A car engine, a rusting bridge, a glowing hand warmer, and the digestion of your lunch seem like completely different events. Yet at the molecular level, all of them follow the same logic: particles collide, atoms rearrange, bonds break, new bonds form, and energy shifts from one form to another. Chemistry becomes much more powerful when you stop seeing reactions as mysterious changes and start seeing them as billions of tiny collisions with consequences.
A chemical process is any change in which atoms are rearranged to form new substances. In a chemical reaction, the atoms in the starting substances do not disappear, and no new kind of atom is created. Instead, the same atoms are connected in new ways. For example, when hydrogen reacts with oxygen, the atoms in \(\textrm{H}_2\) and \(\textrm{O}_2\) rearrange to make water:
\[2\textrm{H}_2 + \textrm{O}_2 \rightarrow 2\textrm{H}_2\textrm{O}\]
The key idea is that chemical change is really about rearranging connections between atoms. The identity of the atoms stays the same, but the substances change because the atoms are bonded differently. This is why chemistry explains both visible changes, such as burning paper, and invisible ones, such as the reactions inside your cells.
Matter is made of atoms, and atoms can join to form molecules or extended structures. In a physical change, the identity of the substance stays the same. In a chemical change, atoms are reorganized into new substances with different properties.
Many reactions happen all around you without being obvious. Iron slowly reacts with oxygen and water to form rust. Fuel reacts with oxygen in an engine in a tiny fraction of a second. In your body, glucose reacts through many steps to release usable energy. The difference between these examples is not whether collisions occur; particles are always moving and colliding. The difference is whether the collisions are effective and how much energy is involved.
