You can smell popcorn before you ever see it. You can watch sugar disappear into tea. You can squeeze air into a smaller space, even though it seems like "nothing." These everyday events seem simple, but they give scientists an important clue: matter is made of tiny pieces too small to see with our eyes.
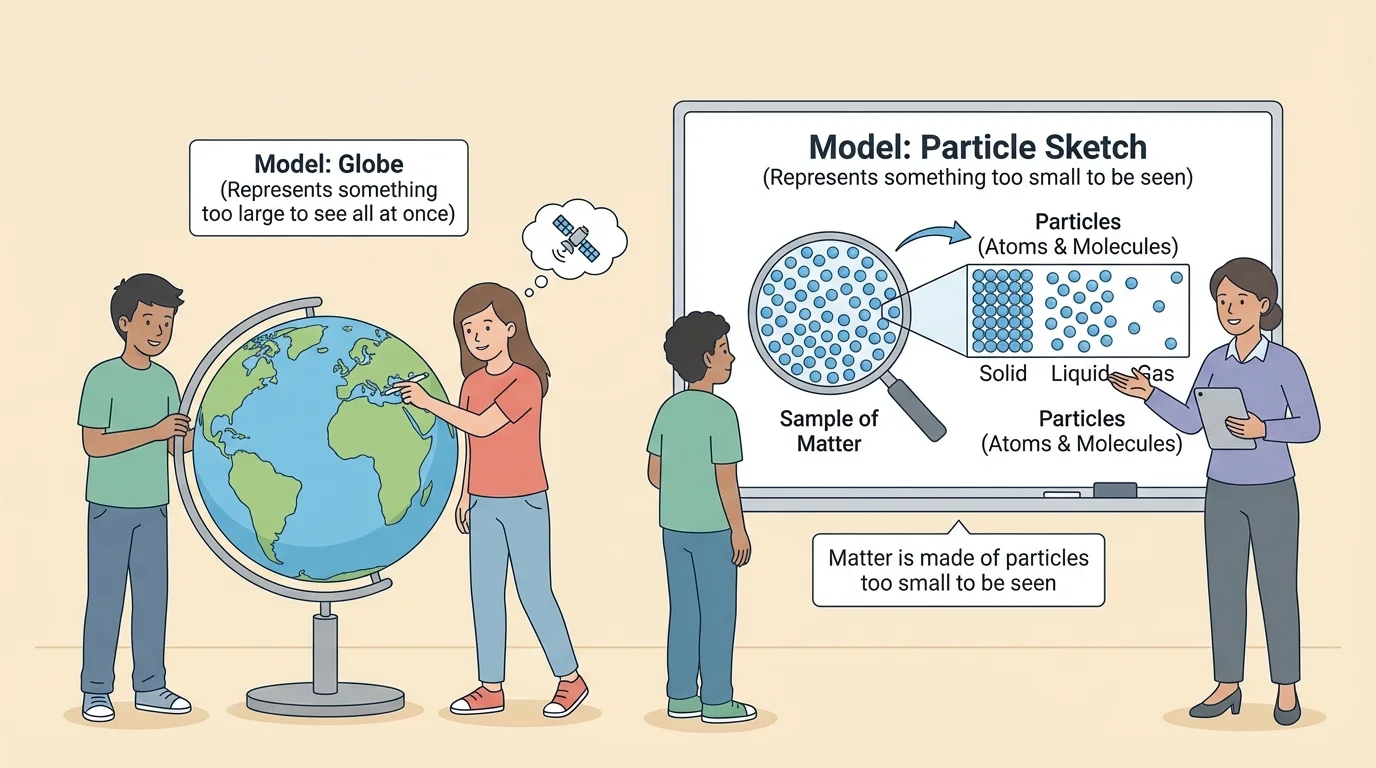
Scientists cannot always look directly at something. Sometimes it is too far away, like a distant planet. Sometimes it is too small, like the particles that make up matter. So scientists build models to explain what they observe. A model is not the real thing. It is a useful way to think about how something works.
A model is a representation of an object, system, or idea. It can be a drawing, a diagram, a physical object, or even a written explanation. In science, models help explain things we cannot directly observe, as [Figure 1] illustrates. A globe is a model of Earth. A weather map is a model of changing conditions. A particle diagram is a model of matter.
