One headline says that microwaves are dangerous because they are "radiation." Another says sunlight is healthy, while a different article warns that part of sunlight can damage DNA. Hospitals use X-rays to diagnose and treat patients, yet everyone is told not to get unnecessary X-ray exposure. These statements are not all equally strong, and they do not all mean the same thing. To judge them well, you need both physics and critical thinking.
Electromagnetic radiation is part of daily life. It carries music to your car radio, heats food in a microwave oven, lets you see colors, powers fiber-optic communication, helps doctors diagnose injuries, and can also damage cells under certain conditions. The key scientific question is not simply whether radiation exists, but what frequency it has, how much energy is absorbed, what kind of matter absorbs it, and what evidence supports the claim being made.
Published materials include textbooks, news articles, advertisements, company websites, social media posts, and scientific papers. They do not all use evidence equally well. A scientifically strong claim must match what is known about waves and photons, and it must be supported by trustworthy observations or experiments.
People often react emotionally to the word electromagnetic radiation. That is understandable, because the word appears in discussions of cancer, nuclear medicine, wireless devices, and space science. But electromagnetic radiation is a broad category. It includes low-frequency radio waves and very high-frequency gamma rays. Treating all of it as equally dangerous is a scientific mistake.
A better approach is to ask precise questions. What part of the spectrum is involved? Is the radiation being absorbed or mostly reflected? Does the absorbed energy mainly make molecules vibrate and warm up, or can it remove electrons from atoms? Is the source close or far away? Was the claim tested in humans, in isolated cells, or only in computer models? Good evaluation begins with careful questions, not quick reactions.
Waves carry energy. Electromagnetic waves do not need matter to travel, unlike sound waves. You may also recall that light can be described both as a wave and as packets of energy called photons. Both models help explain how radiation behaves and how it interacts with matter.
This topic also matters because many real decisions depend on it: using sunscreen, limiting unnecessary medical imaging, choosing protective equipment for welding, understanding why your phone uses radio waves, and judging online claims about "radiation exposure."
The electromagnetic spectrum, shown in [Figure 1], arranges electromagnetic radiation from low frequency to high frequency: radio waves, microwaves, infrared, visible light, ultraviolet, X-rays, and gamma rays. As frequency increases across the spectrum, the wavelength becomes shorter, and each photon carries more energy.
There are two useful ways to think about this. In the wave model, electromagnetic radiation has wavelength and frequency. In the photon model, radiation comes in packets called photons. A key relationship is that photon energy increases with frequency: \(E = hf\), where \(E\) is photon energy, \(h\) is Planck's constant, and \(f\) is frequency. For example, if frequency doubles, photon energy doubles. You do not need detailed calculations here; the important idea is qualitative: higher-frequency radiation can produce different effects because each photon carries more energy.
This does not mean that every high-frequency source is automatically dangerous in every situation. It means that frequency affects what kinds of interactions are possible. Low-frequency radiation is often associated with communication and bulk heating. Higher-frequency radiation can trigger chemical changes or ionization if enough energy is absorbed.
