A star can hold a planet in orbit across millions of kilometers. A tiny charged balloon can bend a thin stream of water without touching it. A magnet can pull a paper clip across a desk. These effects seem almost mysterious because nothing visible connects the objects. Physics explains them with one of its most powerful ideas: space can contain fields that allow objects to affect one another at a distance.
Some forces require direct contact. If you push a box, the force comes from atoms in your hand interacting with atoms in the box. But other forces are noncontact forces. Gravity acts between masses, electric forces act between charges, and magnetic forces act on magnets or moving charges. Even though the objects are separated, the interaction is real and measurable.
To make sense of this, physicists use the idea of a field. A field assigns a physical quantity to every point in space. In simple terms, a field tells you what force an object would experience if it were placed there. A gravitational field tells how a mass would be pulled. An electric field tells how a positive test charge would be pushed or pulled. A magnetic field tells how a moving charge or magnet would be affected.
Force at a distance is an interaction in which one object affects another without direct contact.
Field is a region of space in which an object with the right property, such as mass or charge, experiences a force.
Inverse-square relationship means that when distance doubles, the effect becomes \(\dfrac{1}{4}\) as large; when distance triples, it becomes \(\dfrac{1}{9}\) as large.
Fields are not just convenient drawings. They help explain how energy can move through space. For example, sunlight transfers energy from the Sun to Earth through electromagnetic fields. A falling object gains kinetic energy because of Earth's gravitational field. A motor works because magnetic fields exert forces on moving charges in wires.
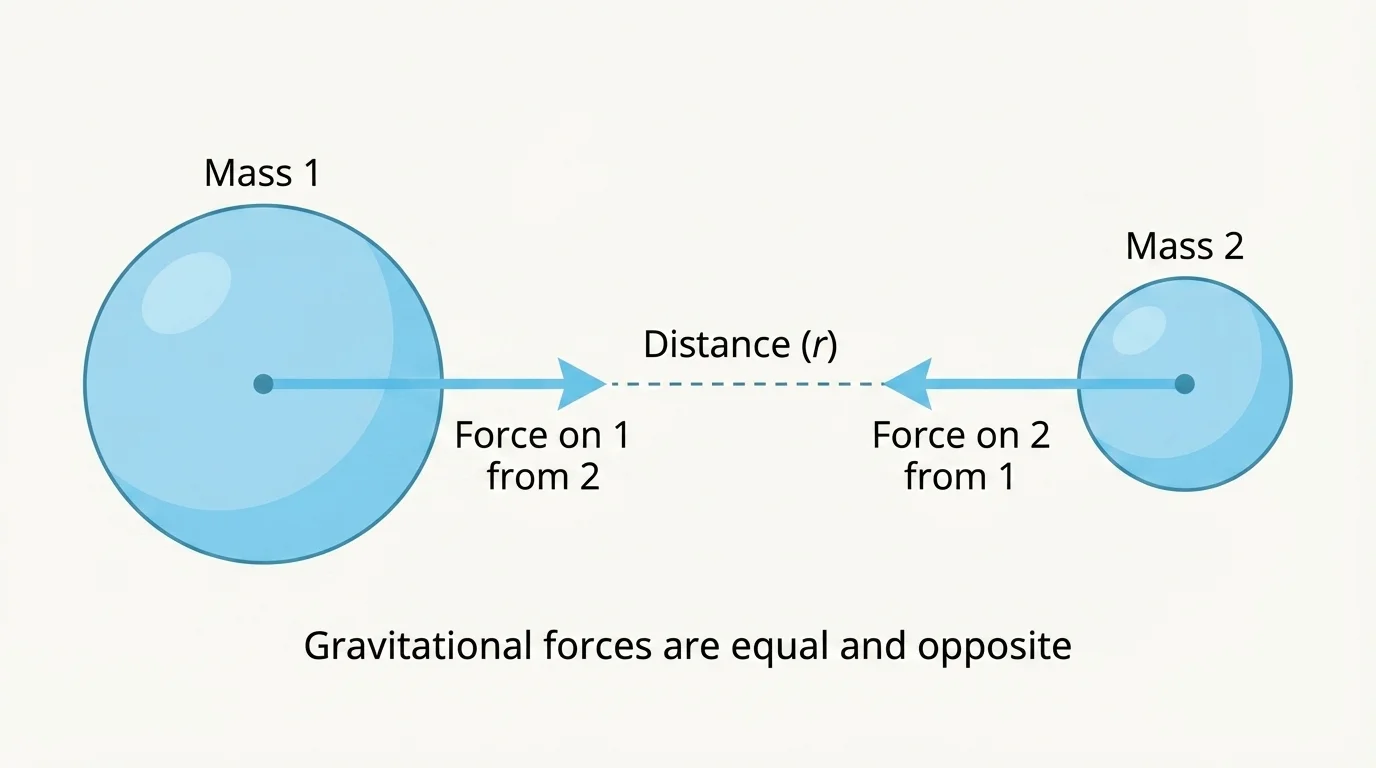
[Figure 1] Gravity is the universal attraction between objects that have mass. Every object with mass attracts every other object with mass. The force is usually noticeable only when at least one of the objects is very massive, such as Earth, the Moon, or a planet. As [Figure 1] shows, the way this force changes with mass and distance is described by Newton's law of universal gravitation. The force acts along the line joining the centers of the two objects, and it becomes much weaker as distance increases.
The law is written as
\[F_g = G\frac{m_1m_2}{r^2}\]
