Earth is sometimes called the "blue planet," but that nickname hides a deeper scientific truth: water is not just something found on Earth's surface; it is one of the main reasons Earth functions the way it does. Oceans, glaciers, rivers, lakes, groundwater, water vapor, and even tiny amounts of water locked inside rocks all help control weather, climate, erosion, life, and volcanic activity. A world with far less liquid water would not simply be a drier Earth. It would be a fundamentally different planet.
The abundance of liquid water at Earth's surface is unusual in the Solar System. Earth's temperature and pressure allow water to exist naturally in all three physical states: solid, liquid, and gas. That matters because water moves constantly among the atmosphere, hydrosphere, geosphere, and biosphere. As it moves, it carries energy and materials, reshapes land, affects ecosystems, and even helps generate magma deep below the surface. Understanding Earth's systems means understanding water.
Water covers about 71 percent of Earth's surface, mostly in the oceans. Because so much of the planet is covered by liquid water, the hydrosphere strongly interacts with the atmosphere above it and the solid Earth below it. Winds blow across warm or cool ocean surfaces, storms form from evaporating seawater, rivers cut valleys through rock, and groundwater reacts chemically with minerals underground.
What makes water especially important is not just how much of it there is, but also its unusual set of properties. Many substances can absorb heat, freeze, dissolve materials, or flow through cracks. Water does all of these in ways that are unusually effective. It can absorb and store large amounts of energy, release that energy during phase changes, transmit light into aquatic environments, expand when it freezes, dissolve many substances, and influence the physical behavior of rocks.
Hydrosphere is all of Earth's water in liquid, solid, and gaseous forms. Phase change is the transformation of a substance from one state of matter to another, such as evaporation, condensation, freezing, or melting. Density is mass per unit volume, which helps explain why some materials float while others sink.
These properties are not random. They come from the structure of the water molecule itself and the way water molecules attract one another.
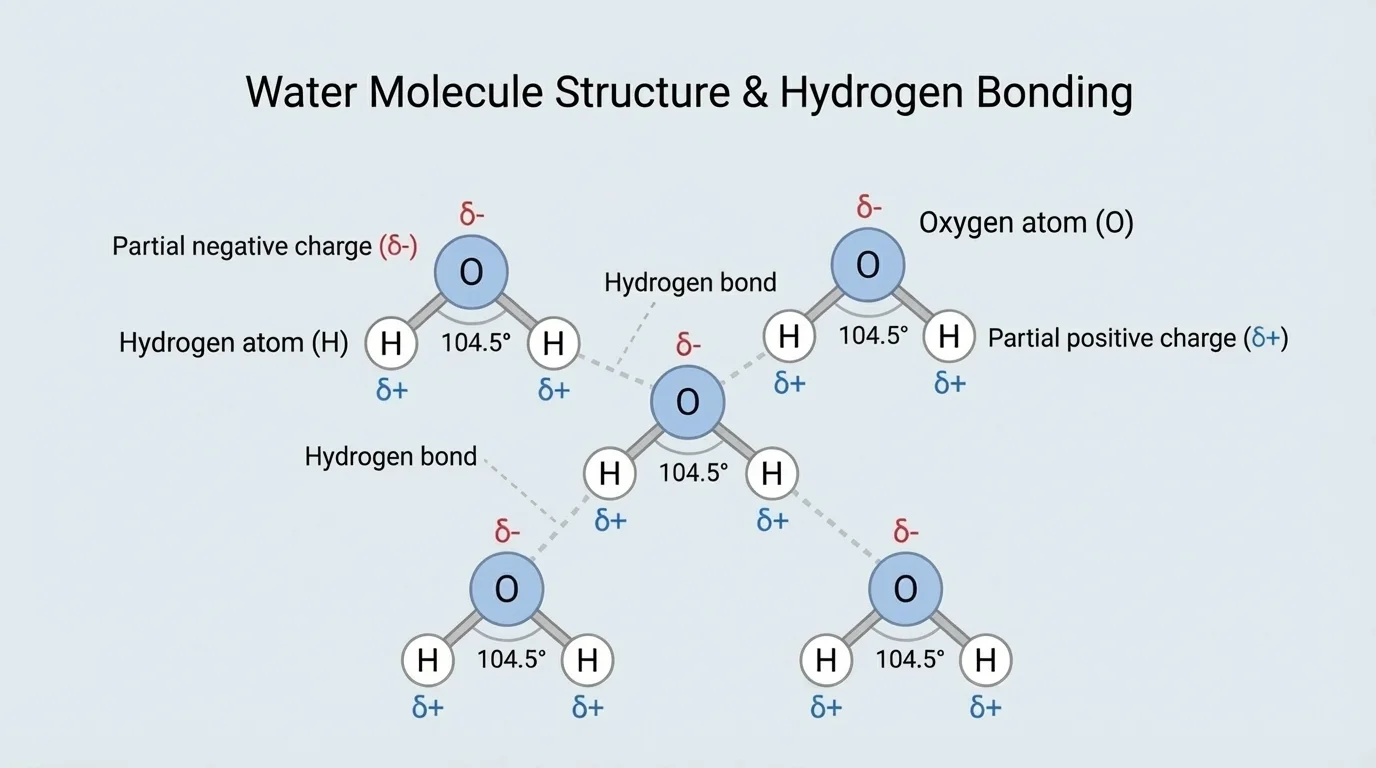
Water's unusual behavior begins with its molecular structure, as [Figure 1] illustrates. A water molecule, \(H_2O\), has two hydrogen atoms bonded to one oxygen atom. Because oxygen pulls shared electrons more strongly than hydrogen does, the molecule has an uneven charge distribution. This makes water a polar molecule, with a slightly negative region near the oxygen atom and slightly positive regions near the hydrogen atoms.
