A beam of light can carry your internet through fiber-optic cables, warm food in a microwave oven, reveal broken bones in an X-ray image, and trigger electricity in a solar panel. That is impressive. A more surprising fact is that the same electromagnetic radiation can be described in two different ways: as a wave and as a stream of photons. At first, that sounds contradictory. In science, though, a model is a tool, and different tools can be useful for different jobs. The real challenge is not choosing one side forever. It is learning how to evaluate the claims, evidence, and reasoning behind each model.
Electromagnetic radiation includes radio waves, microwaves, infrared, visible light, ultraviolet, X-rays, and gamma rays. These forms differ in wavelength and frequency, but they all transfer energy without needing a material medium. That is why sunlight travels across space to Earth. Understanding how to describe this radiation helps explain both natural phenomena and many modern technologies.
Electromagnetic radiation is energy that travels through space as changing electric and magnetic fields. It moves at the speed of light in a vacuum, approximately \(3.0 \times 10^8 \textrm{ m/s}\). A useful relationship connects speed, wavelength, and frequency:
\[c = f\lambda\]
Here, \(c\) is the speed of light, \(f\) is frequency, and \(\lambda\) is wavelength. If frequency increases, wavelength decreases, because the product stays equal to \(c\) in a vacuum.
From earlier studies of waves, remember that wavelength is the distance from one crest to the next, frequency is the number of waves passing a point each second, and amplitude relates to the size of the oscillation. These ideas still matter when discussing electromagnetic waves.
For example, if a radio wave has frequency \(1.0 \times 10^8 \textrm{ Hz}\), then its wavelength is \(\lambda = \dfrac{c}{f} = \dfrac{3.0 \times 10^8}{1.0 \times 10^8} = 3.0 \textrm{ m}\). A much higher-frequency wave, such as visible light, has a much shorter wavelength. This simple relationship shows that different parts of the electromagnetic spectrum have different wavelengths and frequencies, which helps explain why they behave differently in real situations.
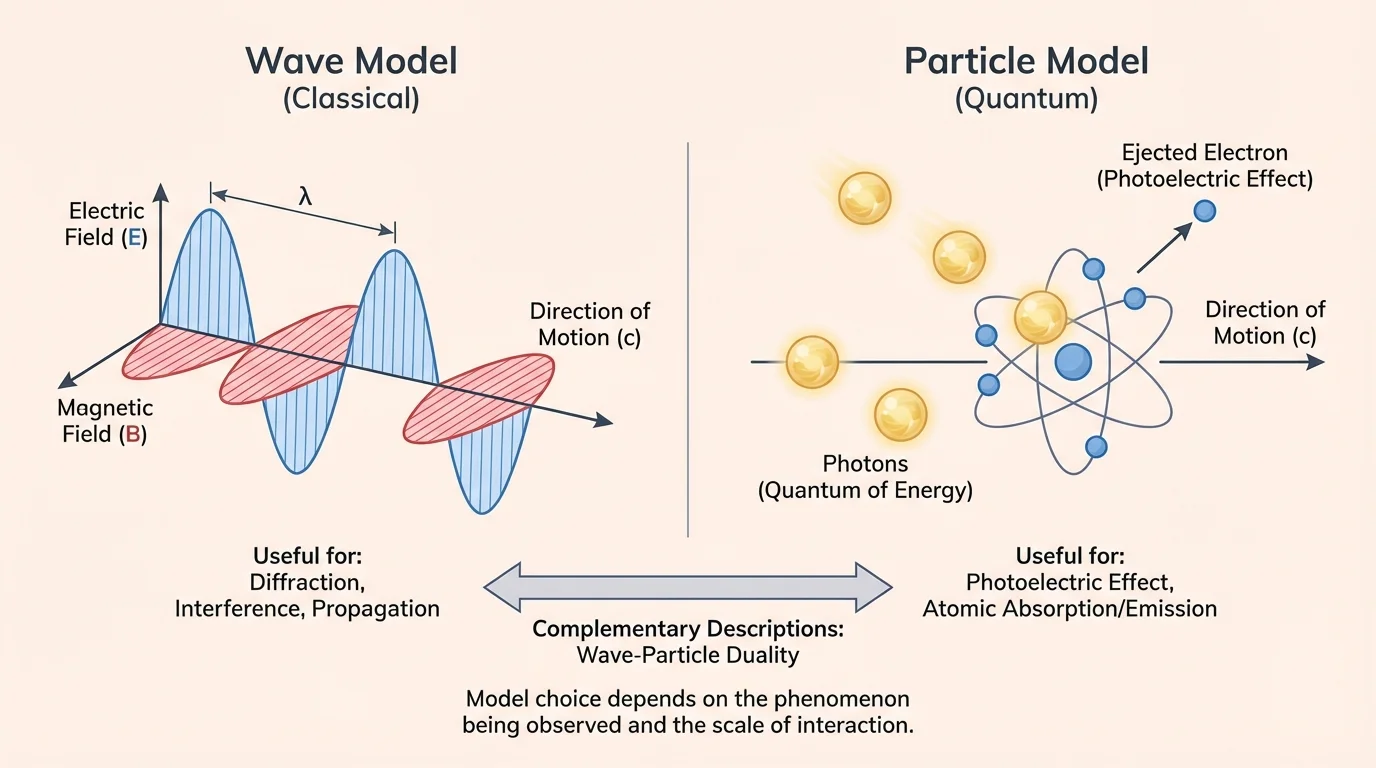
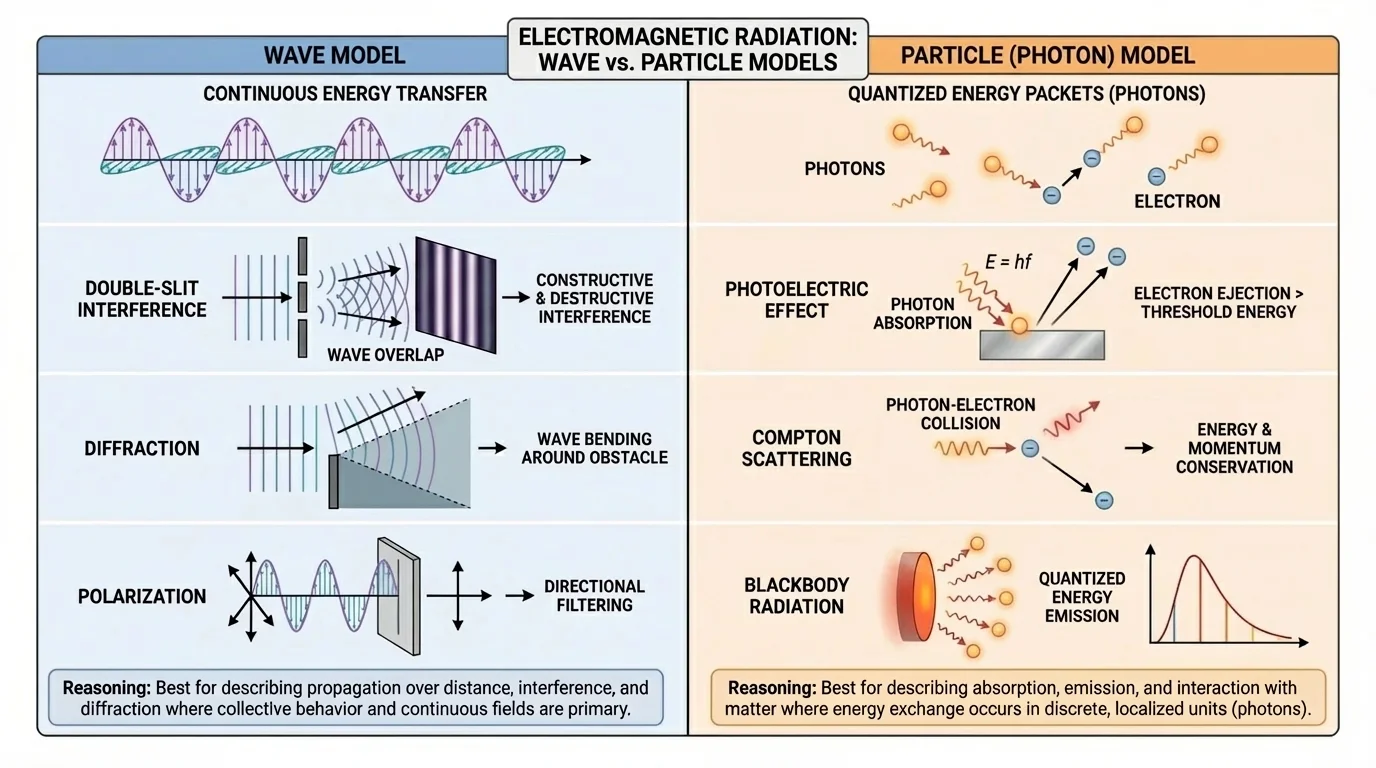
The wave model describes electromagnetic radiation as a wave made of oscillating electric and magnetic fields. [Figure 1] These fields are perpendicular to each other and to the direction the wave travels. This model is especially powerful when we want to explain patterns produced by overlapping waves or how electromagnetic radiation spreads through space.
