When you stir sugar into tea, the sugar seems to disappear. But when you mix baking soda and vinegar, bubbles rush up right away. Both situations involve mixing, yet they are not the same kind of change. Scientists investigate these differences by asking a careful question: Did mixing create a new substance, or did the original substances only change form? Learning how to answer that question helps us understand cooking, cleaning, nature, and even how medicines are made.
Many things around you are made by mixing substances. Cake batter, muddy water, sports drinks, and wet cement all begin with ingredients brought together. Sometimes the mixed materials stay the same substances, just combined. Other times, the substances react and make something new with different properties.
A substance is a kind of matter with its own set of properties. Properties are features scientists can observe, such as color, smell, texture, and whether the material is a solid, liquid, or gas. If a new substance forms, its properties are different from the properties of the starting substances.
Matter is anything that has mass and takes up space. Solids, liquids, and gases are all forms of matter, and all matter can be observed, measured, and described by its properties.
Because appearances can be tricky, scientists do not guess based on one quick look. They gather evidence. They compare what the materials were like before mixing and after mixing. Then they decide whether the evidence supports the idea that a new substance formed.
Scientists often think about tiny particles when they study matter, as [Figure 1] shows. If substances are only mixed together, the particles of each substance are still there, even if they are spread out or hard to see. If a chemical reaction happens, the particles are rearranged and new substances are produced.
A physical change happens when a substance changes size, shape, or state, but remains the same substance. Melting ice, tearing paper, and dissolving salt in water are physical changes. A chemical reaction happens when substances interact and form one or more new substances.
Suppose salt is stirred into water. The salt seems to vanish, but it has not been destroyed. It is still present in the water. If the water evaporates, the salt remains. That is strong evidence that no new substance was formed.
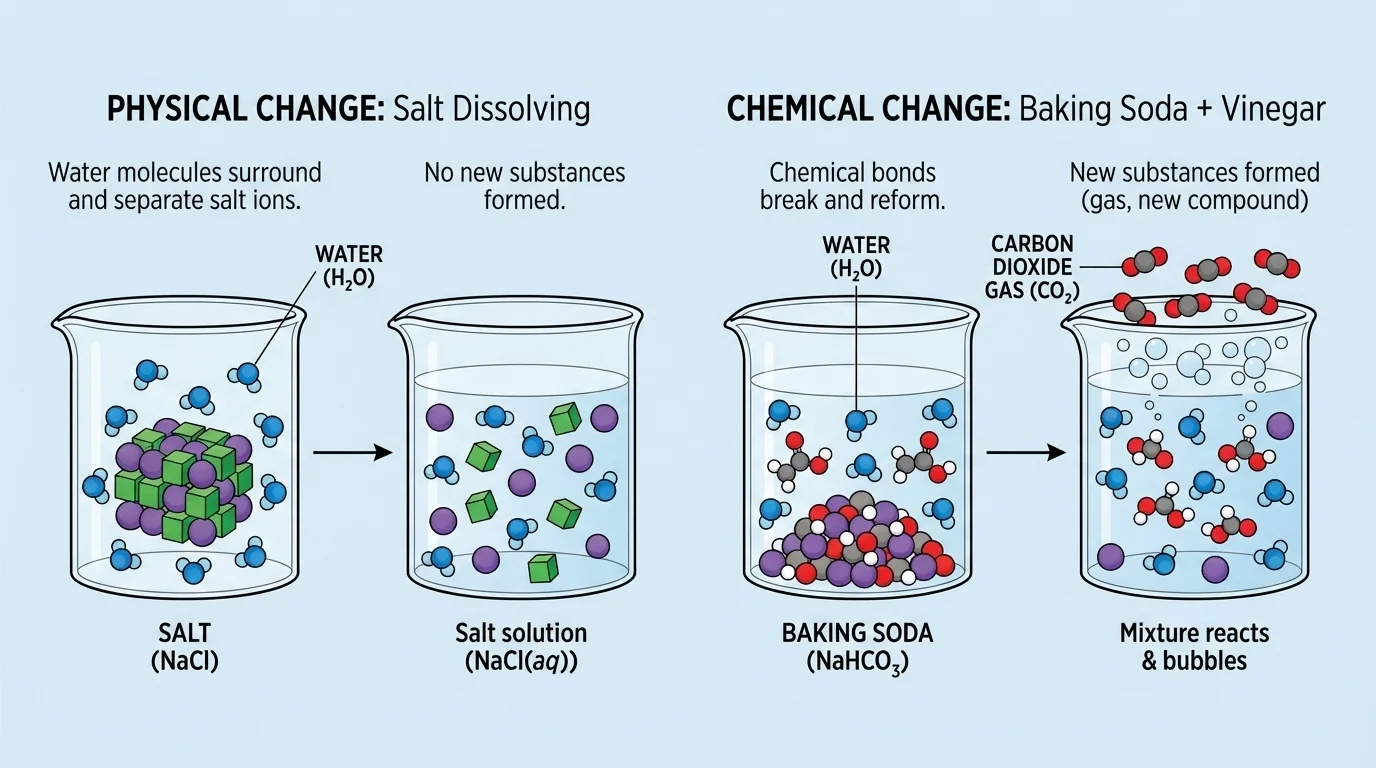
Now think about baking soda mixed with vinegar. The mixture fizzes and produces a gas. The starting materials had one set of properties, and the materials after mixing include a gas and other products with different properties. That is evidence of a chemical reaction.