That polarity allows neighboring water molecules to form hydrogen bonds, which are weak attractions between the positive part of one water molecule and the negative part of another. A single hydrogen bond is not extremely strong, but in a large group of water molecules these attractions create major effects. They help explain water's high boiling point, its ability to absorb large amounts of heat, its surface tension, and its unusual behavior when freezing.
If water molecules did not attract each other this way, Earth's climate, weather, oceans, and life systems would all be less stable. Microscopic and planetary scales are directly connected.
One of water's most important properties is its high specific heat capacity, as [Figure 2] shows through ocean-climate interactions. This means water can absorb a large amount of thermal energy before its temperature rises very much. Compared with land, the ocean warms and cools more slowly. That is why coastal regions often have milder climates than inland areas.
On a hot summer day, sand at the beach can become painfully hot while the nearby ocean stays much cooler. The reverse can happen in winter, when the ocean releases stored heat and helps keep nearby air temperatures less extreme. This thermal buffering effect is essential to Earth's climate system.
Water also absorbs and releases energy during phase changes. To evaporate liquid water into water vapor, energy must be absorbed. To condense water vapor back into liquid droplets, energy is released. This is one reason evaporation cools surfaces. When sweat evaporates from skin, it removes energy from the body. At the planetary scale, evaporation from the ocean stores energy in water vapor, and condensation in clouds releases that energy into the atmosphere.
That released energy can power storms. Hurricanes, for example, intensify over warm ocean water because evaporation transfers energy from the ocean to the atmosphere. When the water vapor condenses in towering storm clouds, latent heat is released and fuels rising air. The same basic idea operates in thunderstorms and many large-scale weather systems.
Latent heat and hidden energy transfer
When water changes state, energy moves without necessarily changing temperature at that moment. During evaporation, water absorbs energy to break intermolecular attractions. During condensation and freezing, it releases energy. During melting, it absorbs energy. This hidden transfer helps connect the ocean, atmosphere, and land in a continuous energy exchange.
A simple formula for heat transfer helps show why oceans store so much energy:
\(Q = mc\Delta T\), where \(Q\) is heat energy, \(m\) is mass, \(c\) is specific heat capacity, and \(\Delta T\) is the change in temperature.
For example, if \(1\,\mathrm{kg}\) of water warms by \(5^\circ\mathrm{C}\) and water's specific heat capacity is about \(4{,}186\,\mathrm{J/(kg\cdot ^\circ C)}\), then \(Q = 1 \times 4{,}186 \times 5 = 20{,}930\,\mathrm{J}\). That is a large amount of energy for a small temperature change. Multiply that by the mass of the oceans, and the scale becomes enormous.