The collision theory of reactions says that particles must collide for a reaction to occur, and not every collision succeeds, as shown in [Figure 1]. For a collision to produce new molecules, the particles must hit with enough energy and in the right orientation. If they collide too gently, or if the reactive parts do not line up, they simply bounce apart.
This explains why reactions do not happen instantly every time molecules meet. Chemical bonds are not rearranged automatically. Existing bonds resist change, so particles must overcome an energy barrier before atoms can be reorganized. That minimum required energy is called activation energy.
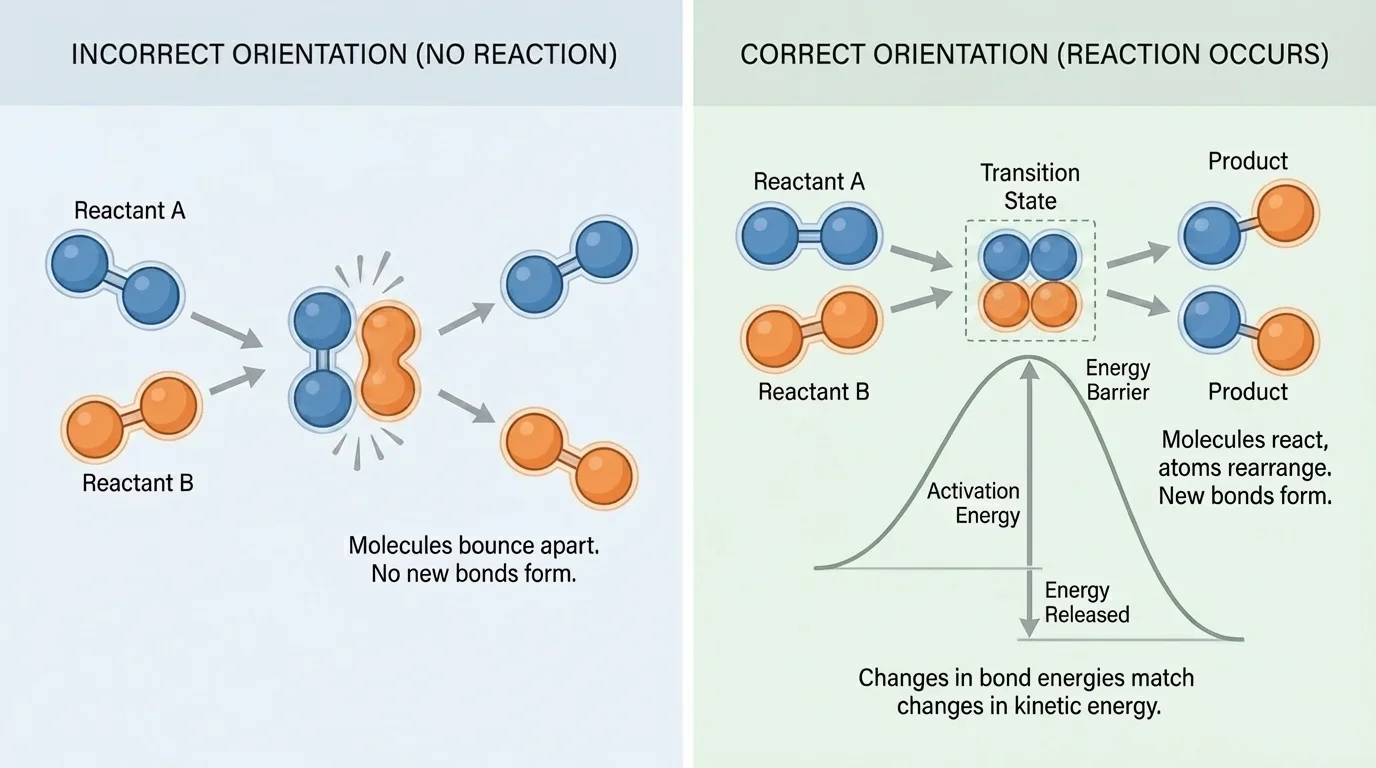
You can think of activation energy as the push needed to start a reaction. Even if a reaction would release energy overall, the particles may still need an initial input to begin. A match provides activation energy for wood to burn. A spark provides activation energy for gasoline in an engine. Once enough particles react, the energy released can help more particles overcome the barrier.
Orientation matters too. If two complex molecules collide, only certain parts may be able to interact. This is especially important in biology, where enzymes often work because they hold reacting molecules in positions that make effective collisions more likely. The molecules are still colliding, but the enzyme makes successful collisions much more common.
Activation energy is the minimum energy particles must have during a collision for a chemical reaction to begin.
Effective collision is a collision in which particles have enough energy and the correct orientation to rearrange atoms and form new substances.
The idea from [Figure 1] also helps explain why some mixtures can sit together for a long time without reacting quickly. The particles may collide often, but very few of those collisions may have both the right energy and the right geometry. Chemistry is not only about contact; it is about the quality of collisions.
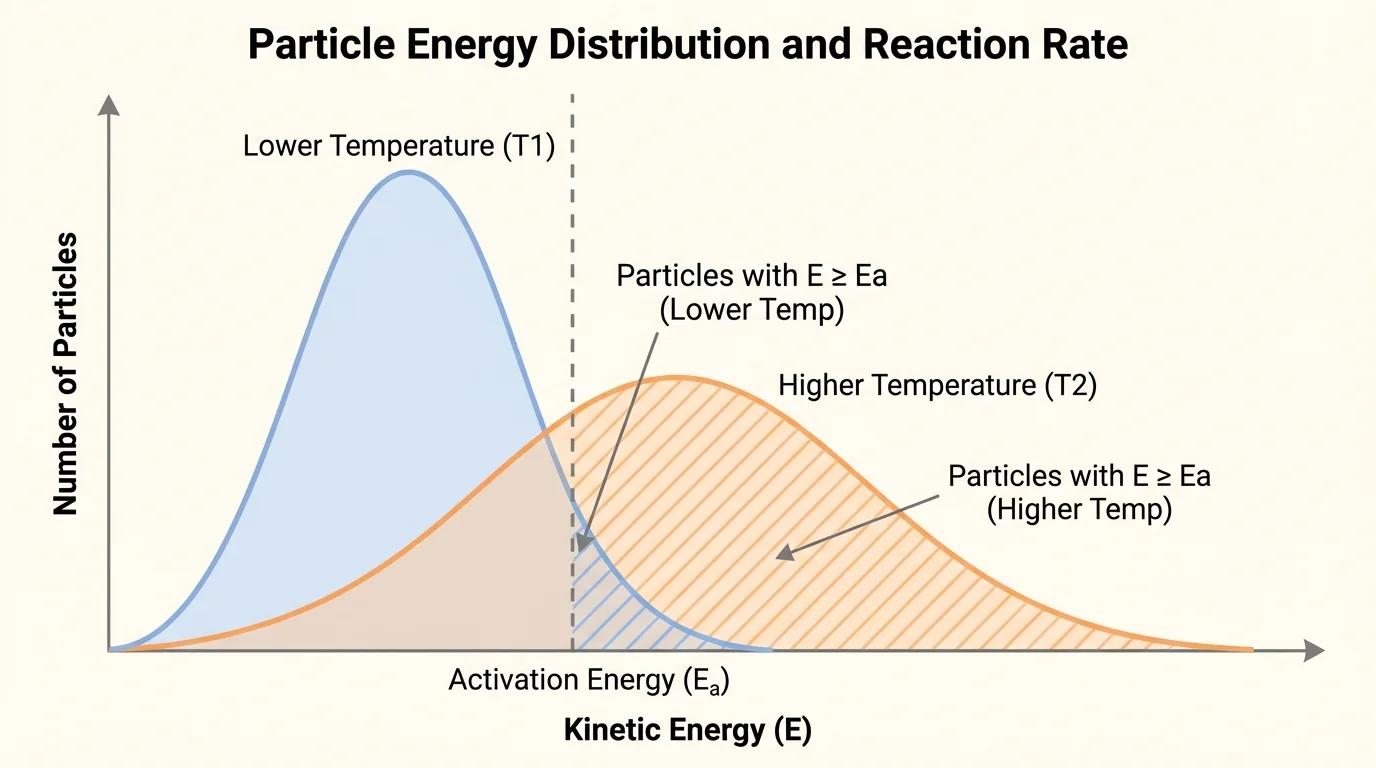
The reaction rate describes how quickly reactants are converted into products. Some reactions are extremely fast, like the explosion of hydrogen and oxygen under the right conditions. Others are slow, like the corrosion of iron. Rate depends on how often particles collide and how many of those collisions are effective, as shown in [Figure 2].