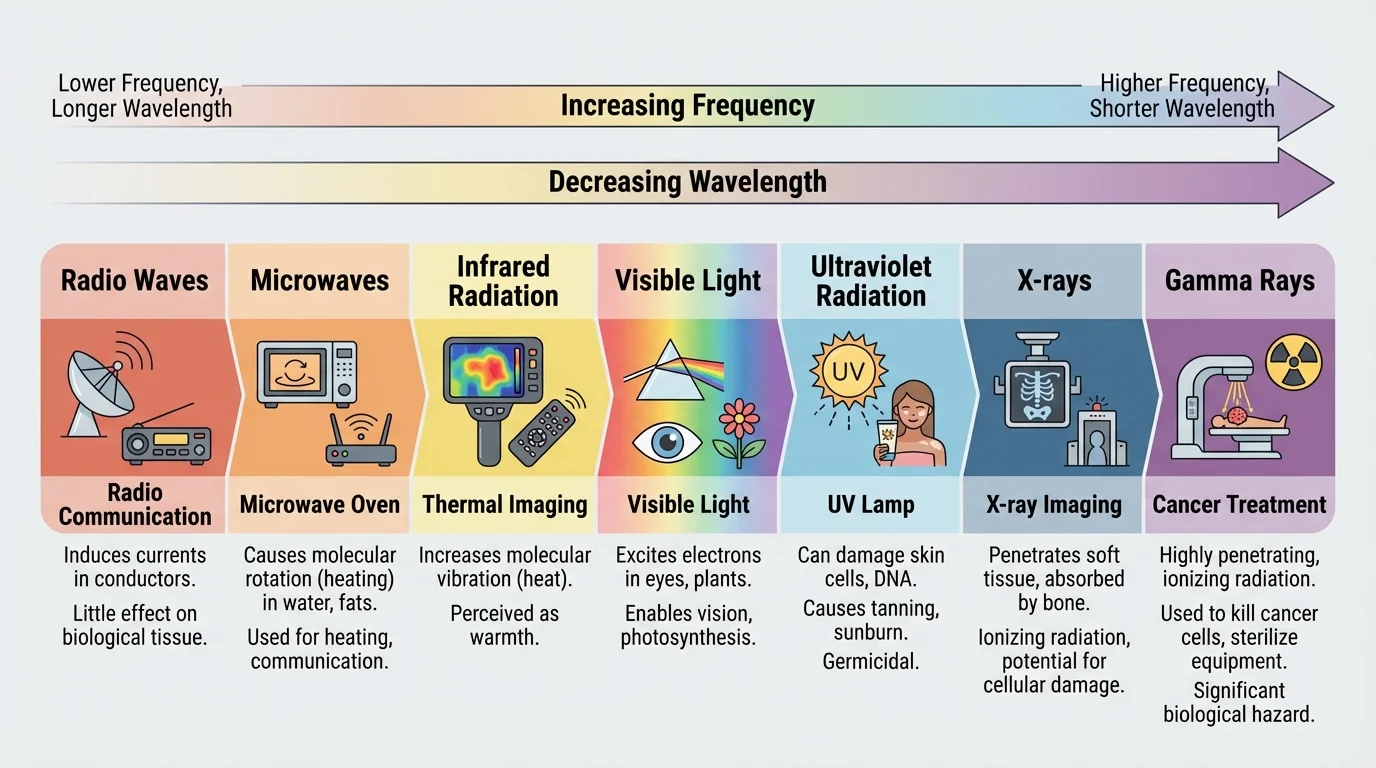
Visible light is only a small part of the spectrum. Human eyes detect it, but matter interacts with many frequencies we cannot see. Your skin absorbs infrared radiation from sunlight and feels warm. A TV remote sends infrared signals. Wi-Fi uses microwaves. Medical X-ray machines use much higher-frequency radiation to pass through soft tissue while being absorbed more strongly by bone.
Frequency is how many wave cycles pass a point each second. Wavelength is the distance between repeating parts of a wave. Photon is a packet of electromagnetic energy. Absorption happens when matter takes in the energy of radiation instead of reflecting or transmitting it.
The distinction between wave and photon descriptions is not a contradiction. The wave model helps explain spreading, interference, and communication technologies. The photon model helps explain why ultraviolet light can cause chemical changes and why X-rays can ionize atoms. As we saw in [Figure 1], the position of radiation on the spectrum gives an important first clue about its likely effects.