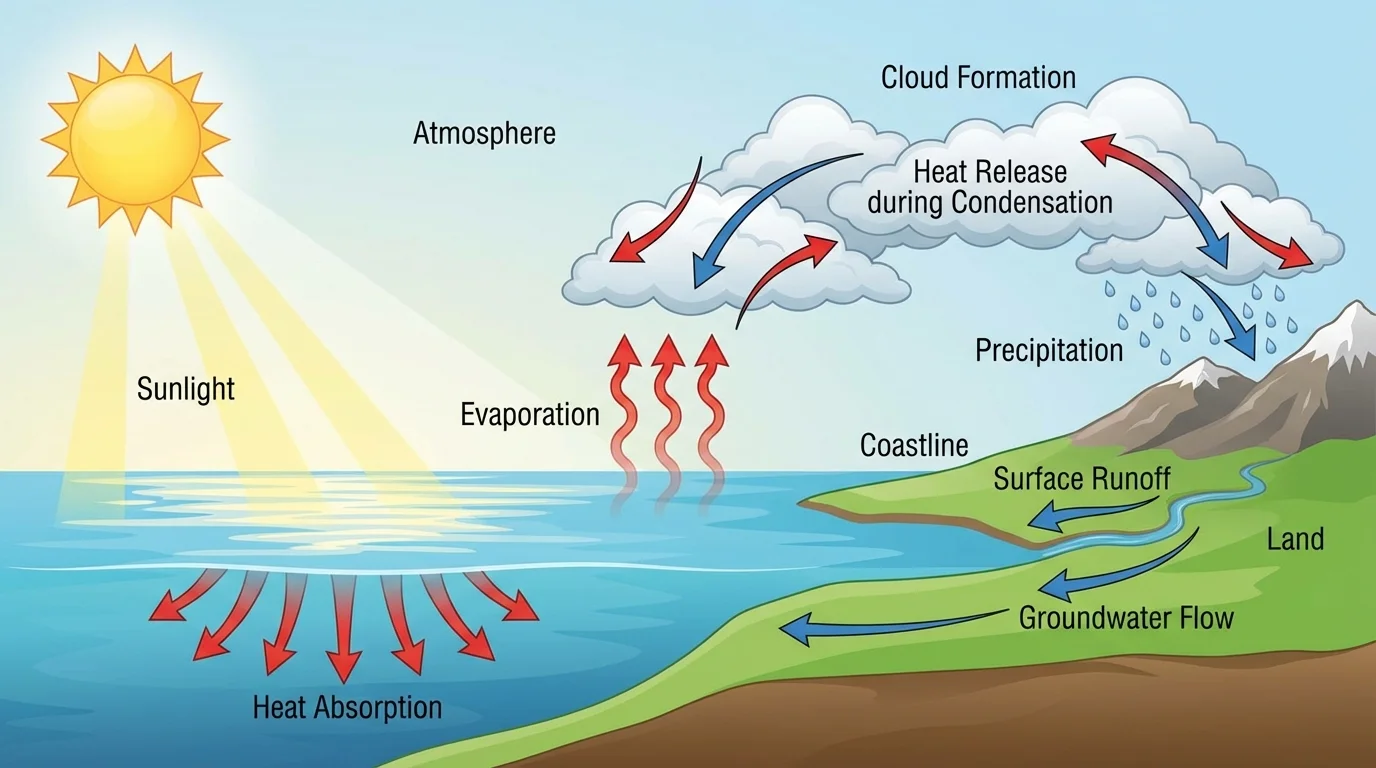
Ocean currents then move that stored heat from place to place. Warm currents can raise the temperature of nearby coasts, while cold currents can cool them. In this way, water is not only a heat reservoir but also a heat transport system. The climate patterns experienced by millions of people are shaped by the movement of water and energy together.
This is one reason climate change is closely tied to the oceans. As greenhouse gases trap more energy in the Earth system, much of that added energy is absorbed by seawater. The large heat capacity of water slows some atmospheric warming in the short term, but it also means the climate system can store changes for a long time.