When scientists make a model, they base it on evidence. That means they look at what happens, make careful observations, and design a model that fits those observations. If new evidence appears, the model can be changed. That is one reason science is powerful: it keeps improving.
For matter, the model says that everything is made of tiny particles too small to be seen. We do not need to define exactly what those particles are for this lesson. What matters is that this model helps explain many things we can observe.
Think about a bag of marbles. If the bag is full, the marbles take up space. If you pour them into a box, they still take up space, but now they fit the shape of the box differently. Matter is not actually made of marbles, but this comparison helps us start picturing how tiny pieces of matter can help explain what we see.
Matter is anything that has mass and takes up space.
Particle is a tiny piece of matter in a scientific model, too small to be seen with the unaided eye.
Evidence is information from observations or measurements that supports an explanation.
Everything around you that is made of matter has mass and takes up space: books, water, air, desks, clouds, and your own body. Light is not matter because it does not take up space or have mass like matter does. Sound is not matter either. But the air that carries sound is matter.
Matter comes in different forms called states of matter. The three most familiar states are solids, liquids, and gases. A rock is a solid. Juice is a liquid. The air in a balloon is a gas. Even though these forms seem very different, the particle model says they are all made of tiny particles.
This idea helps explain why matter can flow, hold its shape, be compressed, or spread out depending on its state. The particles are not all arranged the same way, and they do not move in the same way in every state.
You already know that using your senses can help you observe matter. You can see shape, feel texture, hear a solid object tap on a desk, or notice a smell traveling through air. In science, these observations become clues that help us explain what is happening.
Even if we cannot see the particles themselves, we can see the effects of how matter behaves. That is how we know the model is useful.
One strong clue comes from the way smells spread. If someone peels an orange in one part of a room, people across the room soon notice the smell. The orange does not jump across the room in one big piece. Instead, tiny bits of matter spread through the air, as shown in [Figure 2]. This suggests that matter is made of particles that can move and mix.
Another clue comes from dissolving. When you stir sugar into water, the sugar seems to vanish. But if you taste the water, the sugar is still there. It has not stopped existing. The particle model explains this by saying that sugar particles spread out among the water particles so evenly that you can no longer see the sugar crystals.
Air gives us more evidence. Air may seem invisible, but it is matter because it has mass and takes up space. If you trap air in a syringe and carefully push the plunger, the air takes up less space. This tells us that there are spaces between particles in a gas, so the particles can be pushed closer together.

A balloon also gives evidence. When you blow into a balloon, it gets larger because more air enters it. If air were not matter, the balloon would not expand. The balloon stretches because the air inside takes up space.
Food coloring in water is another familiar example. Put one drop of coloring into a glass of still water and wait. The color slowly spreads. No one stirs it, yet it moves. This spreading suggests that tiny particles are moving and mixing through the water.
Using evidence from everyday life
Suppose a student says, "Sugar disappears in tea, so it is gone." How can we use evidence to answer?
Step 1: Observe what happens.
The sugar crystals are visible at first, but after stirring, they are not visible anymore.
Step 2: Look for evidence that the sugar is still present.
The tea tastes sweet, so the sugar must still be there.
Step 3: Use the particle model.
The sugar breaks into particles too small to see and spreads through the liquid.
The evidence supports the idea that matter is made of tiny particles and does not simply vanish.
None of these observations lets us see the particles directly. Instead, they give us evidence that a particle model makes sense.
The particle model helps explain the differences among solids, liquids, and gases. In each state, matter is still made of particles. What changes is how closely the particles are arranged and how they move.
[Figure 3] In a solid, particles are packed closely together. This helps explain why solids keep their own shape. A book, a spoon, and an ice cube do not spread out to fill a container. Their particles stay in a more fixed arrangement.
In a liquid, particles are still close together, but they can move past one another. That is why liquids flow and take the shape of their container. Water in a cup, for example, changes shape when poured into a bowl, but the amount of water stays the same.
In a gas, particles are much farther apart than in solids or liquids. They move freely and spread out to fill the whole space available. That is why air fills a room, and why the gas in a balloon spreads to every part inside the balloon.