Here, \(F_g\) is the gravitational force, \(G\) is the gravitational constant, \(m_1\) and \(m_2\) are the masses, and \(r\) is the distance between their centers. The equation shows two key ideas: larger masses produce stronger attraction, and greater separation produces weaker attraction according to an inverse-square pattern.
Suppose two students estimate the gravitational force between two \(60 \textrm{ kg}\) people standing \(1.0 \textrm{ m}\) apart. Using \(G \approx 6.67 \times 10^{-11} \textrm{ N m}^2/\textrm{kg}^2\), the force is \(F_g = 6.67 \times 10^{-11} \cdot \dfrac{60 \cdot 60}{1^2}\), which is about \(2.4 \times 10^{-7} \textrm{ N}\). That is far too small to feel. But when one mass is Earth, gravity becomes strong enough to keep oceans, air, and people near the surface.
Gravity explains why planets orbit stars and moons orbit planets. An orbit is not the absence of gravity. It is continuous falling combined with sideways motion. A satellite moves forward while Earth's gravity bends its path into a curved orbit.
Numeric example: how distance changes gravity
If the distance between two objects doubles while their masses stay the same, what happens to the gravitational force?
Step 1: Start with the relationship \(F_g \propto \dfrac{1}{r^2}\).
Step 2: Replace \(r\) with \(2r\).
The new force is \(F_{new} \propto \dfrac{1}{(2r)^2} = \dfrac{1}{4r^2}\).
Step 3: Compare the new force with the original force.
\[F_{new} = \frac{1}{4}F_g\]
Doubling the distance makes the gravitational force four times smaller.
This inverse-square behavior matters in space exploration. If a spacecraft moves farther from Earth, Earth's pull drops quickly. The same pattern appears in many field-based interactions.
A gravitational field describes the gravitational effect of a mass on the space around it. Near Earth, this field causes objects to accelerate downward. Close to the surface, the field strength is approximately \(9.8 \textrm{ N/kg}\), which is also \(9.8 \textrm{ m/s}^2\) for free-fall acceleration.
If a \(2.0 \textrm{ kg}\) object is near Earth, the gravitational force on it is \(F = mg = 2.0 \cdot 9.8 = 19.6 \textrm{ N}\). That simple calculation near Earth is really a special case of universal gravitation. Far from Earth, we use Newton's full law because the field strength changes significantly with distance.
Gravitational fields also connect force with energy. When a book falls from a shelf, gravitational potential energy decreases while kinetic energy increases. No visible rope pulls the book downward, but the field allows energy to transfer through space. In orbits, the exchange between kinetic and gravitational potential energy helps determine speed and path.