Temperature is one of the most important factors. When temperature increases, particles move faster on average. That means collisions happen more often, and a larger fraction of collisions have enough energy to exceed activation energy. This is why food spoils faster at room temperature than in a refrigerator and why many reactions speed up when heated.
Concentration also matters. If more reactant particles are packed into the same volume, collisions become more frequent. For gases, pressure plays a similar role because higher pressure squeezes particles closer together. Surface area matters for solids: a powdered solid reacts faster than a large chunk of the same mass because more particles are exposed for collisions.
Catalysts provide another major control over rate. A catalyst is a substance that increases reaction rate without being permanently used up. It works by providing a different reaction pathway with a lower activation energy. Lowering the energy barrier means more collisions are successful even at the same temperature.
This is why catalysts are so important in industry and biology. Car catalytic converters help harmful gases react into less harmful ones. Enzymes in your body catalyze reactions that would otherwise be too slow to sustain life. Even though the catalyst changes the rate, it does not change which atoms are present before and after the reaction; it changes how easily the rearrangement happens.
Why temperature changes rate so strongly
Not all particles in a sample have exactly the same energy. At a higher temperature, the whole energy distribution shifts so that more particles have energies above the activation-energy threshold. That is why even a moderate increase in temperature can make a reaction much faster.
Looking back at [Figure 2], the important idea is not just that particles move faster, but that the number of particles able to react rises sharply. This is why the effect of temperature on rate is often dramatic rather than small.
