A tablet dropped into water can fizz and seem to disappear. A piece of metal can rust. Cake batter can turn into cake. These changes may look dramatic, but one big science idea stays true: the total weight of the substances does not change. Even when matter changes form or new substances appear, the amount of matter stays the same.
When you see bubbles, smoke, or a solid disappearing into a liquid, it is easy to think some matter has vanished. But matter does not simply pop out of existence. It changes. It may spread out, dissolve, or combine with other matter. If all the matter is kept together, the total weight stays the same.
That idea helps scientists study the world carefully. Whether matter melts, freezes, breaks apart, dissolves, or reacts chemically, the same amount of matter is still there. At this grade level, we use the word weight to describe what is measured on a scale.
Matter is anything that has weight and takes up space. A substance is a kind of matter with its own set of properties. A chemical reaction is a change in which substances are rearranged to form one or more new substances with different properties.
To understand reactions, it helps to think about matter as being made of tiny particles too small to see. During changes, those particles are not destroyed. Instead, they move around or join in new ways. That is why a chemical reaction can make something new while still keeping the same total weight.
Every substance has properties. Properties are characteristics we can observe or measure, such as color, smell, texture, hardness, or whether something dissolves in water. Some properties are easy to notice right away. Others need tools or tests.
For example, sugar is white, grainy, and sweet. Water is clear and liquid at room temperature. Iron is hard and magnetic. Baking soda is a white powder. Vinegar is a sour-smelling liquid. If two substances are mixed and a new substance forms, the new substance may have properties different from either starting material.
This is an important clue: when the properties change in a major way, a chemical reaction may have happened. The starting substances are called reactants, and the new substances made are called products.
You may already know that matter can be a solid, liquid, or gas. Solids keep their shape, liquids flow and take the shape of their container, and gases spread out to fill available space. In chemical reactions, matter can change from one form to another, but it is still matter.
Sometimes a change is easy to reverse. Ice can melt into water, and water can freeze into ice again. Sometimes a change creates something different that is not easy to turn back. Wood burning into ash and gases is an example of that kind of change.
A physical change changes the form, size, or state of a substance, but not the substance itself. A chemical reaction forms new substances. The particle model in [Figure 1] helps explain this difference: in a physical change, particles are still the same substance, but in a chemical reaction, particles are rearranged into something new.
If ice melts, it is still water, written as \(\textrm{H}_2\textrm{O}\). The water particles are the same before and after the change. The only difference is how they move and how closely they are packed. Melting, freezing, cutting paper, and crushing a can are examples of physical changes.
If vinegar and baking soda are mixed, they react and produce new substances, including a gas. The original materials had one set of properties, and the products have different properties. That means a chemical reaction has taken place.