Astronauts in orbit are not beyond gravity. They are still strongly attracted to Earth, but they are moving sideways fast enough that they keep missing the ground.
We can now notice another important feature: the two gravitational forces are equal in size and opposite in direction. Earth pulls on the Moon, and the Moon pulls on Earth with the same force, even though Earth's motion changes much less because Earth has much more mass.
Matter can carry electric charge, a property that comes in two types: positive and negative. Unlike charges attract, and like charges repel. This interaction can be much stronger than gravity. That is why electric forces dominate the behavior of atoms, molecules, and most everyday materials.
The mathematical model for the force between two point charges is Coulomb's law:
\[F_e = k\frac{|q_1q_2|}{r^2}\]
Here, \(F_e\) is the electric force, \(k\) is Coulomb's constant, \(q_1\) and \(q_2\) are the charges, and \(r\) is the distance between them. The absolute value reminds us that the formula gives the size of the force; the signs of the charges determine whether the force is attractive or repulsive.
Consider two small charges, \(q_1 = 2.0 \times 10^{-6} \textrm{ C}\) and \(q_2 = 3.0 \times 10^{-6} \textrm{ C}\), separated by \(0.50 \textrm{ m}\). Using \(k \approx 8.99 \times 10^9 \textrm{ N m}^2/\textrm{C}^2\), the force is \(F_e = 8.99 \times 10^9 \cdot \dfrac{(2.0 \times 10^{-6})(3.0 \times 10^{-6})}{(0.50)^2}\), which is about \(0.216 \textrm{ N}\). For such tiny amounts of matter, that is a surprisingly strong force.
Why electric forces often matter more than gravity
Gravity is always attractive and depends on mass, but electric force can attract or repel and is much stronger at the scale of atoms and molecules. In everyday objects, positive and negative charges usually balance overall, so large electric forces are often hidden unless charges are separated, as in static electricity or electrical circuits.
