Every breath you take contains carbon atoms that may once have been part of a tree, a dinosaur, a shell on the ocean floor, or even volcanic gas. That is not science fiction. Carbon moves continuously through Earth's systems, changing form and location but never disappearing. To understand life on Earth, climate change, food webs, and even fossil fuels, you need to understand how carbon cycles.
Carbon is especially important because it can form many stable bonds with other elements. That makes it a central ingredient in the molecules of living things, including carbohydrates, fats, proteins, and nucleic acids. Carbon is also found in the air as carbon dioxide \((\mathrm{CO}_2)\), dissolved in water, and locked in rocks and sediments.
In ecosystems, matter is conserved. That means carbon atoms are not created by organisms and they are not destroyed when organisms die. Instead, the atoms are rearranged and transferred. A carbon atom might move from the atmosphere into a leaf, from the leaf into a deer, from the deer into decomposers, and eventually back into the air, water, or soil.
Conservation of matter is the idea that matter is not lost as it moves through a system. In biology, this means atoms such as carbon, oxygen, nitrogen, and hydrogen are continually recycled.
This is why the carbon cycle is not just a biology topic. It also connects to chemistry, Earth science, and environmental science. Living organisms interact with air, water, and rock, so the movement of carbon links the biotic and abiotic parts of Earth.
Scientists often describe carbon movement by organizing Earth into major systems, as [Figure 1] shows by using reservoirs and arrows between them. A reservoir is a place where carbon is stored, and a flux is the movement of carbon from one reservoir to another.
The biosphere includes all living things and recently living organic matter. Forests, grasslands, plankton, animals, fungi, and soil organisms are all part of it. The atmosphere contains gases in the air, including \((\mathrm{CO}_2)\). The hydrosphere includes oceans, lakes, rivers, groundwater, and dissolved substances in water. The geosphere includes rocks, sediments, fossil fuels, and Earth's crust.