When a reaction occurs, atoms are conserved. That means the total number of each kind of atom stays the same before and after the reaction. Chemical equations show this conservation by being balanced. Consider the formation of water again:
\[2\textrm{H}_2 + \textrm{O}_2 \rightarrow 2\textrm{H}_2\textrm{O}\]
On the left side there are \(4\) hydrogen atoms and \(2\) oxygen atoms. On the right side there are also \(4\) hydrogen atoms and \(2\) oxygen atoms. Nothing has vanished. The atoms have simply been reorganized into a new arrangement.
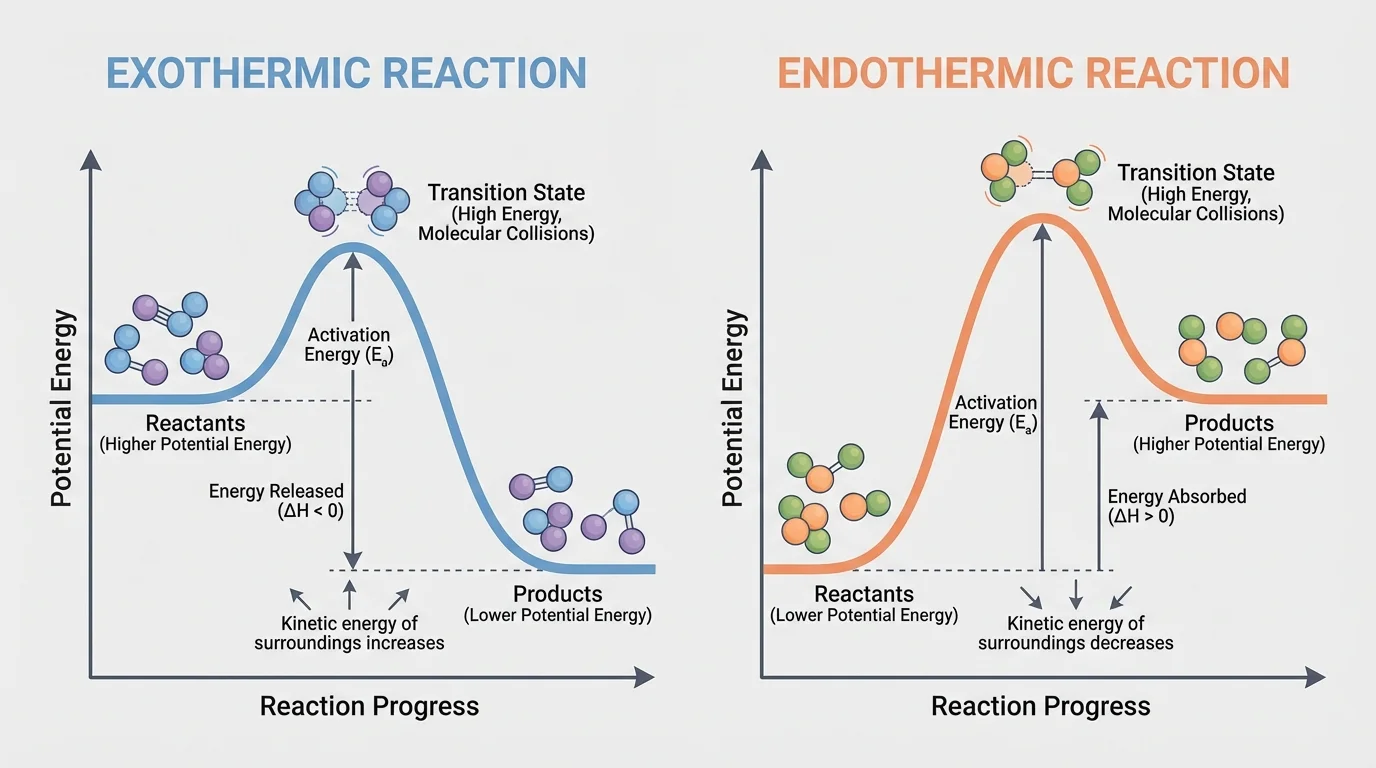
This conservation matters when thinking about energy too. Since the same atoms are present, the energy difference between reactants and products comes mainly from how those atoms are bonded, as [Figure 3] illustrates. A molecule with one pattern of bonds can store a different amount of energy than a molecule made from the same atoms bonded in another pattern.