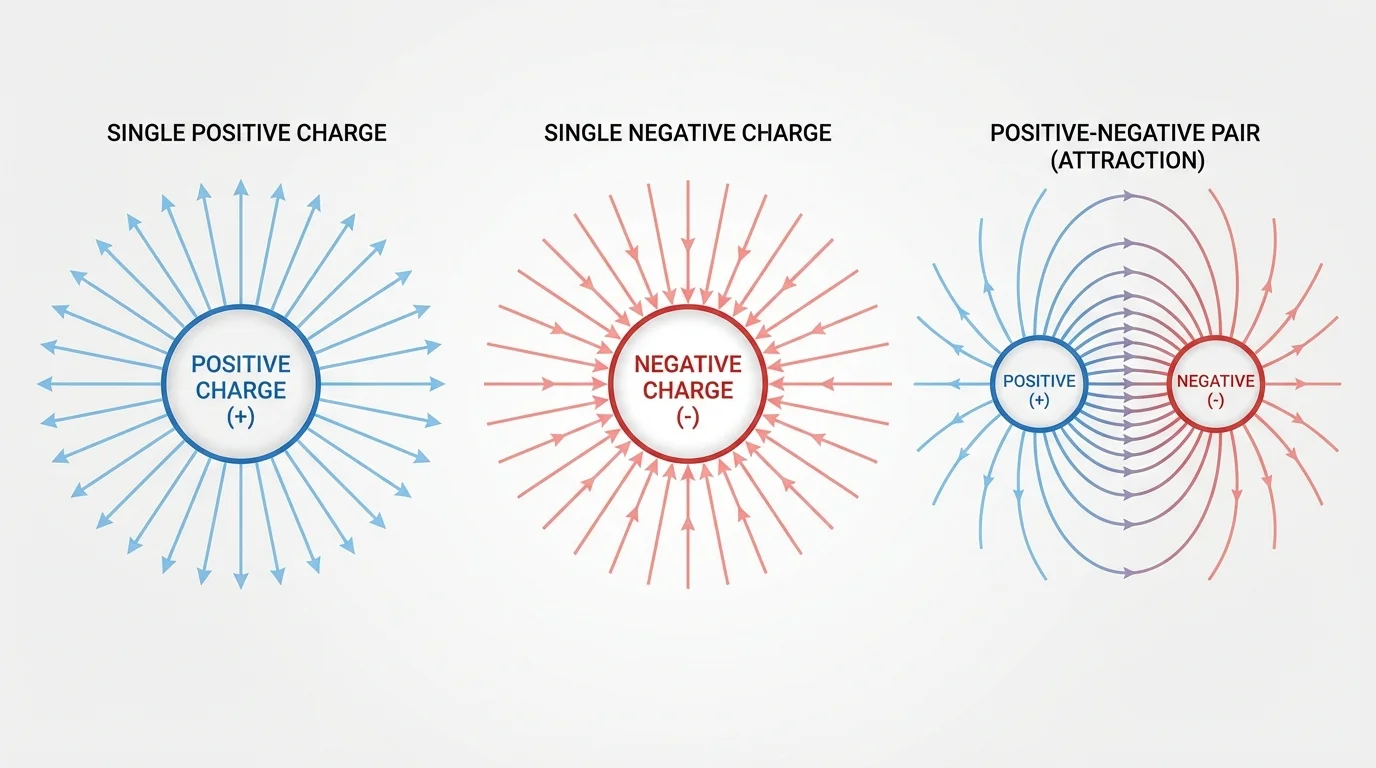
[Figure 2] Like gravity, Coulomb's law follows an inverse-square relationship. If the distance doubles, the electric force becomes \(\dfrac{1}{4}\) as large. But unlike gravity, electric force can push objects apart as well as pull them together.
An electric field describes how a charge influences the space around it. The field points in the direction a positive test charge would move. Field lines point outward from positive charges and inward toward negative charges. Between opposite charges, the lines run from positive to negative.
The electric field strength at a point is defined by \(E = \dfrac{F}{q}\), where \(F\) is the force on a test charge \(q\). If a \(2.0 \times 10^{-6} \textrm{ C}\) test charge feels a force of \(0.040 \textrm{ N}\), then \(E = \dfrac{0.040}{2.0 \times 10^{-6}} = 2.0 \times 10^4 \textrm{ N/C}\).
One fascinating effect of electric fields is polarization. In a neutral object, positive and negative charges are balanced overall. But an external electric field can shift charges slightly, making one side more positive and the other more negative. This is why a charged balloon can stick to a wall and why a plastic comb rubbed on hair can attract small paper pieces.
Static electricity is charge that builds up on surfaces. Rubbing materials together can transfer electrons from one material to another. A classic example is walking across a carpet and then touching a metal doorknob. The sudden spark is a rapid discharge of accumulated charge through the air.
[Figure 3] This pattern also helps explain why pointed conductors are important in lightning rods. Strong electric fields concentrate near sharp points, making it easier for charge to move and reducing dangerous buildup.