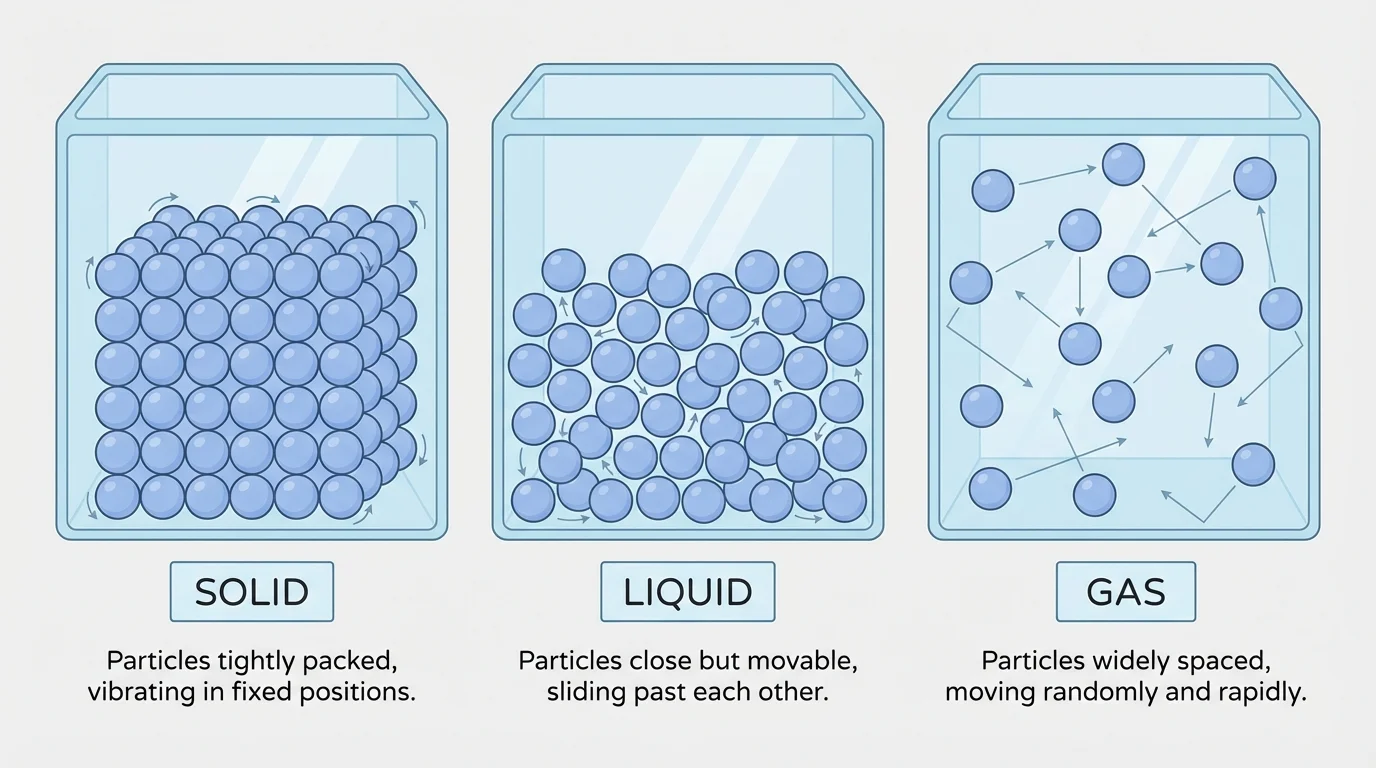
This model explains several familiar facts. Solids are hard to squeeze because their particles are already packed closely. Liquids are also hard to squeeze much. Gases are easier to compress because there is more empty space between particles.
The model also helps explain why a sealed container can feel different when filled with different kinds of matter. A bottle filled with water and a bottle filled with air do not behave the same way because the particles inside are arranged differently.
When you compare these states, remember that the model is simple on purpose. It is meant to explain observations you can make, not every tiny detail. Still, it is very useful. The differences in shape, flow, and ability to be compressed all make sense when we think about the arrangement and motion of particles.