Water is also important because it transmits sunlight better than many liquids and solids. Sunlight can penetrate the upper layers of oceans and lakes, especially when the water is clear. This allows photosynthetic organisms such as algae and phytoplankton to live in sunlit surface waters. Those organisms form the foundation of many aquatic food webs and produce a large share of Earth's oxygen.
Light does not pass equally through all depths or wavelengths. Red light is absorbed relatively quickly, while blue light penetrates farther, which is one reason clear ocean water often appears blue. As depth increases, light intensity decreases, and photosynthesis becomes difficult or impossible below the sunlit zone.
The ability of water to transmit light links physical properties to biological systems. If light could not enter surface waters, marine ecosystems would be radically different. Fisheries, ocean food chains, and carbon cycling would all be affected.
Some of the most productive regions of the ocean are not the warmest or clearest ones. Areas where deep, nutrient-rich water rises to the surface can support huge blooms of phytoplankton because water carries both sunlight near the top and dissolved nutrients from below.
Water's transparency also matters in lakes. Seasonal changes in heating and cooling can produce layers of different temperature and density. These layers affect how much light reaches deeper water and how oxygen and nutrients are distributed.
Most substances become denser when they freeze, but water behaves differently. When liquid water freezes into ice, hydrogen bonding organizes the molecules into a more open crystal structure. That open arrangement takes up more space, so solid water is less dense than liquid water.
[Figure 3] This is why ice floats. It seems like a simple fact, but it has enormous consequences. If ice sank, lakes and oceans in cold regions could freeze from the bottom up. Instead, floating ice forms an insulating layer at the surface, helping the liquid water below remain unfrozen. Aquatic life can survive underneath winter ice because of this unusual density pattern.
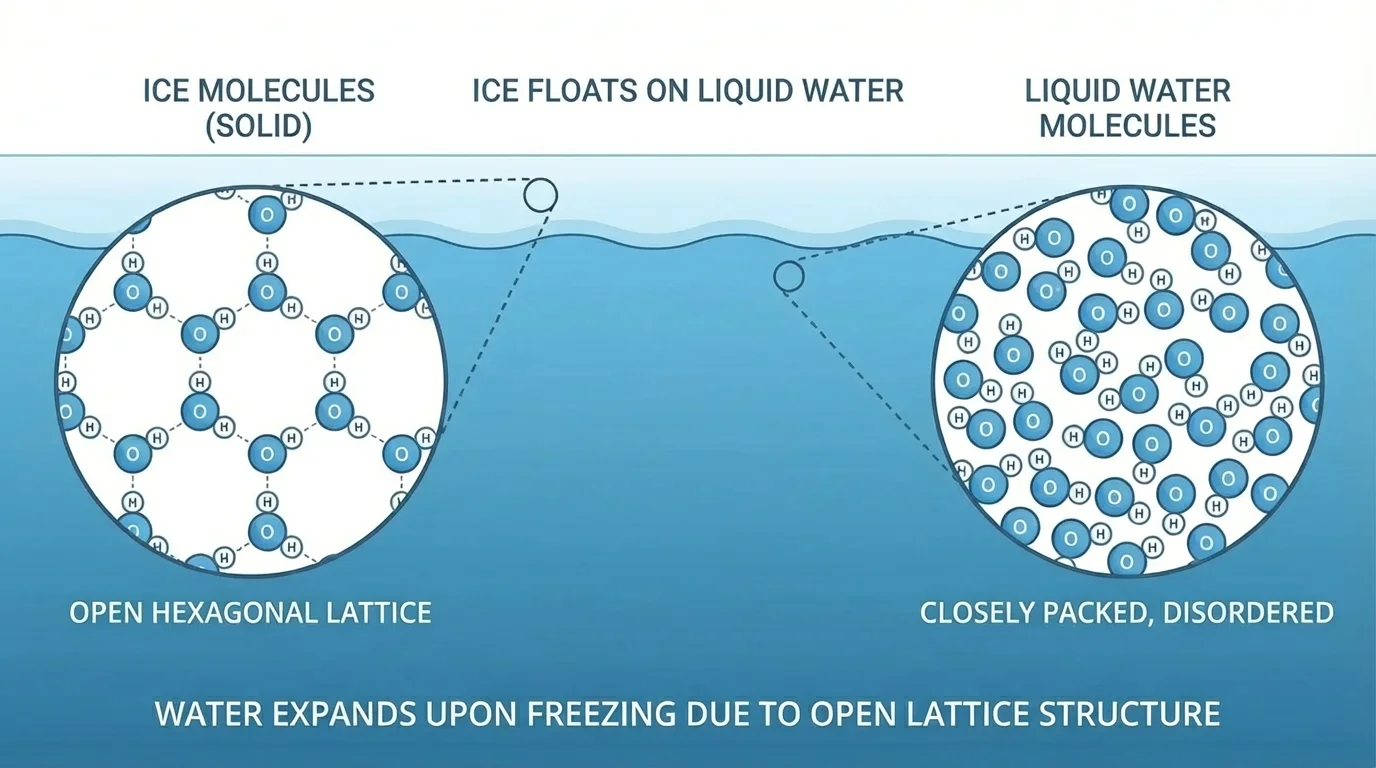
Water reaches its maximum density at about \(4^\circ\mathrm{C}\). As lake water cools in autumn, surface water becomes denser and sinks until the whole lake approaches this temperature. Further cooling produces less dense water at the top, which eventually freezes. This helps explain seasonal mixing and stratification in lakes.
The expansion of water during freezing also contributes to physical weathering. Water can enter cracks in rocks, freeze, expand, and exert pressure on the surrounding material. Repeated freeze-thaw cycles can break rocks apart over time. This process is important in mountain landscapes, road damage, and soil formation.