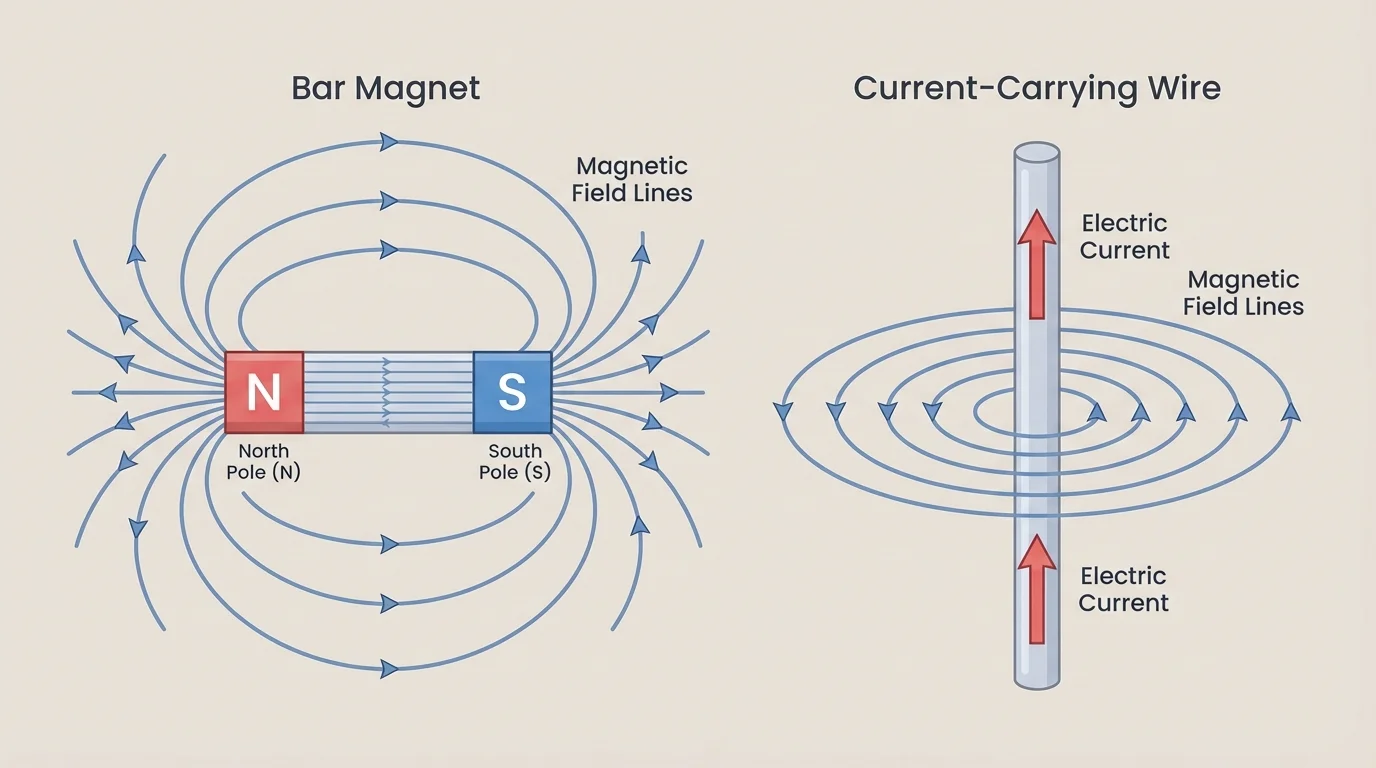
A magnetic field surrounds magnets and moving electric charges. A bar magnet produces looping field patterns, and a straight current-carrying wire produces circular magnetic field lines around the wire. This is a major clue that electricity and magnetism are deeply connected.
Magnets have north and south poles. Opposite poles attract and like poles repel. But magnetic interactions are not exactly the same as electric ones. Isolated magnetic poles have not been observed in ordinary situations; magnets always come with both poles. If you cut a bar magnet in half, you get two smaller magnets, each with a north and a south pole.
Electric currents create magnetic fields. This is the principle behind electromagnets. Wrapping wire into a coil and sending current through it can produce a strong magnetic field, especially if the coil surrounds an iron core. Doorbells, scrapyard cranes, electric motors, and speakers all rely on this effect.
Real-world example: electromagnet in a scrapyard crane
A scrapyard crane lifts steel objects by sending current through a large coil.
Step 1: Current flows through coils of wire.
Moving charges in the wire create a magnetic field around each loop.
Step 2: The fields from many loops add together.
This creates a stronger overall magnetic field, especially with an iron core.
Step 3: The magnetic field attracts ferromagnetic materials such as iron and steel.
When the current is switched off, the field weakens and the load is released.
This ability to turn magnetism on and off makes electromagnets extremely useful in technology.
[Figure 4] The same ideas power electric motors. A current-carrying wire in a magnetic field experiences a force. That force can make a loop of wire rotate, turning electrical energy into mechanical motion.
Electric and magnetic fields are linked so strongly that changing one can create the other. A changing magnetic field causes an electric field. This principle is called electromagnetic induction. It is the basis of generators, which convert mechanical energy into electrical energy.