New substance means a material formed by a chemical reaction that has different properties from the starting substances.
Evidence means observations and measurements that help support a scientific claim.
Not every change means a new substance was made. Chocolate chips melting in a warm cookie are still chocolate. Water freezing into ice is still water, written as \(H_2O\). The form changes, but the substance does not.
Scientists look for signs that suggest a chemical reaction may have happened. One clue is the production of a gas. If bubbles appear and the gas was not just trapped air escaping, a new substance may have formed. This is what happens in the baking soda and vinegar reaction.
Another clue is a temperature change. If two substances are mixed and the container becomes colder or warmer without being heated or cooled from outside, energy is changing during the reaction. This can be evidence that a new substance formed.
A third clue is the formation of a precipitate, which is a new solid that appears when two liquids are mixed. For example, when milk is mixed with lemon juice or vinegar, solid curds form. Those curds have different properties from the original liquid milk.
Scientists may also notice a color change or a new odor. But one clue by itself is not always enough. Food coloring changes water's color, but that does not mean a new substance formed. Good science uses several observations together.
Some reactions are easy to notice, like fizzing or curdling, but others happen slowly. Rust forming on iron is a chemical reaction that can take days, weeks, or even longer to become obvious.
That is why scientists record as much evidence as possible. They watch carefully, measure when they can, and compare before and after.
To investigate whether mixing substances forms new substances, scientists organize materials and observations carefully, as [Figure 2] illustrates. A good investigation begins with a question such as, Does mixing substance A with substance B form a new substance?
Next comes a prediction. A prediction is a statement about what you think will happen based on what you already know. Then scientists decide what they will keep the same and what they will change. This is part of making a fair test.
In a fair test, only one main factor is changed at a time. For example, if you are comparing two mixtures, you should use the same amount of each substance, the same type of container, and the same time for observing whenever possible. That helps you know which change caused the results.
Careful observations matter. Scientists may record color, smell, bubbling, temperature, and whether a solid forms. They may also measure mass before and after mixing. Mass tells how much matter is present.