In the wave model, several properties matter. Wavelength and frequency describe the wave's size and timing. Amplitude is related to the energy carried by the wave. The wave model helps explain interference, which happens when two or more waves overlap, and diffraction, which is the bending and spreading of waves around edges or through openings. It also explains polarization, a property that applies to transverse waves such as light.
Suppose two light waves meet. If their peaks line up with peaks, they produce brighter light through constructive interference. If peaks line up with troughs, they can reduce brightness through destructive interference. This kind of behavior is difficult to explain if light is treated only as tiny independent particles with no wave properties. The wave model naturally predicts these results.
The wave model is also useful for understanding antennas, radio broadcasting, and wireless communication. A radio station creates electromagnetic waves of specific frequencies. A receiver detects those waves and extracts the information they carry. Engineers often think in terms of wavelength, frequency, reflection, refraction, and interference because these ideas predict how signals travel through air, bounce off buildings, and weaken over distance.
Why the wave model is powerful
A good scientific model does not need to explain everything to be valuable. The wave model is powerful because it successfully explains how electromagnetic radiation spreads, combines, bends, and forms patterns. Whenever the question involves wave behavior such as interference, diffraction, or polarization, this model gives strong predictions that match observations.
One familiar example is noise-canceling technology in sound, which uses destructive interference. Light can also interfere, although we often need special setups to notice it clearly. Thin films like soap bubbles and oil slicks display color patterns because reflected light waves interfere. The colors shift as the thickness of the film changes.
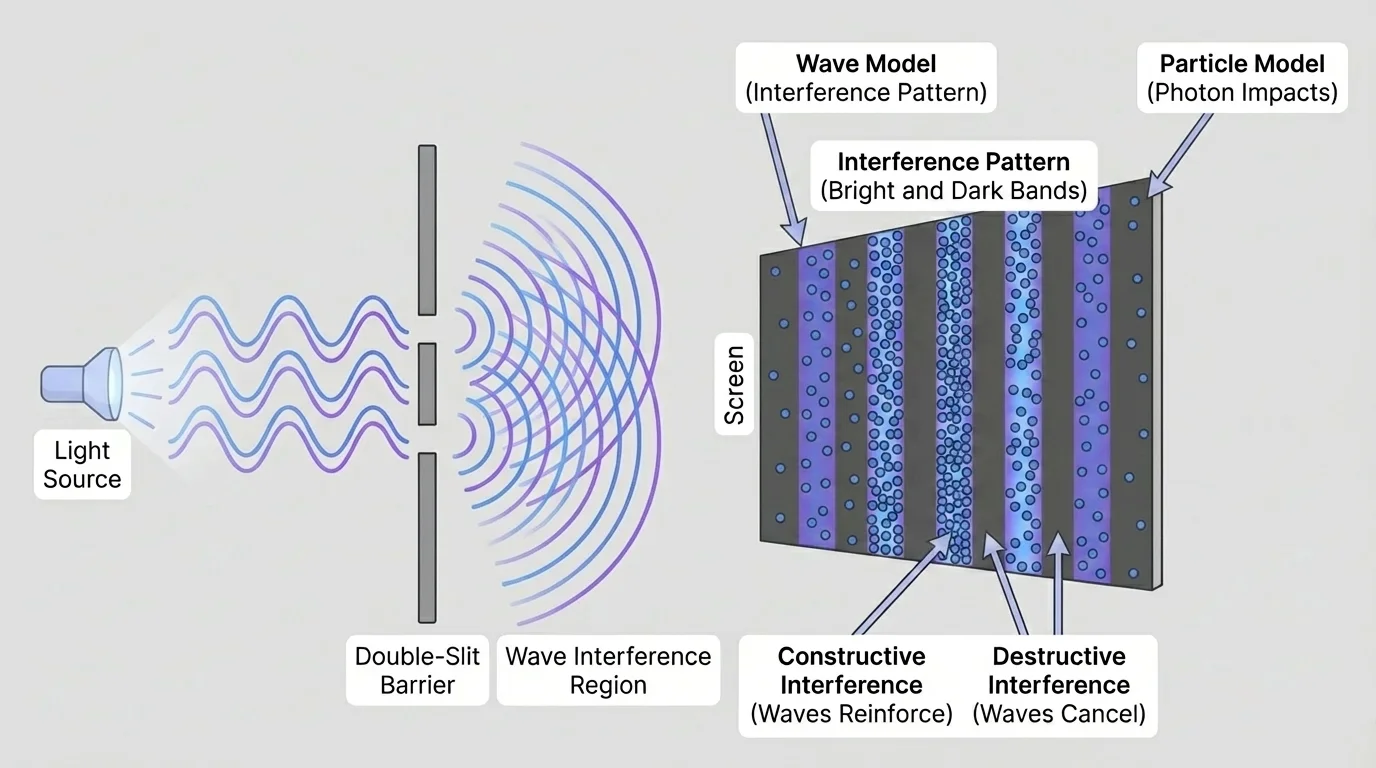
[Figure 2] A major claim of the wave model is that light behaves like a wave. Strong evidence comes from experiments that produce interference patterns. When light passes through two narrow slits, alternating bright and dark bands appear on a screen. Bright bands form where waves reinforce one another, and dark bands form where they cancel.