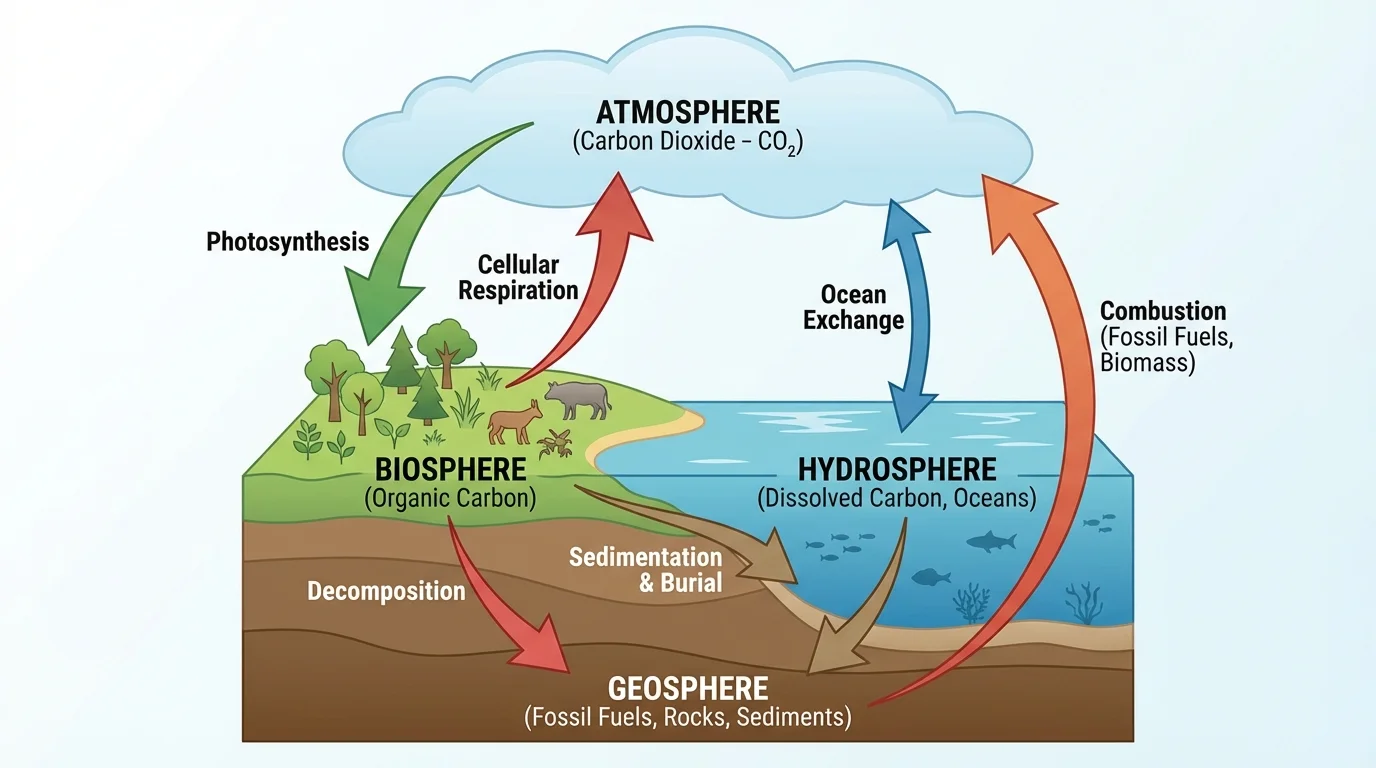
Carbon can move rapidly or very slowly between these systems. A plant may absorb atmospheric \((\mathrm{CO}_2)\) in minutes or hours. By contrast, carbon buried in sedimentary rock may remain stored for millions of years. This difference in timescale matters because short-term and long-term carbon storage affect climate and ecosystems differently.
When you model the carbon cycle, it helps to think in terms of boxes and arrows. The boxes are reservoirs, and the arrows are processes such as photosynthesis, respiration, decomposition, diffusion between air and water, burial, weathering, and combustion. Some arrows are large and fast, while others are small and slow.
Carbon cycle is the continuous movement of carbon among Earth's systems. Reservoir means a storage location for carbon, and flux means the rate or pathway by which carbon moves from one reservoir to another.
Because carbon exists in many forms, a model must track both where the carbon is and what form it takes. It may be part of \((\mathrm{CO}_2)\) in air, dissolved carbon compounds in water, organic molecules in organisms, or carbonate minerals in rock.
[Figure 2] Photosynthesis is one of the most important carbon-moving processes on Earth. Plants, algae, and some bacteria use light energy to build sugar molecules from carbon dioxide and water.
A simplified way to represent photosynthesis is:
\[6\textrm{CO}_2 + 6\textrm{H}_2\textrm{O} \rightarrow \textrm{C}_6\textrm{H}_{12}\textrm{O}_6 + 6\textrm{O}_2\]
This equation shows the overall inputs and outputs, not the specific chemical steps. That is enough for understanding the carbon cycle. The key idea is that carbon from \((\mathrm{CO}_2)\) becomes part of glucose and other organic molecules. Those molecules can then become wood, roots, fruits, leaves, or stored compounds in a plant's body.
For example, when a young tree grows from a sapling into a large oak, most of the added mass does not come from soil. A major source of that mass is carbon taken from atmospheric \((\mathrm{CO}_2)\) during photosynthesis. The tree is literally building itself from carbon atoms that were once in the air.
