure 3] becomes essential for explaining why some surfaces repel polar substances better than others.
Specific molecular structures explain why materials perform differently in real products. A strong explanation identifies a visible structural feature and connects it directly to function, as the high-strength polymer example shows.
Polyethylene is widely used in bags, bottles, and containers. Its repeating carbon chain structure can appear more linear or more branched depending on the type. More linear polyethylene packs more tightly, often giving higher density and greater stiffness. More branched polyethylene packs less efficiently, making it softer and more flexible. This is why not all plastic bags or bottles feel the same even when they are made from related materials.
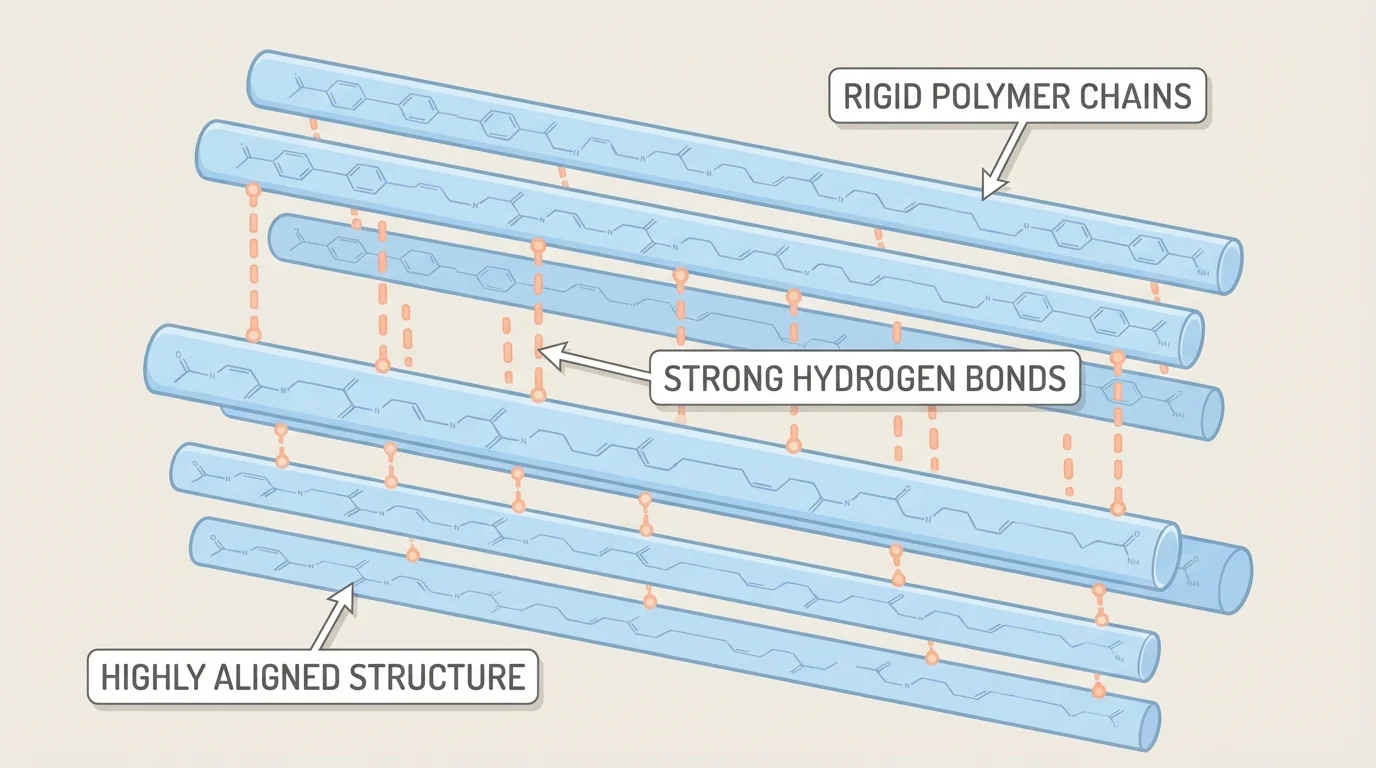
[Figure 4] Kevlar is used in protective gear because its molecular structure contains stiff, rod-like chains that align well. Strong attractions between neighboring chains help prevent them from sliding apart under tension. The result is a material with very high tensile strength for its mass.
Silicone materials are useful in sealants, bakeware, and medical devices. Their structures allow flexibility over a wide temperature range. In many silicones, the backbone and side groups lead to chain mobility, so the material remains bendable while also resisting water and heat better than many ordinary plastics.
Polytetrafluoroethylene, often known for nonstick coatings, has a structure surrounded by fluorine atoms. This arrangement creates a very low-friction surface and weak interaction with many other substances. As a result, it is useful where sticking and chemical reactivity must be reduced.
Hydrogels are polymer networks that absorb large amounts of water. Their structures include hydrophilic groups that attract water and cross-links that keep the material from dissolving completely. Because of this combination, hydrogels can be soft and wet while still holding a shape, making them useful in contact lenses, wound dressings, and drug delivery systems.