The same property matters in everyday engineering practice. Pipes can burst in winter because water expands when it freezes. Buildings, roads, and bridges in cold climates must be designed with this behavior in mind. What happens in a freezer at home is connected to what happens in glaciers, cliff faces, and infrastructure.
Because water is polar, it is an excellent solvent for many ionic and polar substances. Salts such as \(\mathrm{NaCl}\) dissolve because water molecules surround and separate the ions. Many gases also dissolve in water to some degree, including \(O_2\) and \(CO_2\). This makes water an important transport medium in both Earth systems and living systems.
When rainwater and groundwater move through soil and rock, they dissolve minerals and carry them elsewhere. This process contributes to chemical weathering, cave formation, nutrient transport, and the chemistry of rivers and oceans. Water can pick up calcium, sodium, silica, bicarbonate ions, and many other dissolved substances as it flows.
Rivers do not transport only liquid water. They also carry dissolved ions, suspended sediments, organic matter, and pollutants. In that sense, a river is both a flow of water and a flow of materials. The chemistry of river water reflects the rocks, soils, vegetation, and human activities of its watershed.
Case study: How water dissolves and moves materials
Consider rain falling on a limestone region, where limestone is mainly \(\mathrm{CaCO_3}\).
Step 1: Carbon dioxide from the air and soil dissolves into rainwater.
\(CO_2 + H_2O \rightarrow H_2CO_3\)
Step 2: This weak acid reacts with limestone and helps dissolve it.
A simplified reaction is \(\mathrm{CaCO_3} + H_2CO_3 \rightarrow Ca^{2+} + 2HCO_3^{-}\).
Step 3: Flowing groundwater carries the dissolved material away.
Over long periods, this can enlarge fractures into caves and underground drainage systems.
Water's role is both chemical and physical: it reacts, dissolves, and transports.
Water's ability as a solvent is essential for life, but in Earth science it also has a second side: it can spread contamination. Fertilizers, heavy metals, road salts, and industrial chemicals can dissolve or become suspended in water and move through streams or aquifers. That is why water quality is such an important environmental issue.
As seen earlier in [Figure 2], water moves energy through evaporation and condensation; here it also moves matter. Earth systems rarely move one thing at a time. Energy flow and material transport are tightly linked.