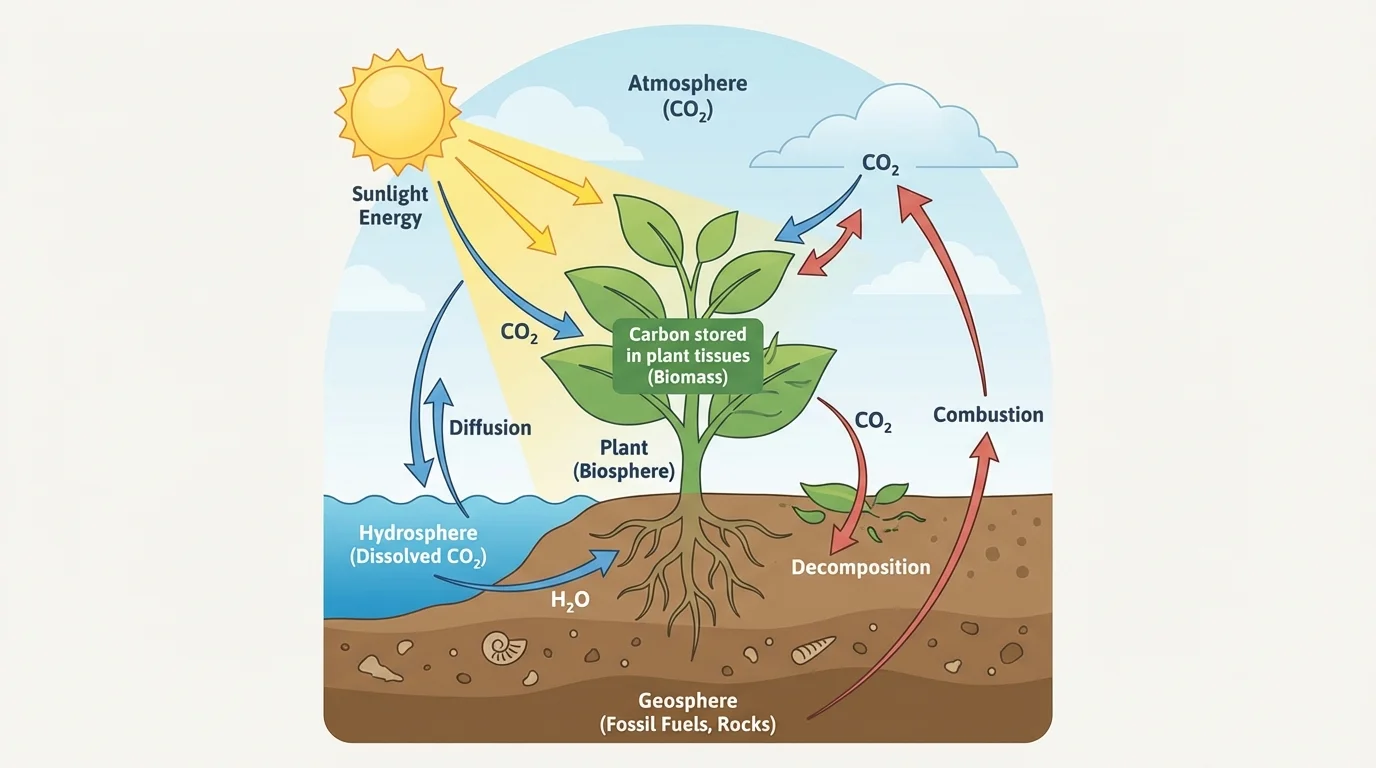
That carbon then enters food webs. If a caterpillar eats the leaves, some of the carbon becomes part of the caterpillar's body. If a bird eats the caterpillar, the carbon moves again. Photosynthesis therefore serves as a major entry point for carbon into the biosphere.
Numeric example: carbon gain in a growing plant
Suppose a plant absorbs \((120\ \mathrm{g})\) of carbon in one week through photosynthesis. During the same week, the plant releases \((80\ \mathrm{g})\) of carbon through respiration.
Step 1: Identify the two opposite carbon flows.
Carbon absorbed by photosynthesis is \((120\ \mathrm{g})\). Carbon released by respiration is \((80\ \mathrm{g})\).
Step 2: Find the net carbon stored.
\((120 - 80 = 40)\), so the plant stores \((40\ \mathrm{g})\) of carbon.