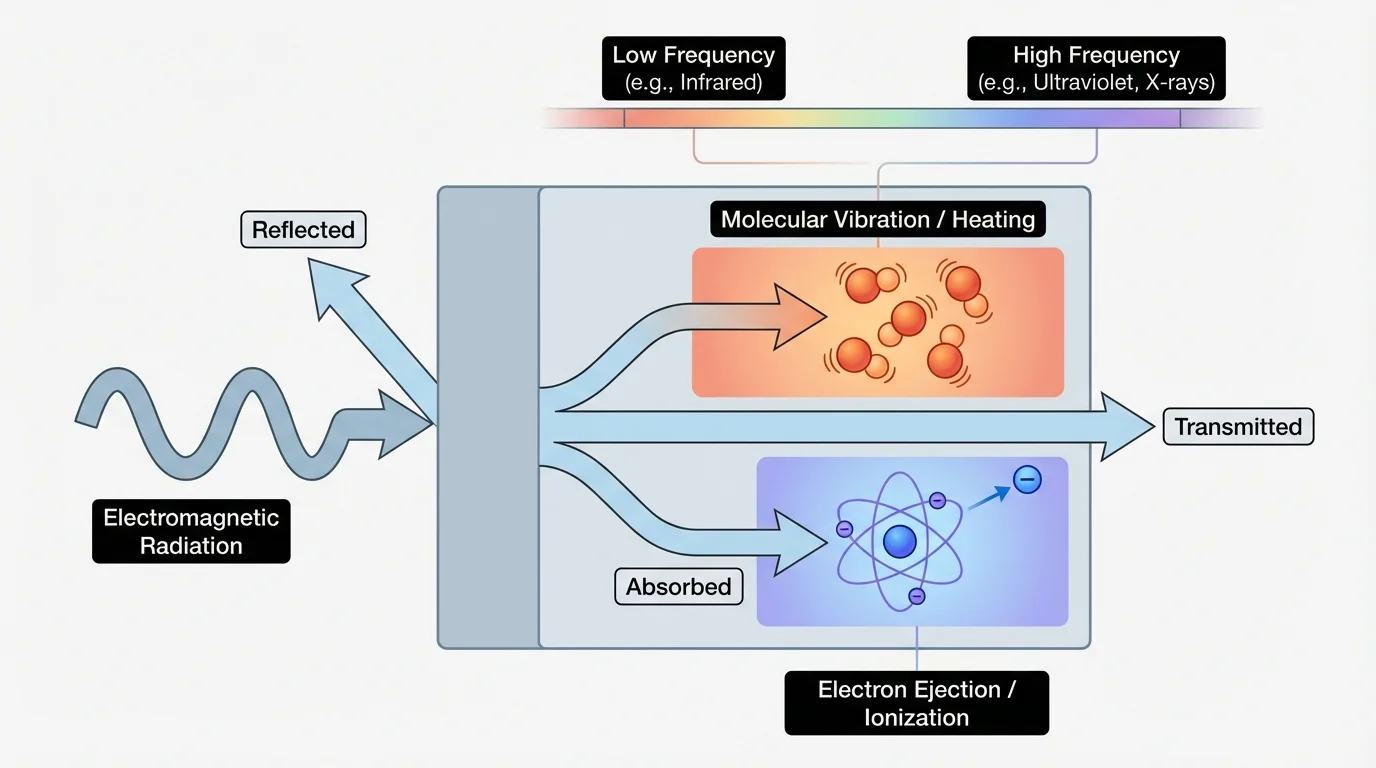
When electromagnetic radiation reaches matter, several things can happen: it can be reflected, transmitted, or absorbed. Which outcome happens depends on the frequency of the radiation and the structure of the material. A black shirt absorbs more visible light than a white shirt, which is why it gets hotter in sunlight.
[Figure 2] Absorption means energy is transferred from the radiation to the material. That energy can have different effects. In some cases, molecules begin to move or rotate more, leading to thermal effects, or heating. In other cases, electrons in atoms or molecules gain energy and change arrangement, which can lead to chemical reactions. At even higher energies, radiation may remove electrons from atoms or molecules, creating ions.
This is why the same word "radiation" can refer to very different situations. Microwaves in an oven primarily cause certain molecules, especially polar molecules in water-containing materials, to rotate in ways that increase thermal motion. Ultraviolet radiation can trigger chemical changes in skin cells. X-rays and gamma rays can be ionizing radiation, meaning they can knock electrons off atoms and molecules.
It is important to keep the assessment focus qualitative. You do not need to calculate exact photon energies to reason well. Instead, think in categories: lower-frequency radiation is usually linked to communication and heating, while higher-frequency radiation can produce chemical changes or ionization because each photon carries more energy.
Why frequency matters
The effect of absorbed radiation depends not only on the total energy involved but also on how that energy is packaged. Many low-energy photons can heat a substance, but a single high-energy photon may trigger a chemical change or ionize an atom. That is why frequency is central when evaluating claims about biological or material effects.
The material matters too. Glass transmits visible light well but blocks much ultraviolet radiation. Metal reflects many radio and microwave frequencies. Skin absorbs some ultraviolet, which is why UV exposure can lead to sunburn. Bone absorbs X-rays more strongly than surrounding soft tissue, creating contrast in medical images.