| Reaction view | What stays the same | What changes |
|---|---|---|
| Atoms | Number and type of atoms | How atoms are connected |
| Matter | Total mass in a closed system | Substances present |
| Energy | Total energy | How energy is stored or transferred |
Table 1. Comparison of what is conserved and what changes during a chemical reaction.
The energy changes in reactions make sense when you track chemical bonds. Breaking bonds requires energy input, while forming new bonds releases energy. The total energy change depends on comparing these two amounts.
If more energy is released when new bonds form than is required to break the original bonds, the reaction releases energy overall. Such a reaction is called exothermic. If more energy is required to break bonds than is released by forming new ones, the reaction absorbs energy overall. That type is an endothermic reaction.
Combustion is a familiar exothermic process. In methane combustion, bonds in methane and oxygen are broken, and new bonds in carbon dioxide and water are formed:
\[\textrm{CH}_4 + 2\textrm{O}_2 \rightarrow \textrm{CO}_2 + 2\textrm{H}_2\textrm{O}\]
The products have a lower total chemical energy than the reactants, so energy is released to the surroundings as heat and light. That released energy can increase the kinetic energy of nearby particles, causing the temperature to rise.
Cold packs often use endothermic processes. In some instant cold packs, dissolving a salt such as ammonium nitrate in water requires more energy than is released by the interactions formed, so energy is taken in from the surroundings. The surroundings then lose thermal energy and feel colder.
Some reactions can be highly exothermic even though they still need activation energy to start. A fuel-air mixture in an engine contains stored chemical energy, but it does not react rapidly until a spark helps enough particles overcome the activation barrier.
[Figure 3] also helps distinguish between the activation energy and the overall energy change. Activation energy is the initial barrier that must be crossed. The overall energy change compares the chemical energy of reactants and products after the reaction is complete.
Chemists often estimate energy change using bond energy, which is the amount of energy needed to break a particular bond in a mole of molecules. Bond energies are average values, so they are useful for estimates rather than exact values. The basic relationship is:
\[\Delta H \approx \sum \textrm{bond energies of bonds broken} - \sum \textrm{bond energies of bonds formed}\]
If the result is negative, the reaction tends to be exothermic. If the result is positive, the reaction tends to be endothermic.
Estimating the energy change for forming hydrogen chloride
Consider the reaction
\[\textrm{H}_2 + \textrm{Cl}_2 \rightarrow 2\textrm{HCl}\]
Suppose the average bond energies are: \(\textrm{H–H} = 436 \textrm{ kJ/mol}\), \(\textrm{Cl–Cl} = 243 \textrm{ kJ/mol}\), and \(\textrm{H–Cl} = 431 \textrm{ kJ/mol}\).
Step 1: Add the energy needed to break the reactant bonds.
One \(\textrm{H–H}\) bond and one \(\textrm{Cl–Cl}\) bond are broken, so the total energy absorbed is \(436 + 243 = 679 \textrm{ kJ/mol}\).
Step 2: Add the energy released when product bonds form.
Two \(\textrm{H–Cl}\) bonds are formed, so the total energy released is \(2 \times 431 = 862 \textrm{ kJ/mol}\).
Step 3: Compare the two totals.
\(\Delta H \approx 679 - 862 = -183 \textrm{ kJ/mol}\).
The negative value means the reaction releases energy overall, so it is exothermic.
This calculation shows the logic clearly: the reaction absorbs energy to separate atoms from their original partners, but it releases even more energy when stronger or more stable bonds form in the products. That difference is transferred to the surroundings.