The plant's biomass increases because photosynthesis exceeds respiration during that time.
Photosynthesis happens in terrestrial ecosystems and aquatic ecosystems. On land, forests, grasslands, and crops remove large amounts of \((\mathrm{CO}_2)\) from the atmosphere. In water, phytoplankton and algae carry out photosynthesis and play a major role in global carbon cycling.
[Figure 3] Cellular respiration transfers carbon in the opposite general direction from photosynthesis. Organisms break down glucose and other organic molecules to release usable energy for life processes, and carbon is released as \((\mathrm{CO}_2)\).
A simplified overall equation for aerobic cellular respiration is:
\[\textrm{C}_6\textrm{H}_{12}\textrm{O}_6 + 6\textrm{O}_2 \rightarrow 6\textrm{CO}_2 + 6\textrm{H}_2\textrm{O}\]
Again, the important idea here is not the individual chemical steps. The key point is that carbon stored in food molecules returns to the atmosphere or hydrosphere as \((\mathrm{CO}_2)\). This process occurs in plants, animals, fungi, protists, and many bacteria. Plants do not only perform photosynthesis; they also respire.
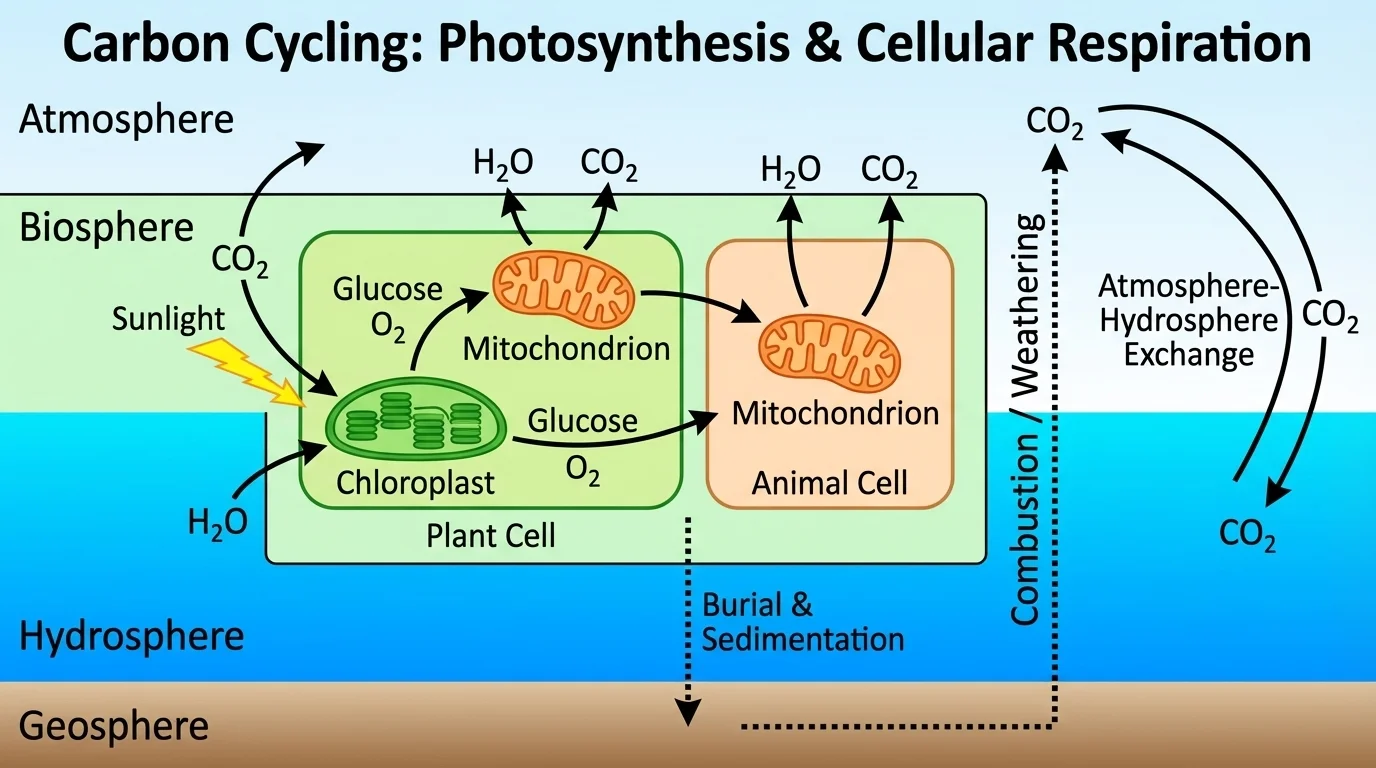
Think about an athlete running hard during a race. Their muscles need more energy, so cellular respiration increases. The runner breathes faster, taking in more oxygen and releasing more carbon dioxide. That exhaled \((\mathrm{CO}_2)\) is carbon returning from the biosphere to the atmosphere.