Case study: explaining a hydrogel
A provided structure shows a cross-linked polymer with several oxygen-containing groups that attract water.
Step 1: Identify the structural features
The structure contains cross-links between chains and polar groups such as bonds involving oxygen. Those features suggest both shape retention and attraction to water.
Step 2: Connect structure to particle behavior
The polar groups attract water molecules, while the cross-links stop the chains from drifting apart completely.
Step 3: State the resulting properties and function
The material swells, stays soft, and remains intact. That makes it useful in products such as contact lenses or wound dressings.
The tightly aligned arrangement discussed for Kevlar remains a powerful example of structure-based reasoning; the same alignment seen in [Figure 4] helps explain why the material resists stretching much better than ordinary flexible plastics.
Being able to understand materials is important, but being able to communicate about them is equally important. In science and engineering, communication means making a claim and supporting it with evidence and reasoning. If you are given a molecular structure, your explanation should answer three questions: What do you observe in the structure? How does that affect interactions or motion at the molecular scale? Why does that matter for the material's job?
A strong technical explanation might sound like this: "The provided polymer has many cross-links between chains. Because the chains are connected, they cannot move freely past one another. This increases rigidity and helps the material keep its shape under stress, so it is useful for a durable molded product." Notice that the explanation does not stop at naming the structure. It links structure to behavior and then to function.
Weak explanations are usually too vague. For example, saying "this material is strong because of its molecules" does not communicate enough. Strong explanations identify which part of the structure matters and how it changes the material.
| Observed structural feature | Likely effect on particle behavior | Possible material property | Possible function |
|---|---|---|---|
| Long linear chains | Closer packing and greater attraction between chains | Higher strength or density | Sturdy container or film |
| Many branches | Less efficient packing | More flexibility, lower density | Soft packaging |
| Cross-links | Chains cannot slide easily | Rigidity, shape retention | Seal, handle, molded part |
| Polar groups | Stronger attraction to polar substances such as water | Water absorption or adhesion | Hydrogel, coating, medical material |
| Very low-attraction outer atoms | Reduced sticking at surfaces | Low friction, nonstick behavior | Cookware coating, protective surface |
Table 1. A structure-property-function guide showing how visible molecular features can support explanations about designed materials.
Scientists also use careful comparison words such as because, therefore, as a result, and this suggests. These words signal reasoning, not just description.
Earlier chemistry learning about bonding, polarity, and attraction between particles is essential here. Materials science builds on those ideas by asking how many particles acting together create large-scale behavior.
If a material is being evaluated for electronics, fields and charge distribution may become central to the explanation. If it is being evaluated for protective gear, chain alignment, bonding, and resistance to stretching may matter more. Communication should match the evidence in the provided structure and the function being discussed.
No material is best at everything. A very rigid material may crack instead of bend. A very soft material may be comfortable but not protective. A water-absorbing material may be useful in medicine but unsuitable for electronics. Engineers often face trade-offs and must choose the structure that gives the right balance of properties.
For example, adding more cross-links can make a polymer stronger and better at keeping its shape, but too many cross-links can also reduce flexibility. Increasing order in a material may improve strength, yet it can also make processing harder. This is why material design is not simply about making something "stronger." It is about matching structure to the demands of the application.
Comparing design choices
Suppose two provided materials are being considered for a flexible phone case.
Step 1: Material A has heavy cross-linking
This suggests high rigidity and good shape retention, but less ability to bend comfortably around the phone.
Step 2: Material B has long chains with fewer cross-links
This suggests more chain motion, so the material is more flexible and can absorb some impact by deforming.
Step 3: Match to function
For a flexible protective case, Material B is likely a better choice because its molecular structure supports bending and energy absorption.
Good communication includes these design trade-offs. Engineers justify choices by showing that a structure gives the most appropriate combination of properties for the job.
In medicine, molecular-level structure helps determine whether a material can safely contact tissue, absorb water, or release medicine slowly. In transportation, lightweight polymers and composites reduce mass while maintaining strength. In sports equipment, molecular design can improve impact resistance, grip, and durability. In electronics, insulating materials and dielectric layers depend on charge distribution and response to electric fields.
Protective clothing offers a clear example. Fibers used in helmets, gloves, and vests are chosen not just because they are "strong," but because their structures resist stretching, tearing, or heat transfer in specific ways. Water-resistant jackets depend on surface chemistry and low attraction to water. Flexible cables depend on polymers that insulate electrically but do not crack easily when bent.
Even sustainable design depends on molecular structure. A material that is durable may reduce replacement waste, while another material may be chosen because its structure supports recycling or controlled breakdown. Function, environment, cost, and safety all matter together.
"Materials do not simply have properties; their properties come from structure."
— Central idea of materials science
When you communicate scientific and technical information about designed materials, the goal is to make the invisible visible through reasoning. You look at a provided structure, identify meaningful features, explain the interactions those features create, and connect them to the material's performance in a real design.