In a power plant, a turbine spins coils of wire in a magnetic field, or spins magnets near coils. The changing magnetic field through the wire produces an electric field that drives charges through the circuit. That is how large-scale electricity generation works, whether the turbine is driven by steam, falling water, wind, or another source.
Changing electric fields are also connected to magnetic effects. Together, electric and magnetic fields form electromagnetic waves. Light, radio waves, microwaves, and X-rays are all examples. Energy travels through space in these fields, even through the vacuum between the Sun and Earth.
Charges can be present even when they are not moving, but current means charges are in motion. This distinction matters because stationary charges create electric fields, while moving charges also create magnetic fields.
Transformers, wireless chargers, antennas, and MRI machines all depend on the relationship between electric and magnetic fields. These are not separate topics glued together by coincidence; they are different expressions of one electromagnetic interaction.
The structure of matter depends mainly on electric attraction and repulsion at the atomic scale. Negatively charged electrons are attracted to positively charged nuclei, while electrons repel other electrons and protons repel other protons. The balance among these interactions determines atomic size, chemical bonding, and the shapes of molecules and solids.
Atoms bond because electrostatic interactions lower the energy of certain arrangements. In ionic compounds such as \(\textrm{NaCl}\), positive and negative ions attract strongly. In covalent molecules such as \(\textrm{H}_2\) or \(\textrm{H}_2\textrm{O}\), shared electrons are attracted to more than one nucleus. In metals, outer electrons move through a lattice of positive ions, helping explain electrical conductivity and malleability.
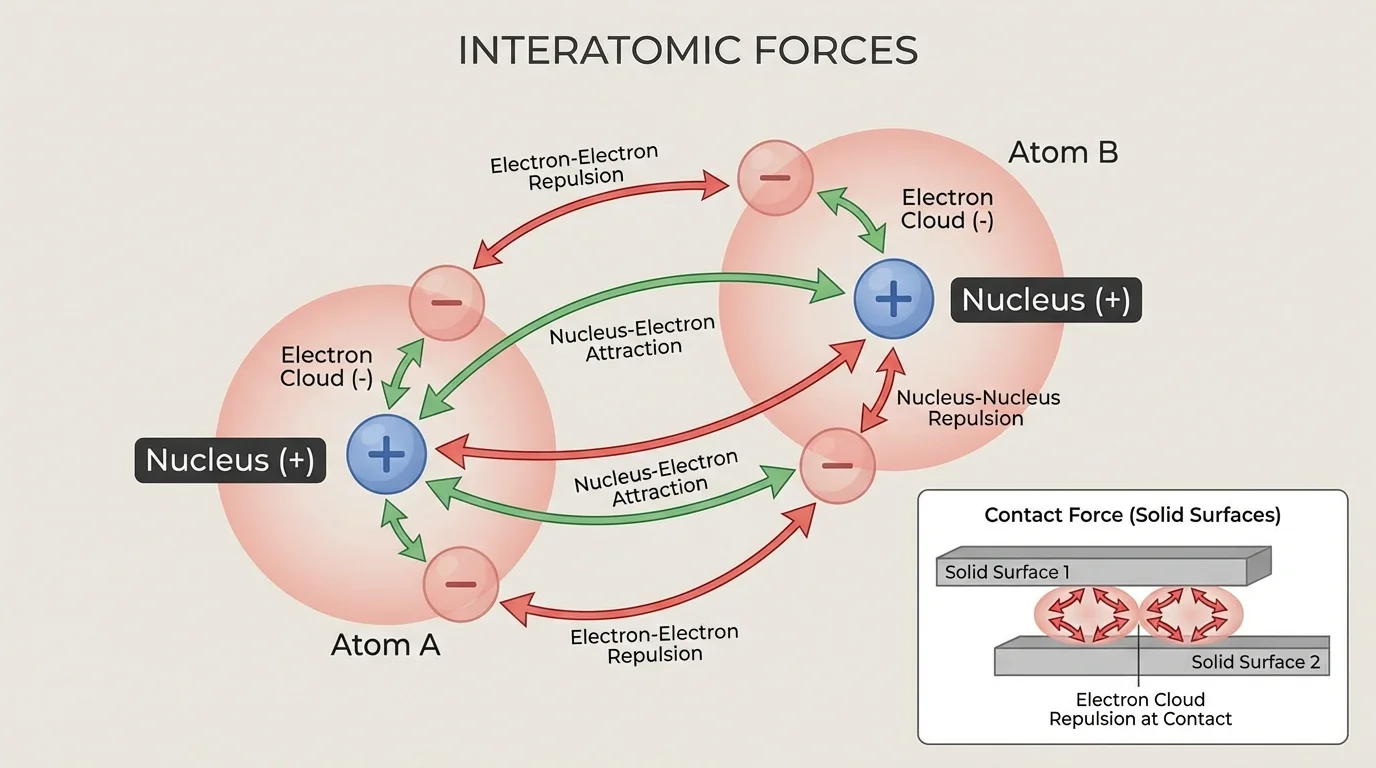
Contact forces, which seem very different from electric forces, are actually caused by electric interactions between atoms. When you press your hand on a table, the atoms in your hand do not sink through the atoms in the table because the electron clouds repel each other strongly at very short distances. The table's upward support force is therefore an emergent effect of atomic-scale electromagnetic interactions.
This also helps explain friction, elasticity, hardness, and why different materials feel and behave differently. Rubber stretches because of the way long molecules interact and return toward lower-energy shapes. Metals bend and conduct because of metallic bonding. Ceramics are often hard and brittle because of strong ionic or covalent bonding patterns.