Respiration also happens in aquatic systems. Fish, aquatic plants, and microorganisms release \((\mathrm{CO}_2)\) into water. Some of that dissolved carbon may later diffuse into the atmosphere, while some stays in the hydrosphere.
Photosynthesis and respiration are linked but not identical opposites. Photosynthesis stores carbon in organic molecules and usually requires light. Cellular respiration releases carbon from organic molecules and occurs continuously in living cells. Together, these processes cycle carbon through ecosystems while transferring energy through living systems.
The balance between photosynthesis and respiration helps determine whether an ecosystem stores carbon or releases it. A fast-growing forest may store more carbon than it releases. A decaying wetland or a burned forest may release more carbon than it stores over a given period.
A scientific model is a simplified representation of a real system. In carbon cycling, a model should show major reservoirs, pathways of movement, and the idea that matter is conserved, as [Figure 4] shows in a systems flowchart. A model does not need every detail to be useful, but it must include the most important relationships.
A strong carbon cycle model for this topic should include at least these reservoirs: atmosphere, producers, consumers, decomposers, water, soil, and geologic storage. It should also include key processes: photosynthesis, feeding, respiration, decomposition, combustion, ocean-atmosphere exchange, and burial into sediments or fossil fuels.
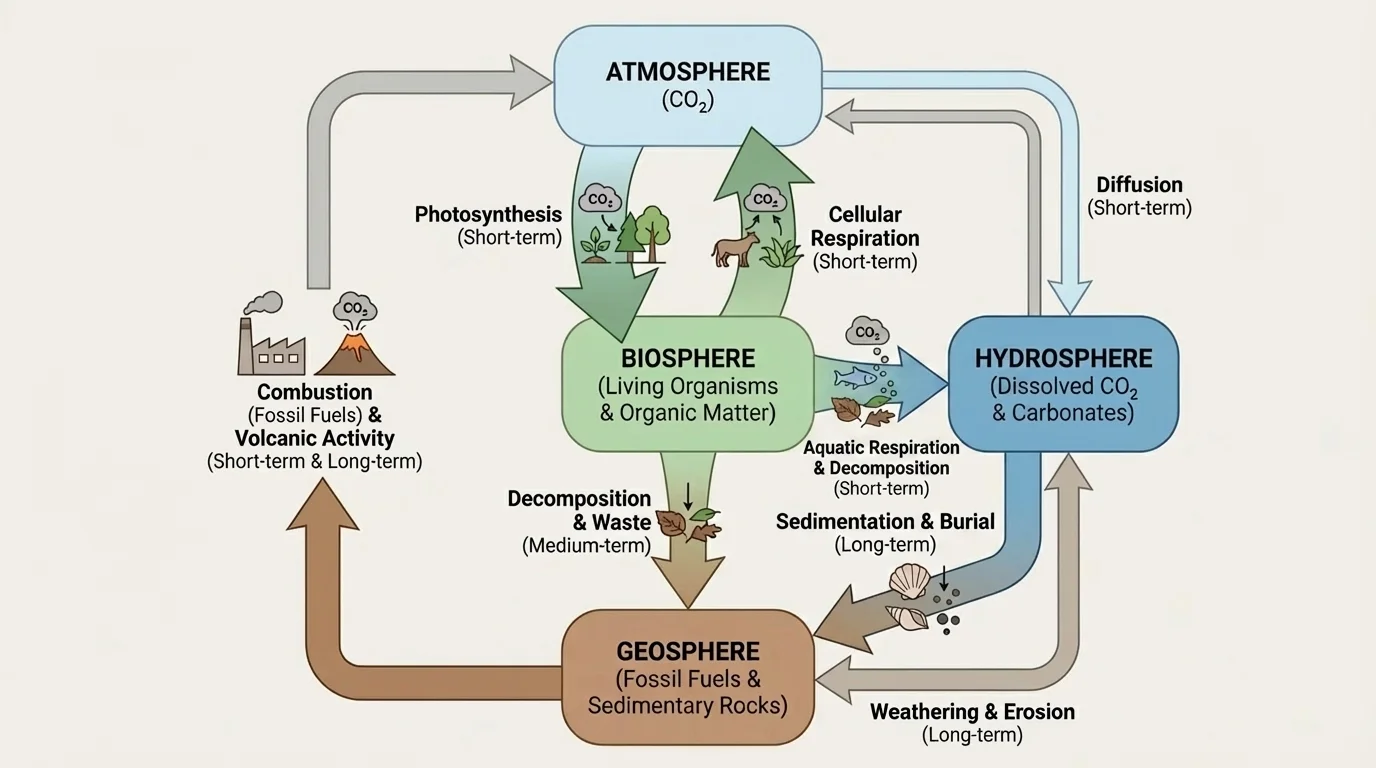
One useful way to build the model is to start with the atmosphere. Draw an arrow from atmospheric \((\mathrm{CO}_2)\) to producers and label it photosynthesis. Then draw arrows from producers to consumers for feeding, from organisms to decomposers for death and waste, and from producers, consumers, and decomposers back to the atmosphere or water for respiration.