Different parts of the spectrum are associated with different common effects when absorbed by matter. These are trends, not absolute rules, but they are scientifically useful.
| Region of spectrum | Typical interaction when absorbed | Common examples | Important caution |
|---|---|---|---|
| Radio | Often used to transfer information; usually low-energy interactions | Broadcasting, communication | Claims of dramatic cell damage need strong evidence |
| Microwave | Often causes heating in suitable materials | Microwave ovens, Wi-Fi, radar | Heating depends on power, time, and material |
| Infrared | Heating and thermal emission | Heat lamps, remote controls, thermal cameras | Can cause burns at high intensity |
| Visible | Excites electrons involved in vision and color | Sight, lasers, screens | Very intense visible light can damage eyes |
| Ultraviolet | Can cause chemical changes in molecules | Tanning, sterilization, fluorescence | Overexposure can damage skin and DNA |
| X-ray | Can ionize atoms and pass through soft tissue | Medical imaging, security scanning | Unnecessary exposure should be avoided |
| Gamma rays | Strongly ionizing interactions | Cancer treatment, nuclear processes | Requires strict shielding and control |
A common misunderstanding is that "non-ionizing" means "harmless in every circumstance." That is not true. Infrared radiation can burn skin. Intense visible lasers can injure eyes. Microwaves can heat tissue if exposure is strong enough. On the other hand, "ionizing" does not mean "never useful." X-rays save lives in diagnosis, and gamma rays can be used carefully in cancer treatment.
Scientific evaluation depends on both type of possible effect and actual exposure conditions. A weak radio signal from a distant source is not equivalent to prolonged exposure to intense ultraviolet light. Frequency helps define the kind of effect that is plausible, while intensity, duration, and shielding affect the level of risk.
Sunlight includes visible light, infrared, and ultraviolet radiation. You feel the warmth mostly because of infrared and other absorbed energy, but sunburn is mainly linked to ultraviolet radiation, especially because it can damage molecules in skin cells.
This is one reason why a claim should never be accepted just because it contains a scientific word. A statement like "all radiation is dangerous" is too broad to be valid. It ignores the major physical differences across the spectrum.
To evaluate whether a claim is valid, follow a sequence of questions rather than trusting the headline. A valid claim is one that matches evidence and uses sound scientific reasoning. It should fit what is known about electromagnetic radiation and matter.
[Figure 3] First, identify the exact claim. Is it claiming heating, DNA damage, ionization, cancer risk, material weakening, or something else? Vague statements are difficult to test. "Phone radiation is bad" is too vague. "High levels of UV exposure increase the risk of skin damage" is much more precise and easier to evaluate.