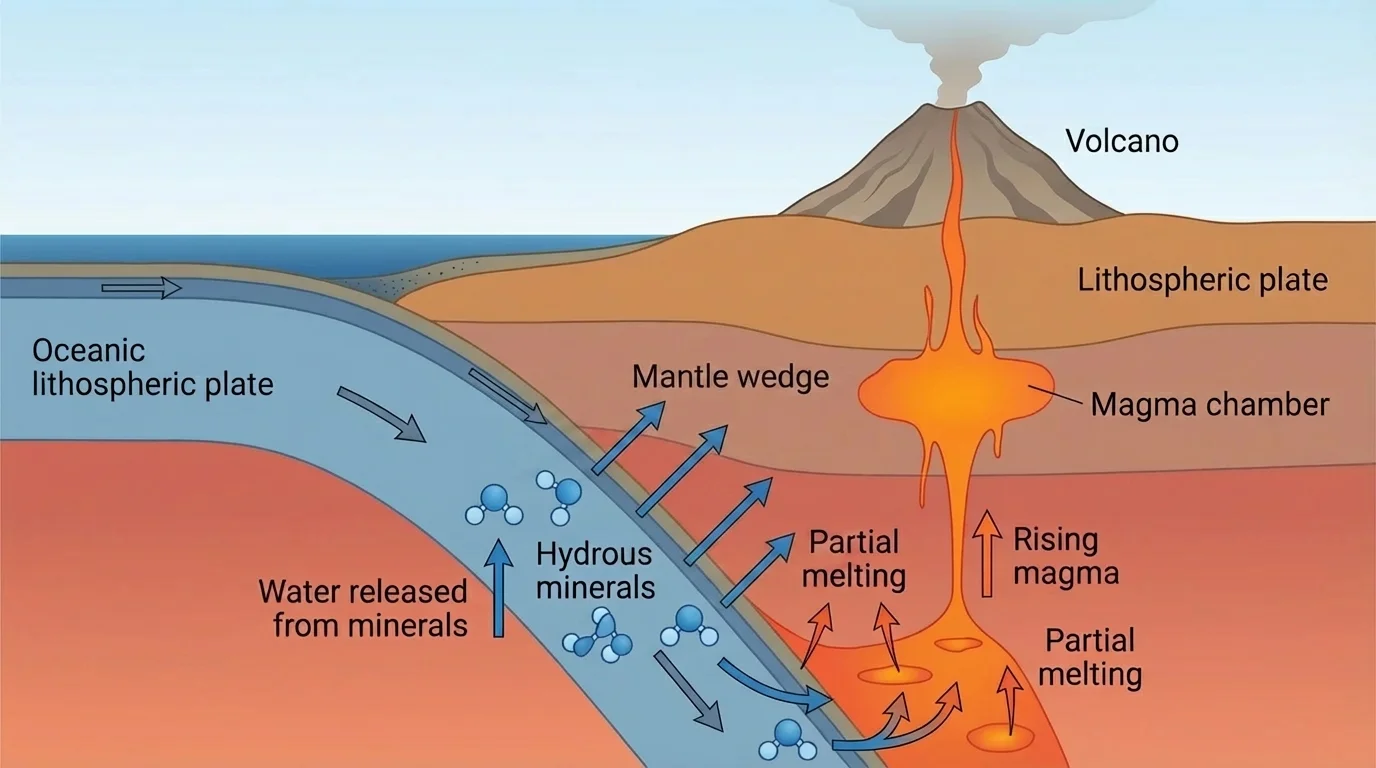
Water affects not only oceans and weather but also the solid Earth deep below the surface, as [Figure 4] demonstrates. In plate tectonic settings, especially at subduction zones, water bound in minerals within oceanic crust and sediments is carried downward into the mantle. As pressure and temperature change, some of that water is released.
That released water lowers the melting point of nearby rocks. In other words, rocks that would remain solid under dry conditions may begin to melt when water is present. This process helps generate magma above subducting plates, which is one reason many volcanoes form in arcs near convergent plate boundaries.
Water also lowers the viscosity of some molten and partially molten materials. Viscosity is a measure of resistance to flow. Lower viscosity means a material can flow more easily. In geological settings, this affects how magma moves, how rocks deform, and how material circulates within Earth.
One way to think about melting point depression is to compare it with road salt melting ice, although the mechanisms are not identical. In both cases, adding another substance changes the conditions under which a solid remains stable. In the mantle, the addition of water can make partial melting possible at lower temperatures than would otherwise be required.