Next, add longer-term pathways. Some dead organic matter becomes buried in sediments. Over long periods, this can contribute to fossil fuel formation or carbon-rich rock. Combustion of fossil fuels returns that stored carbon to the atmosphere much faster than it formed. This is one reason human activity changes the carbon cycle so strongly.
Models can also show relative speed. You might use thicker arrows for large or rapid fluxes and thinner arrows for smaller or slower ones. For example, daily plant respiration is a fast flux, while carbon burial into rock is a slow flux.
Example: checking conservation of carbon in a simple model
Suppose a small pond model tracks carbon over one month. The algae remove \((300\ \mathrm{g})\) of carbon from dissolved \((\mathrm{CO}_2)\) by photosynthesis. During the same month, algae, fish, and decomposers release \((210\ \mathrm{g})\) of carbon by respiration, and \((60\ \mathrm{g})\) becomes buried in sediment.
Step 1: Add the carbon leaving the living biomass.
\((210 + 60 = 270)\), so \((270\ \mathrm{g})\) of carbon leaves the active living reservoir.
Step 2: Compare with the carbon entering by photosynthesis.
\((300 - 270 = 30)\), so \((30\ \mathrm{g})\) remains as additional carbon stored in living organisms.
The model is consistent with conservation of matter because all \((300\ \mathrm{g})\) taken in by photosynthesis is accounted for.
Later, when you evaluate ecosystem data, a model like the one in [Figure 4] helps you decide where carbon is entering, where it is leaving, and where it is accumulating.
Not all carbon moves directly from plants to animals and back into the air. Many pathways branch outward. Decomposers such as bacteria and fungi break down dead organisms and wastes. In doing so, they return carbon to soils, water, and the atmosphere.