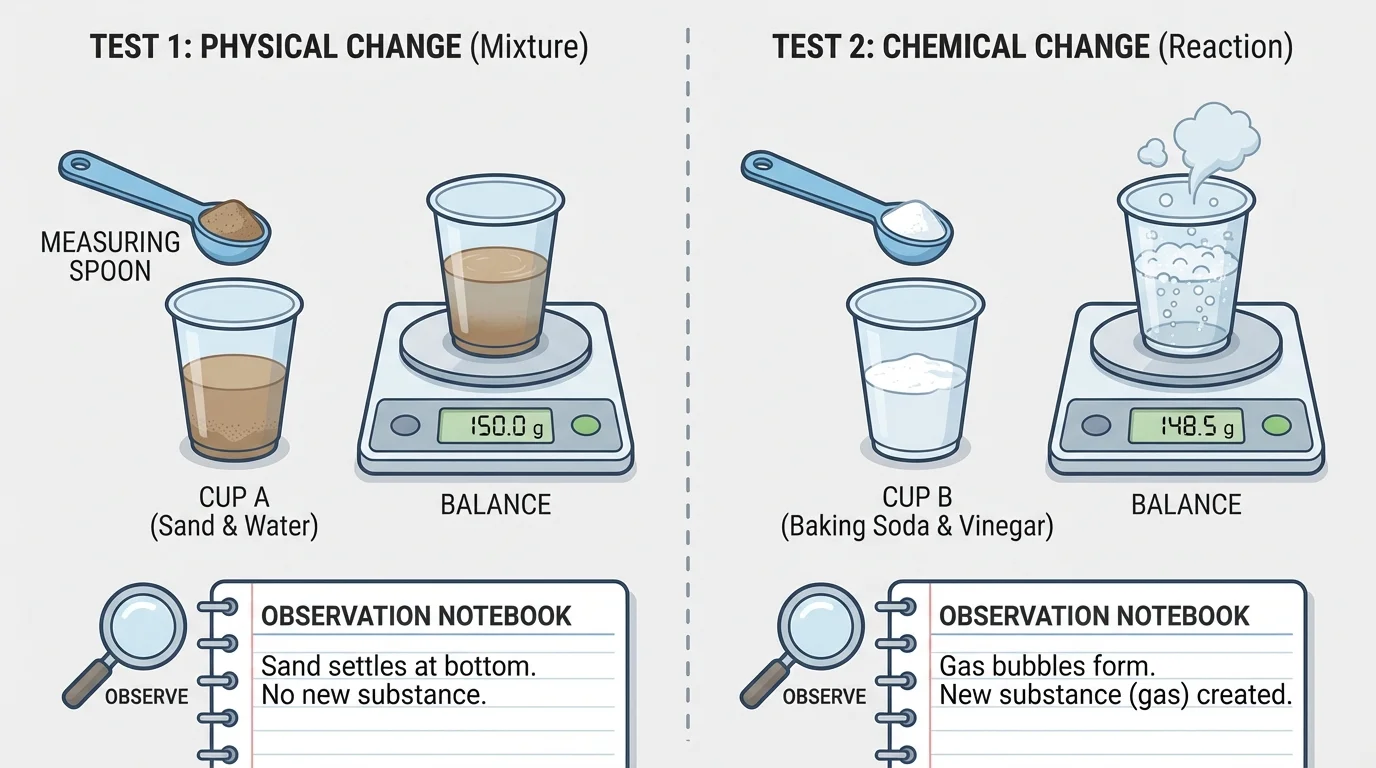
Safety is also part of planning. Students should use only teacher-approved materials, wear eye protection if needed, avoid tasting substances, and wash hands after handling materials. Good science is careful science.
Example investigation: Salt and water
This investigation helps test whether mixing always makes a new substance.
Step 1: Observe the starting substances.
Salt is a white solid. Water is a clear liquid.
Step 2: Mix and observe.
The salt seems to disappear in the water, but there is no fizzing, no new solid, and no clear sign of a new gas.
Step 3: Look for more evidence.
If the water is allowed to evaporate, the salt can be found again. That suggests the salt did not turn into a new substance.
This mixture is best described as a physical change, not a chemical reaction.
Scientists write down their observations in tables so they can compare results clearly.
| Mixture | What was observed? | Possible conclusion |
|---|---|---|
| Salt + water | Salt dissolves; no obvious gas or new solid | No new substance observed |
| Baking soda + vinegar | Fizzing and gas produced | New substances likely formed |
| Milk + lemon juice | Solid curds form | New substances likely formed |
Table 1. Observations from three common mixtures used to investigate whether new substances form.