Chemical transformations are guided by electric forces too. In the reaction \(2\textrm{H}_2 + \textrm{O}_2 \rightarrow 2\textrm{H}_2\textrm{O}\), atoms rearrange as old bonds break and new bonds form. The process depends on attractions between nuclei and electrons and repulsions that prevent atoms from collapsing into one another.
We can connect this back to the same idea: even when two objects appear to touch, what really happens is that countless electric interactions between atoms create the macroscopic forces we call normal force, tension in materials, and much of friction.
Gravitational, electric, and magnetic interactions all involve fields, but they differ in source, strength, and behavior. Seeing them side by side makes the relationships clearer.
| Interaction | Source | Acts on | Can attract? | Can repel? | Typical importance |
|---|---|---|---|---|---|
| Gravitational | Mass | Mass | Yes | No | Planets, stars, falling objects, orbits |
| Electric | Electric charge | Electric charge | Yes | Yes | Atoms, molecules, static electricity, circuits |
| Magnetic | Magnets and moving charges | Magnets and moving charges | Yes | Yes | Motors, generators, electromagnets, storage devices |
Table 1. Comparison of the three major field-based interactions emphasized in this lesson.
A useful pattern appears here: gravity shapes the large-scale structure of the universe because mass is always positive and gravity always attracts, while electric forces dominate the small-scale structure of matter because they are much stronger and can either attract or repel. Magnetism becomes especially important when charges move.
Satellite navigation depends on gravity. Engineers calculate orbital motion using gravitational models so satellites remain at the right altitude and speed. Space missions to the Moon or Mars rely on precise predictions of how gravitational forces from different bodies combine.
Electrostatic painting uses electric force to help paint stick evenly to metal surfaces. Photocopiers and laser printers guide charged toner particles with electric fields. Air purifiers can remove particles by charging them and attracting them to plates with opposite charge.
Magnetic resonance imaging, or MRI, uses strong magnetic fields and electromagnetic signals to produce detailed images of structures inside the body. Electric motors in fans, elevators, and electric cars convert field interactions into motion. Generators in power plants do the reverse, turning motion into electrical energy.
"The most incomprehensible thing about the universe is that it is comprehensible."
— Albert Einstein
The deeper idea behind all of these examples is that fields allow physicists to describe invisible interactions with extraordinary precision. Newton's law predicts planetary motion. Coulomb's law predicts electric force between charges. Field models explain how these interactions fill space, transfer energy, and connect the behavior of galaxies, circuits, magnets, atoms, and matter itself.