Soils are major carbon reservoirs. Fallen leaves, roots, dead microorganisms, and waste materials add carbon to soil. Some soil carbon is quickly respired back as \((\mathrm{CO}_2)\), while some remains for years or centuries. This means that changes in farming, deforestation, or land use can alter whether soil acts as a carbon sink or a carbon source.
The oceans are also crucial. Carbon dioxide can dissolve into seawater, and dissolved carbon can move back into the air. Marine organisms use carbon to build bodies and shells. When some of these organisms die, carbon may sink into deeper water or sediments. Over geologic time, carbon can become part of limestone and other rocks in the geosphere.
Ocean water contains far more carbon than the atmosphere. That makes the hydrosphere a powerful part of the global carbon cycle, but changes in temperature and chemistry affect how much carbon the oceans can absorb.
[Figure 5] Some carbon remains stored for extremely long periods in fossil fuels such as coal, oil, and natural gas. These formed from ancient organic matter under heat and pressure over millions of years. When humans burn them in cars, power plants, and factories, carbon that was once locked in the geosphere rapidly returns to the atmosphere as \((\mathrm{CO}_2)\).
Human activities alter carbon movement among reservoirs. Two especially important changes are fossil fuel combustion and deforestation. Both reduce long-term carbon storage or increase atmospheric carbon release.
Deforestation matters because fewer trees mean less photosynthesis removing \((\mathrm{CO}_2)\) from the atmosphere. If forests are cleared and burned, carbon stored in biomass is released quickly. If forests regrow, they can again act as carbon sinks by taking up carbon during photosynthesis.
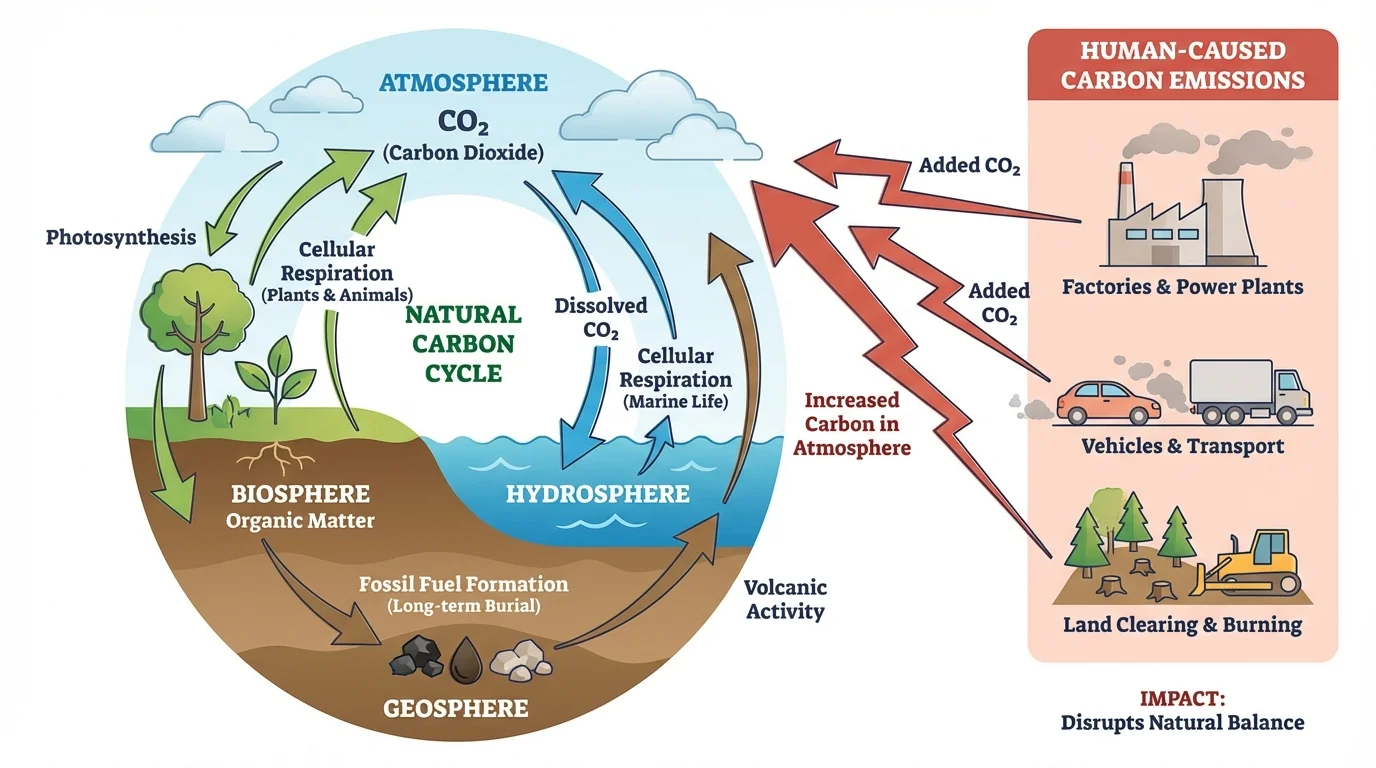
Burning fossil fuels adds carbon that had been stored underground for millions of years. In a natural short-term cycle, photosynthesis and respiration often exchange carbon repeatedly among air, water, and organisms. Fossil fuel use adds extra carbon from long-term geologic storage into that faster cycle.
Scientists monitor carbon cycling using satellites, ocean sensors, atmospheric sampling stations, and ecosystem studies. For example, measurements of atmospheric \((\mathrm{CO}_2)\) over time help reveal seasonal patterns. In the Northern Hemisphere, carbon dioxide often drops during the growing season because plants remove more \((\mathrm{CO}_2)\) by photosynthesis.