Second, ask whether the claim has a plausible physical mechanism. Does the stated frequency have the kind of energy needed for the claimed effect? If someone claims that ordinary visible light from a lamp causes ionization in the way X-rays do, that should immediately raise doubt because it conflicts with the known qualitative differences across the spectrum. This does not prove the claim false by itself, but it means the evidence would need to be extraordinarily strong.
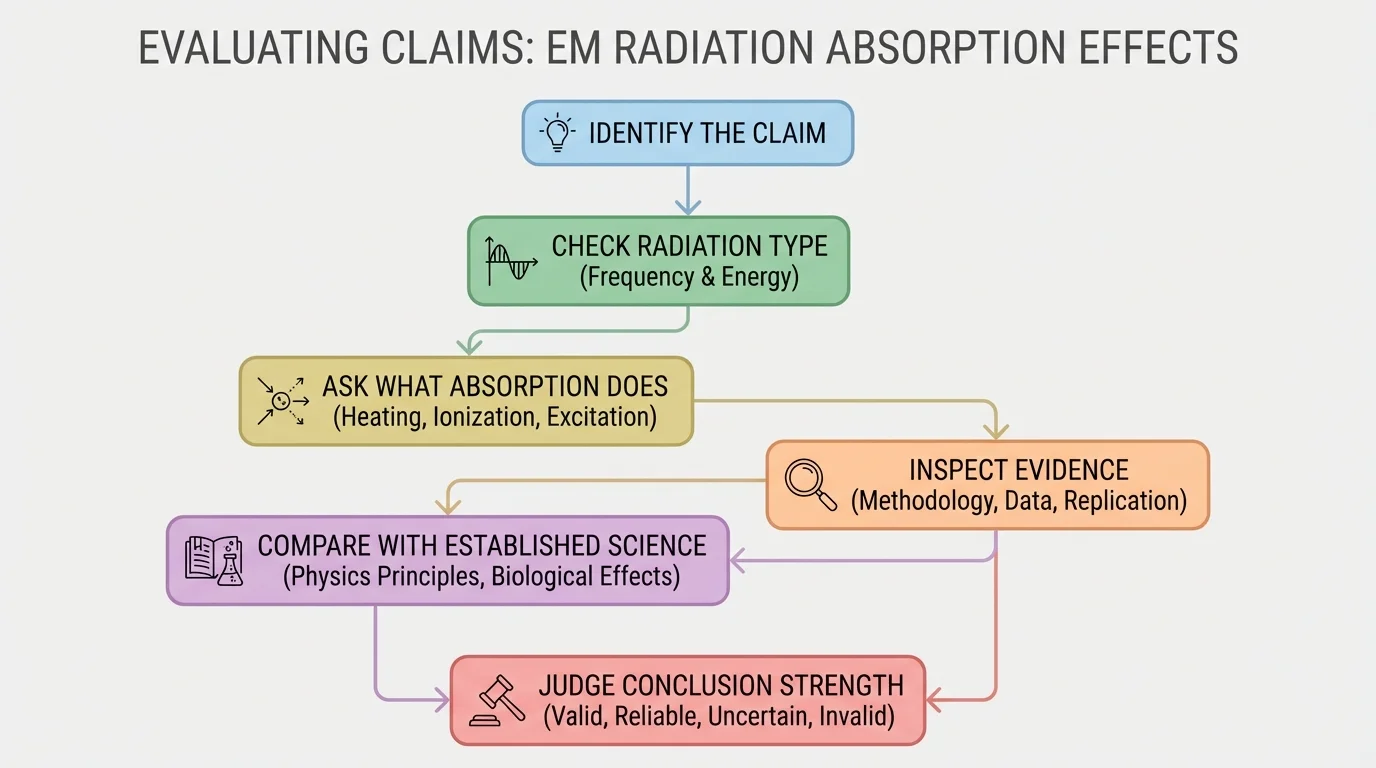
Third, check the evidence itself. Was there a direct experiment? Were there comparison groups? Were conditions controlled? Were the researchers measuring actual absorption and effects, or only guessing? Stronger claims require stronger evidence.
Fourth, examine the language. Scientists often use careful wording such as "may," "is associated with," or "under these conditions." Weak articles often turn these into "proves" or "causes" without justification. If the conclusion goes beyond the data, validity is weakened.
Case check: a weak claim
An advertisement says that a sticker on your phone "blocks all harmful radiation" and "protects your DNA."
Step 1: Identify the claim precisely.
The ad claims that ordinary phone radiation harms DNA and that a sticker prevents this.
Step 2: Check mechanism.
Phone communication uses radio or microwave frequencies, which are non-ionizing under ordinary conditions. A DNA-damage claim needs very strong evidence and a clear mechanism.
Step 3: Check evidence.
If the ad provides no peer-reviewed studies, no test methods, and no independent verification, the claim is weak.
Step 4: Judge validity.
The claim is not well supported and likely exaggerates risk while selling a product.
Validity is about whether the claim itself is scientifically justified. As shown earlier in [Figure 2], absorption can produce different outcomes, but not every outcome is plausible at every frequency.
Reliability is different from validity. A claim might sound plausible, but if it comes from an unreliable source or a poorly designed study, confidence should remain low. Reliability asks whether the evidence and source can be trusted consistently.
Reliable published materials usually have several features. They identify the author or organization. They explain where the information came from. They describe methods clearly enough for others to examine. They are often reviewed by experts. Their results are consistent with other evidence, or, if they differ, they explain why.
Unreliable sources often rely on dramatic wording, anonymous testimony, or cherry-picked data. They may use one unusual study while ignoring many larger studies. They may confuse a laboratory result with a real-world exposure situation. They may also have a financial motive, such as selling a shield, supplement, or device.
Peer review is a process in which other experts examine a study before publication. Replication means other researchers can repeat a study and get similar results. Bias is a tendency that can unfairly influence results or interpretation.
For example, if one small study reports an effect of radio waves on cells in unusual conditions, but many other studies under realistic conditions do not find the same effect, the single study should be treated cautiously. Reliability increases when different researchers using different methods reach similar conclusions.