One useful investigation compares different pairs of substances. This helps students see that some mixtures stay as mixtures while others react.
When baking soda and vinegar are mixed, bubbles form quickly. Those bubbles are evidence of a gas being produced. The gas is carbon dioxide, written as \(CO_2\). The reaction forms new substances that were not present before mixing.
Example investigation: Baking soda and vinegar
Step 1: Observe before mixing.
Baking soda is a white powder. Vinegar is a clear liquid with a strong smell.
Step 2: Mix and watch closely.
The mixture fizzes right away, showing that a gas is being produced.
Step 3: Decide what the evidence suggests.
The gas production is strong evidence that new substances formed during the reaction.
This mixture shows a chemical reaction.
Another investigation uses milk and lemon juice. After mixing, solid clumps called curds appear. Since a new solid forms from two liquids, this is evidence of a chemical reaction.
The particle comparison in [Figure 1] helps explain why these results differ. In a physical change, particles are still the same substances. In a chemical reaction, they are rearranged into new substances with new properties.
One of the most important ideas in chemistry is the conservation of mass, as [Figure 3] shows. This means the total mass of the substances before mixing is the same as the total mass of the substances after the reaction, if all the matter is kept in the system.
Sometimes students think mass disappeared when they see bubbling gas escape into the air. But the matter did not vanish. Some of it simply left the container as a gas. If the reaction happens in a sealed bag or closed container, the gas stays inside, and the total mass can be measured more accurately.
For example, if two substances have masses of \(20 \textrm{ g}\) and \(30 \textrm{ g}\) before mixing, the total mass is \(20 + 30 = 50 \textrm{ g}\). After the reaction, the products together still have a total mass of \(50 \textrm{ g}\), as long as no matter escapes.

This idea does not mean everything looks the same after a reaction. The substances can change greatly. A gas can form, a solid can appear, or the color can change. But the amount of matter stays the same overall.
Why a closed system matters
If a gas leaves an open cup, the cup and its contents may seem to have less mass afterward. In a sealed container, the gas remains part of the system, so the total mass can be measured before and after more fairly.
This setup makes the idea easier to understand because it shows that even when the bag inflates with gas, the balance reading stays the same.
It helps to compare these two kinds of changes clearly, as [Figure 4] presents. Both involve matter, and both can change what something looks like. The big difference is whether a new substance forms.
| Type of change | What happens? | Examples |
|---|---|---|
| Physical change | The substance stays the same, but its form may change | Melting ice, tearing paper, dissolving salt in water |
| Chemical reaction | New substances form with different properties | Baking soda + vinegar, rusting iron, curdling milk |
Table 2. Comparison of physical changes and chemical reactions.
In both cases, students should use evidence rather than guesses. For instance, a color change alone is not enough. A liquid might simply spread out or dilute. But if a color change happens along with gas production or a new solid, the evidence is stronger.