A good scientific model matches evidence
If a model explains many observations clearly, it is useful. The particle model explains why smells spread, why sugar dissolves, why gases can be compressed, and why solids keep their shape. Because it matches evidence from many situations, scientists use it to describe matter.
Earlier, the spreading smell in [Figure 2] showed particles moving through air. That same idea helps explain why gases fill a room and why a spray from one corner can eventually be detected elsewhere.
Observations are important, but measurements make scientific work stronger. Scientists use measurable properties to identify materials. If two objects look alike, measuring their properties can reveal that they are made of different materials.
[Figure 4] One useful property is mass, which is the amount of matter in an object. Another is volume, which is the amount of space an object takes up. Two cubes can have the same volume but different masses if they are made from different materials.
For example, imagine three cubes that are all the same size. One is wood, one is plastic, and one is metal. Since they have equal volume, any difference in mass must come from the kind of material. A metal cube often has a greater mass than a wood cube of the same size.
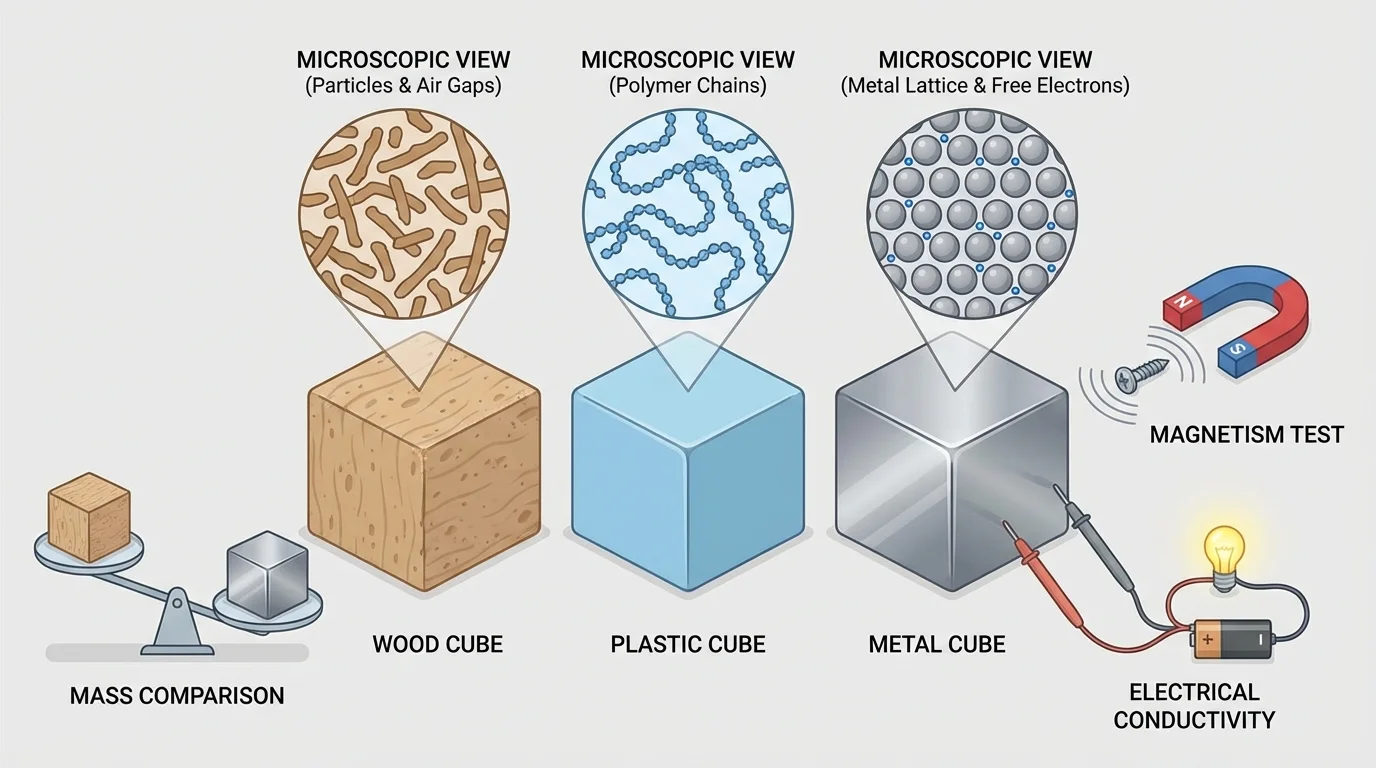
Other measurable properties include magnetism, hardness, and conductivity. A magnet may attract iron but not wood. A metal spoon can conduct heat well, while a wooden spoon does not. These properties help identify what a material might be.