Example: interpreting seasonal carbon data
A monitoring station records atmospheric \((\mathrm{CO}_2)\) at \((420\ \mathrm{ppm})\) in early spring and \((412\ \mathrm{ppm})\) in midsummer.
Step 1: Calculate the change.
\((420 - 412 = 8)\), so the concentration decreases by \((8\ \mathrm{ppm})\).
Step 2: Connect the change to carbon cycling.
A summer decrease suggests that photosynthesis by plants is removing more \((\mathrm{CO}_2)\) from the atmosphere than respiration and decomposition are adding during that period.
This kind of pattern provides evidence for large-scale carbon movement between the atmosphere and biosphere.
Agriculture and ecosystem restoration also depend on understanding carbon cycling. Farmers may use practices that increase soil carbon storage. Conservation scientists may restore wetlands, grasslands, or forests to improve carbon sequestration while supporting biodiversity.
A model is useful only if it matches evidence. If a model shows photosynthesis adding carbon to plants, but does not show respiration, decomposition, or carbon returning to air and water, it is incomplete. Good models include both uptake and release pathways.
When you evaluate a carbon model, ask several questions. Are all four Earth systems represented? Does the model show both short-term and long-term storage? Are the arrows labeled with actual processes rather than vague phrases? Does the model make it possible to trace the path of a single carbon atom through multiple reservoirs?
It is also important to recognize scale. A model of a forest may emphasize trees, animals, and soil. A global model must include oceans, sediments, fossil fuels, and atmospheric circulation. Both can be accurate, but each is designed for a different question.
The earlier Earth-system view in [Figure 1] and the systems flow approach in [Figure 4] remain useful here because they emphasize that carbon cycling is not a single loop. It is a network of pathways operating at different speeds across connected systems.
The central scientific idea is straightforward but powerful: photosynthesis moves carbon into living matter, cellular respiration moves carbon back out, and together with decomposition, ocean exchange, combustion, and geologic processes, they cycle carbon through the biosphere, atmosphere, hydrosphere, and geosphere. Understanding that model helps explain ecosystem function, climate patterns, and the impact of human actions on Earth.