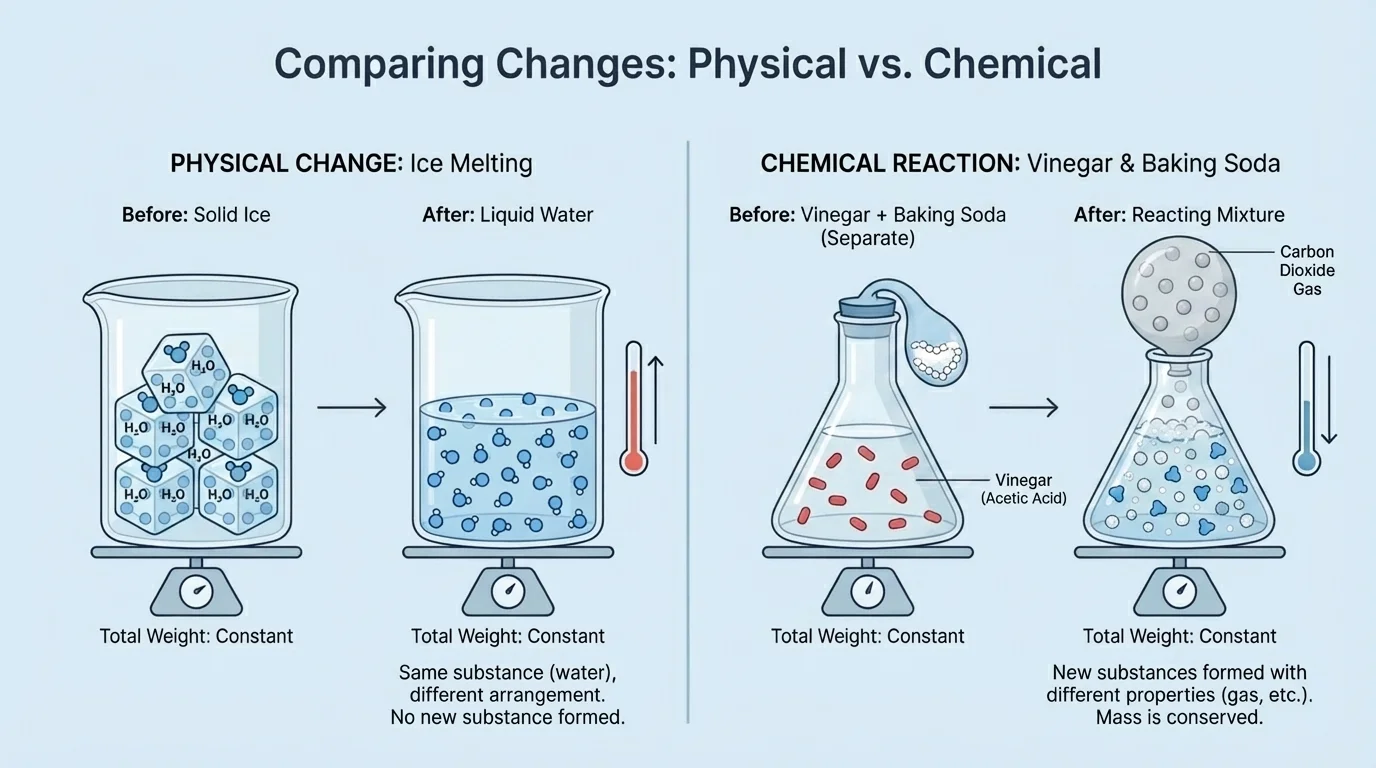
One tricky example is dissolving. When sugar dissolves in water, the sugar seems to disappear, but it is still there. The sugar particles spread through the water. Because no new substance is formed, dissolving sugar in water is usually a physical change.
Cooking can include both kinds of changes. Melting butter is a physical change. But when cake batter bakes and turns into cake, many chemical reactions happen. New smells, textures, colors, and tastes appear.
| Change | What happens | Type of change |
|---|---|---|
| Ice melts | Water changes from solid to liquid | Physical change |
| Sugar dissolves in water | Sugar spreads through water | Physical change |
| Vinegar and baking soda are mixed | New substances form, including a gas | Chemical reaction |
| Iron rusts | Iron reacts with oxygen and forms rust | Chemical reaction |
Table 1. Examples comparing physical changes and chemical reactions.
Scientists look for evidence when deciding whether a chemical reaction happened. No single sign proves it every time, but several clues together can be strong evidence.
One clue is gas production. If bubbles appear when two liquids or a liquid and a solid are mixed, a gas may be forming. In a reaction between vinegar and baking soda, one product is carbon dioxide, written as \(\textrm{CO}_2\).
Another clue is a color change. Shiny gray iron can slowly turn reddish-brown as rust forms. A temperature change is also important. Some reactions release heat, while others take in heat and feel cooler.
A new smell can be a clue too, although smells should never be tested by putting your face close to a chemical. Sometimes a solid forms from two liquids. That new solid is called a precipitate. It is evidence that a new substance may have formed.
Rusting is a slow chemical reaction, but some reactions happen in seconds. The speed of a reaction can vary a lot, even though the same big idea about total weight still applies.
These signs are useful, but scientists also compare properties before and after the change. If the products act differently from the reactants, that is strong evidence of a chemical reaction.
As shown in [Figure 2], in a closed system all the matter is counted before and after the reaction because nothing can escape and nothing from outside can enter. The rule that total weight stays the same is often called conservation of matter.
Think of building with blocks. If you take apart a tower and build a bridge, you still have the same number of blocks. The shape changed, but the amount did not. In a chemical reaction, tiny particles are rearranged in a similar way. The substances change, but the total amount of matter remains the same.
Scientists often describe this idea with a simple relationship:
\[\textrm{total weight before reaction} = \textrm{total weight after reaction}\]
Suppose a sealed container and everything inside it weighs \(150 \textrm{ g}\) before a reaction. After the reaction, the container and everything inside still weigh \(150 \textrm{ g}\). The substances may look different, but the total weight stays equal.

Gases matter too. They may be harder to see, but they still have weight. This is why a gas-forming reaction can keep the same total weight if the gas stays trapped. We can connect this idea back to the particle comparison in [Figure 1]: the particles are rearranged, not lost.
Example: Equal total weight in a sealed reaction
A jar with vinegar and baking soda sealed inside weighs \(240 \textrm{ g}\) before mixing.