This pattern matters because it is not random. It follows the reasoning expected for waves: if two waves travel different distances, their peaks and troughs reach the screen at slightly different times. Some positions on the screen receive constructive interference, and some receive destructive interference. The pattern is direct evidence that wave ideas are useful and predictive.
Another line of evidence comes from diffraction. Light spreads after passing through a very small opening, and the amount of spreading depends on wavelength. Radio waves, which have long wavelengths, can bend around obstacles more effectively than visible light. That is one reason radio signals can sometimes be detected even when the transmitter is not in direct view.
Polarization is also important evidence. Polarizing sunglasses reduce glare by blocking light waves oscillating in certain directions. This makes sense if light behaves as a transverse wave. The wave model gives a clear explanation for why filters can allow one orientation of oscillation to pass while blocking another.
The photon model describes electromagnetic radiation as made of discrete packets of energy called photons. In this description, light interacts with matter in individual energy transfers, as shown in the case of light striking a metal surface and ejecting electrons. This model becomes especially useful when the question is about how radiation is absorbed, emitted, or detected.
A key relationship for photon energy is
\(E = hf\)
[Figure 3] Here, \(E\) is photon energy, \(h\) is Planck's constant, and \(f\) is frequency. This equation shows that higher-frequency radiation carries more energy per photon. This idea is especially useful when explaining how light transfers energy to matter. That is why X-rays and ultraviolet radiation can have stronger effects on matter than radio waves or microwaves.
For a simple calculation, compare two kinds of radiation. If one photon has frequency \(5.0 \times 10^{14} \textrm{ Hz}\), then its energy is \(E = hf = (6.63 \times 10^{-34})(5.0 \times 10^{14}) \approx 3.3 \times 10^{-19} \textrm{ J}\). If frequency doubles, photon energy doubles too. The photon model focuses on these energy packets, not on continuous spreading alone.