Suppose one block has a mass of \(50 \textrm{ g}\) and another same-size block has a mass of \(15 \textrm{ g}\). Since the blocks have equal volume, the heavier one is likely made of a different material. The measurement does not tell us everything by itself, but it gives evidence we can use.
| Property | What it tells us | Example |
|---|---|---|
| Mass | How much matter is present | A metal cube may have more mass than a wood cube of the same size |
| Volume | How much space an object takes up | A full cup of water has a greater volume than a half cup |
| Magnetism | Whether a material is attracted to a magnet | Some metals are attracted, plastic is not |
| Hardness | How easily a material can be scratched or dented | Glass is harder than clay |
| Conductivity | How well a material carries heat or electricity | Metal conducts electricity better than rubber |
Table 1. Measurable properties that help scientists identify and compare materials.
Measurements do not replace the particle model. Instead, they work together. The particle model explains why materials can behave differently, and measurements help us describe those differences carefully.
A single drop of water contains far more particles than you could ever count one by one. Even though we cannot see those particles, their behavior affects everything from drinking water to clouds in the sky.
When scientists compare materials, they often measure several properties, not just one. A shiny gray object might look like a silvery metal, but if it is light, not magnetic, and does not conduct electricity well, it may be something else. Science relies on multiple pieces of evidence.
A model is strongest when it explains many observations with one big idea. The particle model does that. It explains why solids hold shape, why liquids flow, why gases spread out, why smells move through air, and why some materials are easier to compress than others.
But a model must be tested. Suppose a model said matter was one solid piece with no tiny parts. That model would not explain how sugar seems to disappear into water or how air can be compressed in a syringe. The particle model does a better job because it fits the evidence.
Scientists also revise models when needed. If a new observation does not match the model, they ask questions and improve it. That process is part of science and engineering: using evidence, building explanations, and changing ideas when better evidence appears.
Testing whether a particle model fits observations
Consider a sealed syringe containing air.
Step 1: Observe the system.
When the plunger is pushed, the air takes up less space.
Step 2: Ask what a model must explain.
The model must explain how the same air can fit into a smaller volume.
Step 3: Compare possible models.
If matter were one solid chunk, it would be hard to explain this. If air is made of particles with space between them, the particles can be pushed closer together.
The particle model better matches the evidence.
Back in the solid, liquid, and gas comparison from [Figure 3], the gas box helps us understand why air in a syringe can be compressed more easily than water in a sealed container.
The particle model is not just a classroom idea. It helps explain many real-world situations. In cooking, salt spreads through soup because tiny particles mix through the liquid. In medicine, a smell from rubbing alcohol spreads through air because particles move outward. In engineering, choosing the right building material depends on measured properties such as hardness, mass, and conductivity.
Athletes also depend on matter and its properties. The air in a basketball takes up space and pushes outward on the inside of the ball. The material of a running shoe is chosen for properties like flexibility, mass, and how it handles heat and wear.
At home, particle ideas help explain why a sponge can hold water in tiny spaces, why a sealed chip bag puffs up on an airplane, and why a metal pan gets hot faster than a wooden spoon. In each case, scientists and engineers use observations and measurements to understand matter and choose materials wisely.
Even recycling depends on identifying materials correctly. Workers sort metal, plastic, glass, and paper by observing and measuring their properties. A magnet may separate some metals. Mass and hardness may help identify others. Understanding matter helps us use resources better.
"Science is a way of thinking much more than it is a body of knowledge."
— Carl Sagan
The idea that matter is made of particles too small to see may sound strange at first. Yet once you look at the evidence, it becomes a powerful explanation for many things you notice every day.