Step 1: Identify the rule.
The total weight stays the same if the system is sealed.
Step 2: Apply the rule.
Before reaction: \(240 \textrm{ g}\). After reaction: \(240 \textrm{ g}\).
The reaction makes new substances, but the total weight remains the same.
This idea is true not only for reactions in a science classroom, but also for reactions in factories, engines, living things, and Earth's systems.
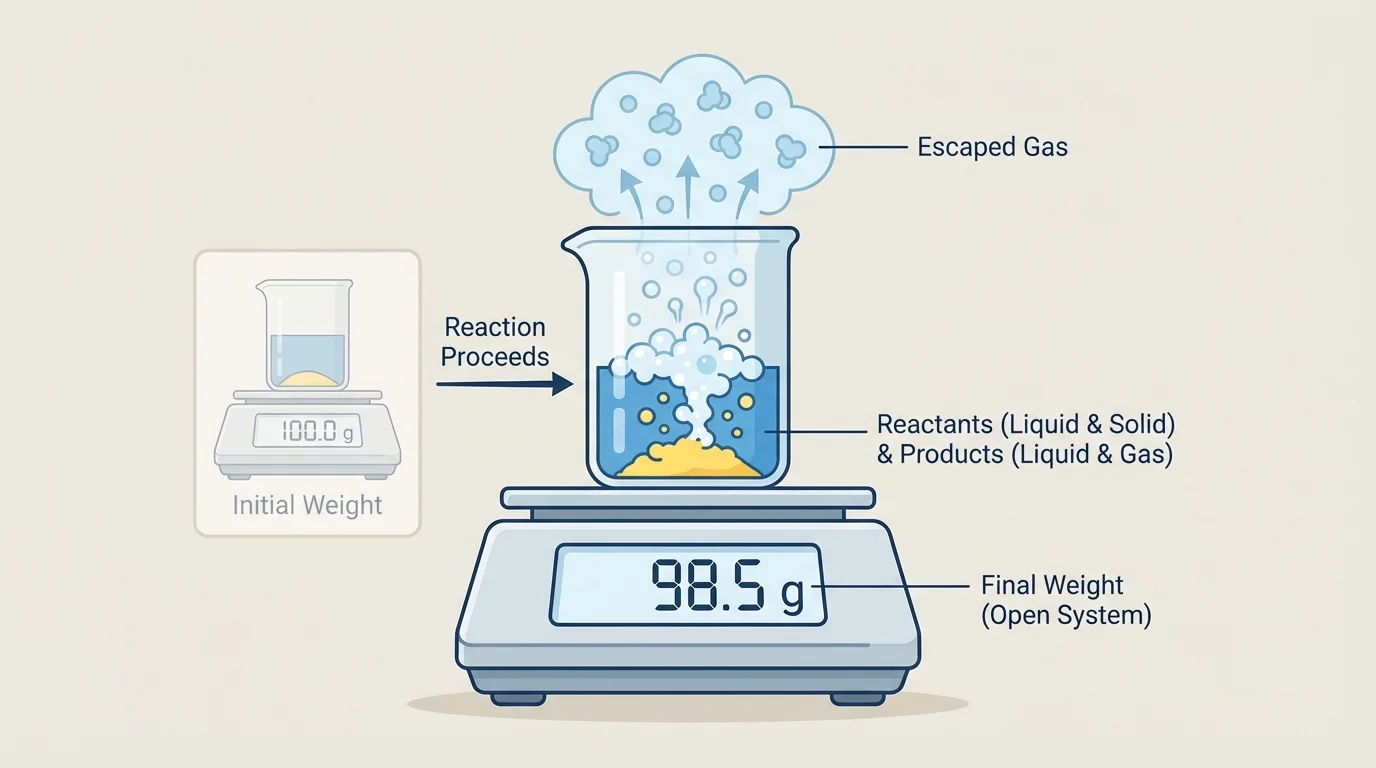
As [Figure 3] illustrates, sometimes a reaction seems to lose weight because a gas leaves an open container and is no longer included in the measurement. The matter is not destroyed; it simply moved somewhere else.
For example, if vinegar and baking soda react in an open cup, carbon dioxide gas can bubble out into the air. If you weigh only the cup and the remaining contents, the reading may be lower after the reaction. But if you could weigh the cup and the escaped gas, the total would match the original amount.
This is why scientists care about whether a system is open or closed. In an open system, matter can leave or enter. In a closed system, matter stays inside. A closed system gives a fairer test of whether total weight stays the same.