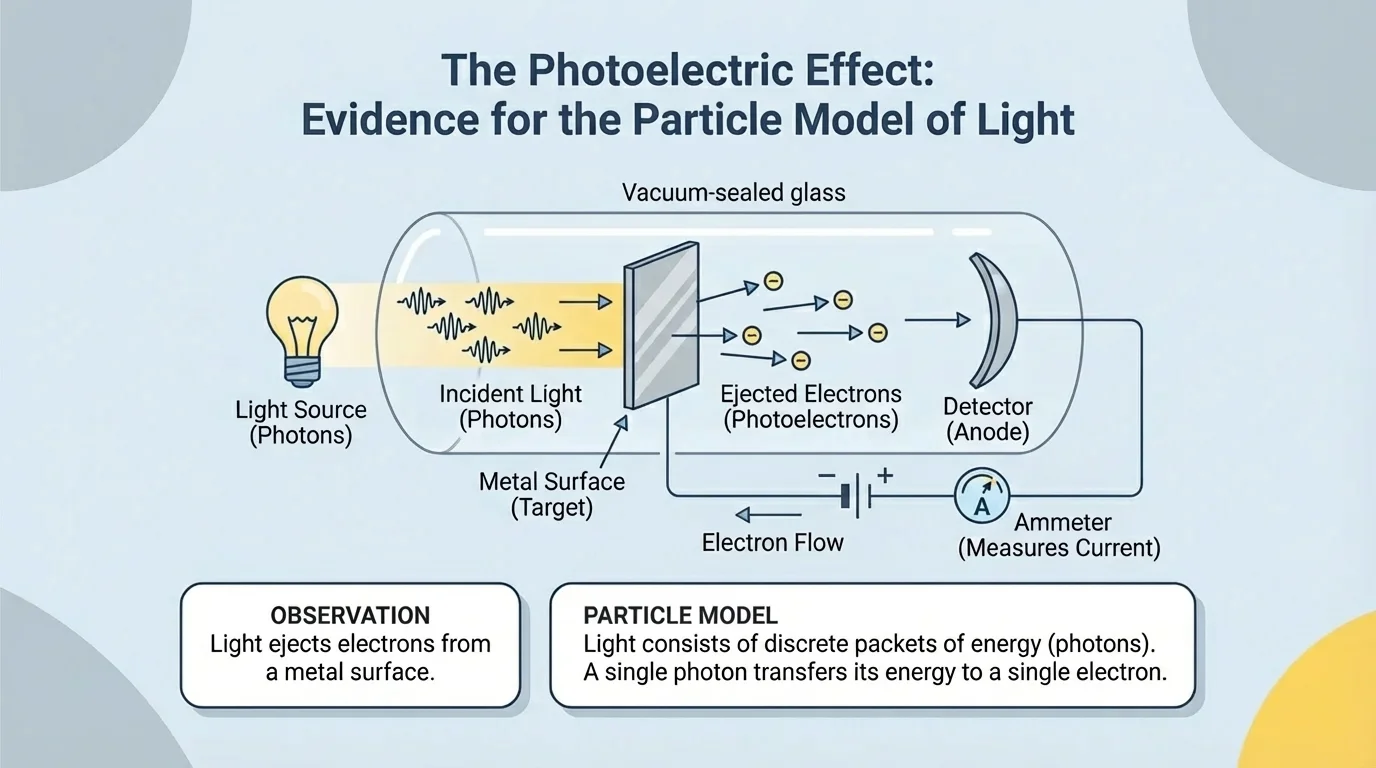
This model helps explain the photoelectric effect, where light shining on certain metals can eject electrons. The important observation is that below a certain frequency, no electrons are emitted, even if the light is very bright. But above that frequency, electrons are emitted. Reasoning with photons explains this well: each electron must receive enough energy from a single interaction. Brighter low-frequency light provides more photons, but each photon still carries too little energy.
Numeric example: comparing photon energies
Consider red light with frequency \(4.0 \times 10^{14} \textrm{ Hz}\) and ultraviolet light with frequency \(1.0 \times 10^{15} \textrm{ Hz}\).