One of the most important ideas in chemistry is that energy released or absorbed during a reaction is matched by changes in other forms of energy, especially the kinetic energy of particles. When a reaction is exothermic, some of the energy released by bond formation can increase the motion of particles in the surroundings. Since temperature measures the average kinetic energy of particles, the temperature may rise.
In an endothermic reaction, energy must come from somewhere. Often it is taken from the kinetic energy of nearby particles. As those particles slow down on average, the temperature drops. This is why an endothermic cold pack feels cold and why evaporation can cool your skin.
For thermal energy changes, a common relationship is
\[q = mc\Delta T\]
where \(q\) is thermal energy transferred, \(m\) is mass, \(c\) is specific heat capacity, and \(\Delta T\) is temperature change.
Numeric example of temperature change from released energy
Suppose a reaction releases \(4.18 \textrm{ kJ}\) of energy into \(100 \textrm{ g}\) of water. The specific heat of water is about \(4.18 \textrm{ J/(g}\cdot\textrm{ ^\circ C)}\). Find the temperature change.
Step 1: Convert the energy to joules.
\(4.18 \textrm{ kJ} = 4,180 \textrm{ J}\).
Step 2: Rearrange the formula.
\(\Delta T = \dfrac{q}{mc}\).
Step 3: Substitute the values.
\(\Delta T = \dfrac{4,180}{100 \times 4.18} = \dfrac{4,180}{418} = 10\).
The water temperature increases by \(10 ^\circ \textrm{C}\).
Although this formula tracks heating of a substance rather than bond energy directly, it helps connect chemistry to something measurable. A reaction that changes chemical energy can produce a temperature change because particle motion changes too.
These ideas explain many technologies and natural systems. In internal combustion engines, fuel molecules collide with oxygen molecules. A spark supplies activation energy, and then an exothermic chain of reactions rapidly forms products such as \(\textrm{CO}_2\) and \(\textrm{H}_2\textrm{O}\). The released energy increases gas particle motion, producing high pressure that pushes pistons.
In metabolism, food molecules do not simply "contain energy" in a magical sense. Their atoms can be rearranged into products with lower chemical energy through enzyme-controlled pathways. The energy difference is transferred in manageable amounts that cells can use. Enzymes are crucial because they lower activation energies without changing the overall energy balance of the reactions.
Industrial chemistry depends heavily on controlling rate. Manufacturers adjust temperature, pressure, concentration, and catalysts to maximize the number of effective collisions. In the Haber process, nitrogen and hydrogen react to form ammonia:
\[\textrm{N}_2 + 3\textrm{H}_2 \rightleftharpoons 2\textrm{NH}_3\]
This reaction requires carefully chosen conditions because the strong bond in \(\textrm{N}_2\) makes activation energy a major challenge. A catalyst helps the reaction occur fast enough to be useful on a large scale.
Why chemistry links microscopic events to macroscopic observations
What you observe in the lab or in everyday life—flames, cooling packs, rust, spoiled food, engine power, battery heating—comes from molecular events. Reaction rate tells how fast the molecular rearrangements happen, while bond-energy changes help explain whether the surroundings warm up or cool down.
Even environmental issues connect to this picture. Smog formation involves many atmospheric collisions and reaction pathways. Catalytic converters are designed to alter reaction pathways and reduce harmful emissions. Understanding collisions and energy changes is therefore not just an academic topic; it is central to engineering, medicine, environmental science, and energy technology.
A chemical reaction can be understood as a sequence of linked ideas. First, particles move and collide. Second, only collisions with enough energy and proper orientation succeed. Third, successful collisions allow atoms to rearrange into new molecules. Fourth, because the new arrangement has different bonds, the total bond energy changes. Finally, that change is balanced by energy transferred to or from the surroundings, often as changes in particle kinetic energy and temperature.
This unified model explains both rate and energy. Rate depends on collisions and activation energy. Energy release or storage depends on the balance between bond breaking and bond forming. When students connect those two ideas, chemistry stops being a list of separate facts and becomes a coherent explanation of how matter behaves.