Measurements can also look different because of spills, evaporation, or tools that are not used carefully. Good science depends on careful measurement and fair tests.
When students first learn this idea, the escaping-gas example is often the most surprising. A reaction may look like it created or destroyed matter, but really it only changed where the matter is.
Chemical reactions are happening all around you. Food cooking in a pan, leaves rotting on the ground, metal rusting outdoors, and batteries powering devices all involve chemical changes.
Rust is a classic example. Iron reacts with oxygen in the air and often with water too. The product, rust, has different properties from the iron you started with. It is more brittle, rougher, and reddish-brown. Even though the material changed, the total weight of all matter involved stays the same.
Baking is full of reactions. In bread and cakes, ingredients are mixed and heated. Gases can form and make the batter rise. New colors, flavors, and textures develop. If everything were measured in a closed system, the total weight would remain equal before and after the reactions.
Why new properties matter
A chemical reaction is not just a substance changing shape or size. It creates products with different properties. That is why raw egg turning solid when cooked, milk souring, and wood burning are all powerful examples of chemical change.
Another familiar example is an antacid tablet dropped into water. The fizz is gas being produced. The bubbling may look like matter is vanishing, but the gas is still matter. If the gas is kept in the system, the total weight stays the same.
Even your body carries out chemical reactions every moment. Digestion breaks food into simpler substances, and cells use reactions to release energy from food. New substances are formed again and again inside living things.
A safe investigation can make this idea clearer. [Figure 4] shows a sealed plastic bag in which students can mix a small amount of vinegar with baking soda and observe both a chemical reaction and conservation of total weight at the same time.
First, place a little vinegar in the bottom of a sturdy sealable bag. Put baking soda in a small tissue or cup inside the bag without mixing it yet. Seal the bag completely. Weigh the sealed bag. Then tip the bag so the substances mix.
The bag inflates because a gas forms. That is evidence of a chemical reaction. If the bag remains sealed and nothing leaks out, the total weight after the reaction should be the same as before. The shape changes, and new substances form, but the amount of matter stays constant.

This investigation is useful because it answers two questions at once: Did a chemical reaction happen? and Did the total weight stay the same? The inflated bag shows gas production, and the unchanged scale reading shows conservation.
Example: Interpreting the bag investigation
A sealed bag weighs \(86 \textrm{ g}\) before mixing vinegar and baking soda.
Step 1: Observe the evidence.
After mixing, the bag puffs up, so a gas formed. That suggests a chemical reaction happened.
Step 2: Compare the weights.
If the bag is still sealed, the final weight should remain \(86 \textrm{ g}\).
Step 3: Explain the result.
The gas did not disappear. It stayed inside the bag, so all the matter was still included in the measurement.
The reaction forms new substances, but the total weight stays the same.
Later, when thinking about open containers again, the same investigation helps. Unlike the open-cup case in [Figure 3], the sealed bag keeps the gas from escaping.
Scientists often use words and symbols to describe reactions. A chemical equation is a short way to show reactants changing into products. For example, water forms when hydrogen reacts with oxygen:
\[2\textrm{H}_2 + \textrm{O}_2 \rightarrow 2\textrm{H}_2\textrm{O}\]
This equation is a model. It shows that the atoms in the reactants are rearranged to make the product. The number of each kind of atom stays the same on both sides of the arrow, which matches the idea that matter is conserved.
Another example involves carbon dioxide gas forming in a reaction. Carbon dioxide is written as \(\textrm{CO}_2\). Water is written as \(\textrm{H}_2\textrm{O}\). These formulas help scientists communicate clearly about substances.
"Matter is neither created nor destroyed in ordinary chemical reactions."
— A central idea of chemistry
You do not need to balance many equations yet, but it is useful to know that chemical equations support the same big idea you see in experiments: matter is rearranged, not lost.
Understanding chemical reactions helps people in many jobs. Cooks depend on reactions to bake food correctly. Engineers choose materials that resist rust. Doctors and pharmacists study reactions in medicines. Environmental scientists track reactions in air, water, and soil.
Recycling and waste treatment also depend on these ideas. If materials are changed or combined, scientists need to know what products will form and whether they are safe. Knowing that total weight is conserved helps with measuring, designing, and testing processes.
Firefighters, food scientists, builders, and even game designers who simulate realistic worlds use knowledge about materials and their changes. Science becomes more powerful when we understand both what changes and what stays the same.
That is the central idea of this topic: when substances change, the properties may be different, a new substance may form, and the change can be dramatic. But the total weight of all the substances involved remains constant if all the matter is included.