Another key issue is whether exposure was measured realistically. A claim about harm from daily technology should use conditions that match actual use. If a study blasts cells with unusually strong radiation for long periods, it may not represent normal life. Reliable interpretation requires matching the experiment to the real-world question.
[Figure 4] Comparing several claims side by side helps reveal which ones are stronger. The same scientific questions apply each time: What frequency is involved? What kind of effect is physically plausible? What evidence supports the claim? How trustworthy is the source?
Claim 1: "Ultraviolet tanning beds are a safe way to get a healthy glow." This is a weak claim. Ultraviolet radiation can cause chemical changes in skin and damage DNA. There is a clear mechanism, strong evidence of risk from overexposure, and no good reason to describe tanning beds as generally "safe" without major qualifications.
Claim 2: "Medical X-rays are always too dangerous to use." This is also weak. X-rays are ionizing, so risk is real, but the claim ignores benefit, dose control, shielding, and medical judgment. A more valid claim would say that X-rays should be used carefully and only when the diagnostic benefit outweighs the risk.
Claim 3: "Microwave ovens make food radioactive." This is false. Microwave radiation heats food by transferring energy, but it does not make the food a radioactive source. The mechanism described by the claim does not match known physics.

Claim 4: "Cell phones emit radiation, so they must cause ionization." This is invalid reasoning. The word "radiation" alone does not determine the effect. Frequency matters. Radio and microwave frequencies do not behave qualitatively the same way as X-rays and gamma rays.
Claim 5: "Sunscreen helps reduce harm from ultraviolet exposure." This is a strong claim when supported by appropriate evidence. It matches a known mechanism: ultraviolet radiation can damage skin, and sunscreen is designed to reduce how much reaches or affects the skin.
Case check: a stronger claim
A health article states that repeated intense ultraviolet exposure raises the risk of skin damage and that protective clothing and sunscreen can reduce that risk.
Step 1: Check mechanism.
Ultraviolet radiation can trigger chemical changes in skin molecules and damage DNA.
Step 2: Check evidence.
This topic has been studied extensively, and the claim matches broad scientific understanding.
Step 3: Check wording.
The article uses cautious language about risk and prevention instead of sensational language.
Step 4: Judge reliability and validity.
The claim is both scientifically valid and likely reliable if it comes from a trusted medical or scientific source.
The comparison in [Figure 4] makes an important pattern visible: the strongest claims are specific, mechanism-based, and supported by multiple lines of evidence.
Many misleading claims become persuasive because readers miss small but important differences in wording. One of the biggest is the difference between hazard and risk. A hazard is something that can cause harm under some conditions. Risk depends on the actual chance of harm in a real situation. X-rays are a hazard, but the risk from one medically necessary image is not the same as the risk from repeated unnecessary exposure.
Another critical distinction is between correlation and causation. If two things appear together, that does not prove one causes the other. Suppose a report finds that people who use a certain technology also report headaches more often. That does not automatically prove the radiation caused the headaches. Other variables might be involved, such as sleep, stress, or screen time.
Words like "proves," "miracle," "toxic," or "completely safe" should make you cautious. Real science is usually more careful. It recognizes uncertainty, conditions, and limits. This does not make science weak; it makes science honest.
"The first principle is that you must not fool yourself—and you are the easiest person to fool."
— Richard Feynman
You should also notice whether a source distinguishes between exposure and absorption. Being near a radiation source is not the same as absorbing a harmful amount of energy. Materials, distance, shielding, orientation, and time all matter.
These ideas are not just for science class. They help with real decisions. When evaluating a product claim, ask whether the seller explains the frequency involved and the mechanism of protection. When reading about health risks, check whether the source is a scientific organization, a medical institution, or a sensational website. When hearing that a technology is "dangerous because radiation," ask: what kind of radiation, what kind of absorption, and what evidence?
In medicine, the goal is not to fear all radiation but to use it wisely. In engineering and communications, radio and microwaves are invaluable for transferring information. In environmental science, ultraviolet monitoring matters because UV exposure affects ecosystems and human health. In each case, understanding the physics helps you evaluate the claim more fairly.
One of the strongest habits in science is proportional judgment. Extraordinary claims need extraordinary evidence. Claims that match known mechanisms and are supported by repeated studies deserve more confidence than dramatic headlines or advertisements. The more a source tries to trigger fear without explaining frequency, absorption, and evidence, the less trust it should receive.