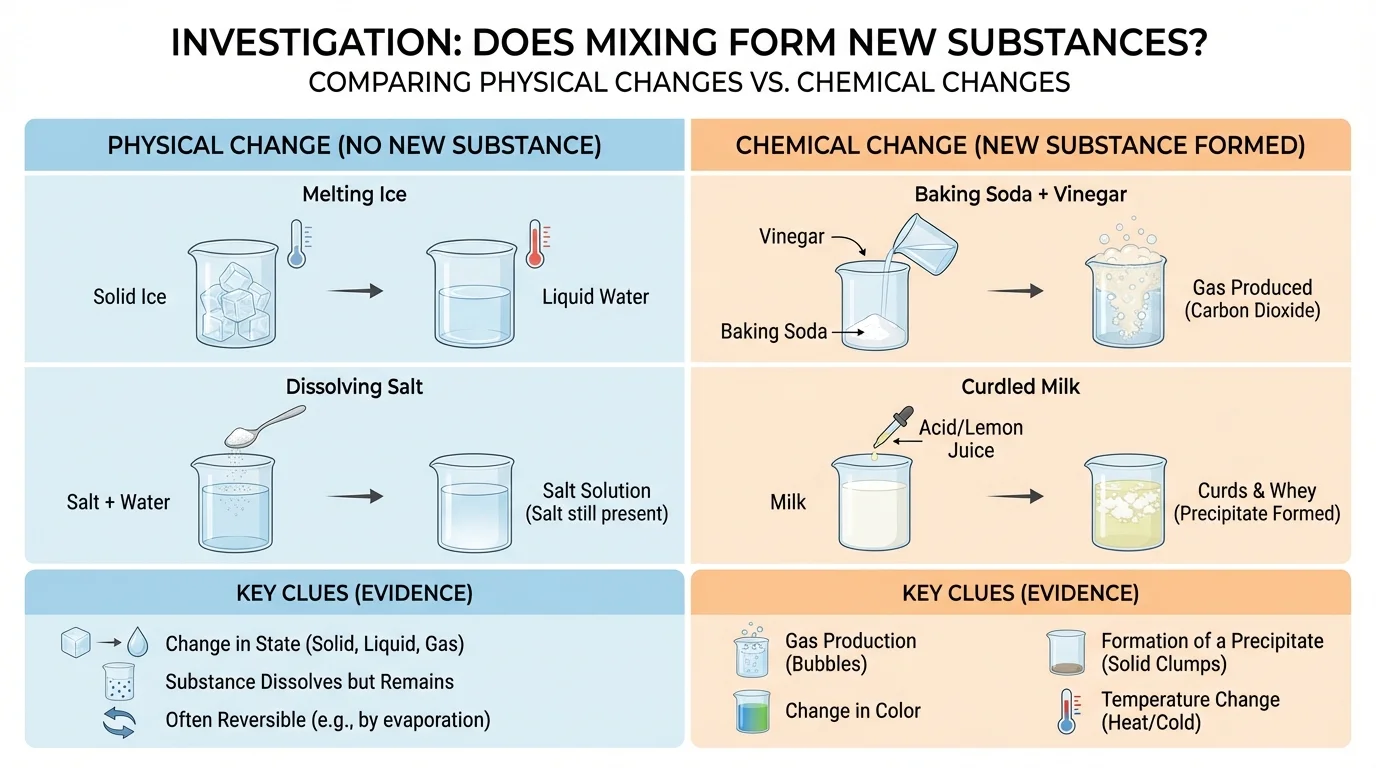
The comparison chart also shows why dissolving and reacting should not be confused. Dissolving can make something hard to see, but the substance is still there.
Chemical reactions are part of daily life. Baking bread depends on reactions that produce gas, which helps dough rise. A cake changes in the oven because heat helps ingredients react and form new substances.
Cleaning tablets that fizz in water are another example. The bubbles show a gas is forming. In nature, leaves decaying, metal rusting, and fruit ripening all involve chemical changes.
Engineers and scientists also need this knowledge. They design products by testing how substances interact. Pharmacists and chemists study reactions when making medicines. Farmers pay attention to soil and fertilizer reactions. Understanding whether a new substance forms helps people make safer choices and better materials.
"Science is a way of thinking much more than it is a body of knowledge."
— Carl Sagan
That idea fits this topic well. The goal is not only to memorize examples, but to think like a scientist by using observations and evidence.
A common mistake is to assume that if something changes shape, size, or state, a new substance must have formed. That is not true. Ice melting and paper being cut are changes, but no new substances appear.
Another mistake is deciding too quickly from one clue. Bubbles might be caused by trapped air, not a reaction. A color change might happen because one substance simply spreads through another. Scientists collect several kinds of evidence before making a claim.
Good investigations use repeated trials, careful notes, and fair comparisons. Students should ask: What did I observe? What evidence supports my idea? Is there another possible explanation?
When students answer these questions, they are doing real science. They are not just watching changes happen. They are investigating whether matter stayed the same or formed something new.