This matters because plate tectonics is not only a mechanical system of moving plates. It is also a chemical and thermal system. Water participates in rock reactions, alters mineral stability, promotes melting, and influences volcanic gases. A dry Earth would likely have a very different style of tectonic and volcanic activity.
Why water changes rock behavior
At high temperature and pressure, water can enter mineral structures, weaken bonds, and change how atoms move through rocks and melts. As a result, rocks can deform more easily, and partial melting can occur under conditions where dry rocks would stay solid. This makes water a powerful agent not only at Earth's surface but also inside the planet.
This connection shows that the movement of water into the mantle links the hydrosphere to volcanism and mountain building. Earth's "surface water story" continues far below the surface.
To understand Earth's systems, it helps to think of water as a connector. It links the atmosphere through water vapor and clouds, the hydrosphere through oceans and rivers, the geosphere through weathering and rock reactions, and the biosphere through all living organisms. Water cycles among these spheres continuously, carrying both energy and matter.
The water cycle includes evaporation, transpiration, condensation, precipitation, infiltration, runoff, groundwater flow, and ice formation or melting. Although the basic sequence is familiar, its consequences are profound. It redistributes freshwater, regulates temperature, drives erosion, replenishes aquifers, and shapes ecosystems.
Humans increasingly alter this cycle. Dams change river flow and sediment transport. Urban surfaces reduce infiltration and increase runoff. Groundwater pumping can lower water tables. Climate change intensifies some parts of the cycle by increasing evaporation and changing precipitation patterns. The same special properties that make water so powerful in natural systems also make changes in water systems especially important for society.
| Property of water | What it means | Earth-system effect |
|---|---|---|
| High specific heat capacity | Warms and cools slowly | Moderates climate and stores ocean heat |
| Latent heat of phase change | Absorbs or releases large amounts of energy during evaporation, condensation, melting, and freezing | Powers weather and redistributes energy |
| Transparency to visible light | Allows sunlight to penetrate surface waters | Supports aquatic photosynthesis and food webs |
| Expansion upon freezing | Ice is less dense than liquid water | Ice floats, insulates lakes, and contributes to weathering |
| Excellent solvent properties | Dissolves and carries many substances | Drives chemical weathering, nutrient transport, and pollutant movement |
| Lowers melting points and viscosities of some rocks and melts | Changes rock and magma behavior | Helps generate magma and influences tectonic processes |
These ideas are not only theoretical. They appear in everyday observations and major natural events. A sea breeze develops partly because land heats faster than water. A winter lake survives beneath ice because frozen water floats. Caves form because groundwater dissolves rock. Volcanic arcs form partly because water released from subducting slabs promotes melting in the mantle.
A simple classroom demonstration can compare heating rates of equal masses of sand and water under the same lamp. The sand usually changes temperature faster, showing that water stores more thermal energy for the same temperature increase. Another demonstration can freeze a bottle partly filled with water to show expansion on freezing. A third can compare how salt dissolves in water but not as easily in a nonpolar liquid, reinforcing the importance of polarity.
Numeric example: comparing heat storage
Suppose \(2\,\mathrm{kg}\) of seawater experiences a temperature increase of \(3^\circ\mathrm{C}\). Using \(Q = mc\) with \(c\) about \(4{,}186\,\mathrm{J/(kg\cdot ^\circ C)}\):
Step 1: Identify known values.
\(m = 2\,\mathrm{kg}\), \(c = 4{,}186\,\mathrm{J/(kg\cdot ^\circ C)}\), and \(\Delta T = 3^\circ\mathrm{C}\).
Step 2: Substitute into the formula.
\(Q = 2 \times 4{,}186 \times 3 = 25{,}116\,\mathrm{J}\)
Step 3: Interpret the result.
Even a small mass of water stores more than \(25{,}000\,\mathrm{J}\) for a modest temperature increase, which helps explain the thermal capacity of lakes and oceans.
The same principles help explain current events such as stronger marine heat waves, coral bleaching, changes in snowpack, and shifts in freshwater supply. Water's special properties are not separate facts to memorize. They are mechanisms that connect climate, geology, ecosystems, and human life.