Step 1: Use the photon energy formula
\(E = hf\)
Step 2: Calculate the red-light photon energy
\(E = (6.63 \times 10^{-34})(4.0 \times 10^{14}) \approx 2.65 \times 10^{-19} \textrm{ J}\)
Step 3: Calculate the ultraviolet photon energy
\(E = (6.63 \times 10^{-34})(1.0 \times 10^{15}) \approx 6.63 \times 10^{-19} \textrm{ J}\)
Each ultraviolet photon carries much more energy than each red-light photon. That difference helps explain why ultraviolet radiation can damage skin cells more easily.
The photon model is also useful for solar panels. In a solar cell, incoming light transfers energy to electrons in the material. Thinking in terms of photons helps explain why some frequencies are more effective than others at producing electric current. The same idea applies to digital camera sensors, where light is detected through many tiny energy-transfer events.
One important claim of the photon model is that electromagnetic radiation transfers energy in discrete amounts. Evidence comes from the photoelectric effect, where electron emission depends on frequency more than brightness. This is not what a simple continuous-wave energy picture would predict. The evidence supports the reasoning that light sometimes acts as if it arrives in separate energy packets.
Another source of evidence is line spectra from glowing gases. When a gas is energized, it emits only certain wavelengths rather than all possible wavelengths. At this stage, the key point is that matter absorbs and emits electromagnetic radiation in discrete amounts, and the photon model helps describe those exchanges. We do not need deeper quantum theory here to see that the particle description is useful.
Remote controls commonly use infrared radiation, not visible light. The receiving device detects bursts of electromagnetic radiation carrying coded information, showing that the same kind of radiation can be treated as a wave for transmission and as photons when detected by electronics.
[Figure 4] Medical imaging also gives practical evidence about photon energy. Different situations are often explained more effectively by different models. X-rays pass through soft tissue more easily than bone because of how they interact with matter. Discussing those interactions often works best with the photon model, since each X-ray photon carries enough energy to affect materials differently depending on their composition and density.
Scientists do not usually ask, "Which model is the one true picture in every situation?" A better question is, "Which model explains the evidence more effectively for this problem?" That comparison is central to evaluating scientific claims. Models are judged by how well they explain observations and predict outcomes.
The wave model is more useful when discussing propagation, interference, diffraction, polarization, and communication signals. The photon model is more useful when discussing energy transfer between radiation and matter, such as emission, absorption, detectors, solar cells, and the photoelectric effect.
| Situation | More useful model | Why it helps |
|---|---|---|
| Double-slit pattern | Wave model | Explains bright and dark bands through interference |
| Radio broadcasting | Wave model | Uses frequency, wavelength, and signal propagation |
| Polarizing sunglasses | Wave model | Depends on wave orientation |
| Photoelectric effect | Photon model | Explains threshold frequency and discrete energy transfer |
| Solar panels | Photon model | Describes light transferring energy to electrons |
| X-ray interactions with tissue | Photon model | Focuses on energy per photon and matter interaction |
Table 1. Situations in which either the wave model or the photon model is more useful for explaining electromagnetic radiation.
This does not mean one model is wrong and the other is right in a simple all-or-nothing sense. Instead, it means each model has strengths. As with maps, one map may show roads while another shows elevation. Neither tells everything, but each is useful for a particular purpose. In science, models are accepted because they work well within their scope.
The double-slit pattern we discussed earlier still stands as strong evidence for wave behavior, as seen in [Figure 2]. By contrast, the photoelectric effect still stands as strong evidence that particle-like energy transfer matters, as introduced in [Figure 3]. Together, these observations push us to use both models intelligently.
Electromagnetic radiation is not just a topic in physics; it is built into daily life and advanced technology. Cell phone networks, Wi-Fi, and satellite communication depend heavily on wave ideas such as frequency, modulation, reflection, and interference. Engineers choose frequencies carefully because wavelength affects how signals travel and what obstacles they can pass around.
Fiber-optic communication also relies on wave behavior. Light reflects repeatedly inside thin glass fibers, allowing information to travel long distances with low loss. At the same time, the detectors at the receiving end respond to light through discrete interactions, reminding us that both descriptions can matter in one system.
In medicine, X-rays are useful for imaging bones because they interact differently with various tissues. Ultraviolet radiation can help sterilize surfaces by damaging microorganisms. Infrared cameras detect heat-related radiation from objects and are used in firefighting, engineering inspections, and environmental studies. In astronomy, telescopes collect electromagnetic radiation across the spectrum, and scientists choose the model that best fits the data they are analyzing.
Numeric example: wavelength from frequency
A microwave oven commonly uses radiation with frequency about \(2.45 \times 10^9 \textrm{ Hz}\). Find its wavelength in air, using \(c = 3.0 \times 10^8 \textrm{ m/s}\).
Step 1: Start with the wave relationship
\(c = f\lambda\)
Step 2: Rearrange for wavelength
\(\lambda = \dfrac{c}{f}\)
Step 3: Substitute values
\(\lambda = \dfrac{3.0 \times 10^8}{2.45 \times 10^9} \approx 1.22 \times 10^{-1} \textrm{ m}\)
The wavelength is about \(0.122 \textrm{ m}\), or about \(12.2 \textrm{ cm}\).
Microwave ovens are a good reminder that electromagnetic radiation transfers energy. Water molecules in food absorb microwave energy efficiently, heating the food. The oven design also depends on wave reflections inside the chamber, so this is another case where the wave model is useful for one aspect and the interaction with matter is important for another.
To evaluate scientific ideas well, separate three parts: the claim, the evidence, and the reasoning. A claim is the statement being made, such as "light behaves like a wave" or "light transfers energy as photons." Evidence is the observation or data, such as interference bands or threshold frequencies in the photoelectric effect. Reasoning explains why the evidence supports the claim.
Consider the claim that electromagnetic radiation can be described by a wave model. Evidence includes interference, diffraction, and polarization. The reasoning is that these behaviors are characteristic of waves, and the wave model correctly predicts them. Therefore, the claim is strongly supported in those contexts.
Now consider the claim that electromagnetic radiation can be described by a particle model. Evidence includes the photoelectric effect and the dependence of energy transfer on frequency. The reasoning is that discrete packets of energy explain why increasing brightness alone does not always eject electrons, while increasing frequency can. Therefore, the photon model is strongly supported in situations involving radiation interacting with matter.
A strong evaluation uses scope
One of the most important reasoning skills in science is recognizing the scope of a model. Saying "the wave model is useful" is stronger than saying "the wave model explains everything." Saying "the photon model explains certain energy-transfer phenomena" is stronger than claiming it replaces all wave ideas. Good scientific reasoning matches the model to the evidence and the question being asked.
That is why the most accurate overall claim is not that electromagnetic radiation is only a wave or only a particle in every classroom-level discussion. A better claim is that both models explain broad features of electromagnetic radiation, and one model may be more useful than the other depending on the situation. The comparison chart in [Figure 4] helps organize that judgment.
Even the basic formulas support this idea. The wave relationship \(c = f\lambda\) helps analyze signal behavior, wavelength, and frequency. The photon relationship \(E = hf\) helps analyze energy carried by individual light packets. Both formulas are valid tools, but they answer different kinds of questions.
Choosing the better model
A student is asked two questions: why a soap bubble shows shifting colors, and why ultraviolet light can trigger electron emission from a metal more effectively than red light.
Step 1: Identify the first phenomenon
Soap-bubble colors come from reflected light waves combining. The best model is the wave model because interference explains the changing colors.
Step 2: Identify the second phenomenon
Electron emission depends on how much energy each light packet carries. The best model is the photon model because higher frequency means higher photon energy through \(E = hf\).
Step 3: Evaluate the reasoning
The chosen model should match the evidence. Interference evidence supports the wave model, while threshold-energy evidence supports the photon model.
This is exactly how scientists use models: not by guessing, but by connecting claims to evidence with clear reasoning.
As shown earlier in [Figure 1], the wave picture remains essential for visualizing how electric and magnetic fields travel together. But when radiation transfers energy to electrons in a detector or metal surface, the particle description introduced with [Figure 3] is often the better tool. The strength of science here is not choosing one image forever. It is using evidence to choose the best explanation for the situation.