When a campfire burns wood, it can look as if the wood simply disappears. But it does not vanish. The atoms in the wood are rearranged into new substances such as ash, smoke, water vapor, and gases in the air. That idea is one of the most important ideas in science: in an ordinary chemical reaction, matter is not created or destroyed. Instead, tiny particles are rearranged.
You have probably seen food cook, toast burn, metal rust, or a fizzy tablet bubble in water. These are all examples of matter changing. Some changes are only physical, such as ice melting into water. Other changes are chemical, meaning new substances are formed. A chemical reaction can make substances look very different, but the total amount of matter is still there.
This can feel surprising at first. If a marshmallow gets smaller in a flame, where did the matter go? Some of it becomes gases that spread into the air. Some becomes black carbon-rich material. The matter has changed form and location, not vanished. To understand this, scientists use models.
Matter is anything that has mass and takes up space. All matter is made of particles too small to see without powerful tools. In middle school science, a useful model is to think of matter as being built from atoms that can join together in different ways.
Models are especially helpful because atoms are far too small to watch directly during a reaction. By drawing them as colored circles or using beads, cubes, or counters, we can keep track of what happens.
An atom is the smallest unit of an element that still has the properties of that element. For example, oxygen atoms are one kind of atom, and hydrogen atoms are another kind. A molecule forms when two or more atoms bond together. Water is a molecule made of hydrogen and oxygen atoms, written as \(\textrm{H}_2\textrm{O}\).
Different substances have different combinations of atoms. Oxygen gas in the air is often found as \(\textrm{O}_2\), which means each oxygen molecule has two oxygen atoms joined together. Carbon dioxide is \(\textrm{CO}_2\), which has one carbon atom and two oxygen atoms. These formulas help us count atoms.
Chemical reaction is a process in which atoms in the starting substances are rearranged to form new substances.
Reactants are the starting substances in a chemical reaction.
Products are the new substances formed by a chemical reaction.
Conservation of mass means the total mass stays the same during a chemical reaction if all matter is included.
Notice what that definition does not say. It does not say atoms are broken out of existence or that new atoms suddenly appear from nowhere. The key idea is rearrangement.
In a chemical reaction, old connections between atoms can break, and new connections can form. This creates different molecules. For example, hydrogen gas and oxygen gas can react to form water. The substances before and after are different, but the atoms involved are the same hydrogen and oxygen atoms.
There are often clues that a chemical reaction is happening: a color change, bubbles, light, heat, a new smell, or a solid forming from liquids. These signs do not prove exactly which atoms are present, but they tell us that a chemical change may be taking place.
Rearrangement, not disappearance
A good way to think about a chemical reaction is as a building set. If you take apart one structure made of blocks and build a different structure, you still have the same total number of blocks. In a reaction, atoms are like those blocks. The arrangement changes, but the atoms are conserved.
This is why scientists often say that reacting substances rearrange to form different molecules. The number of atoms of each element remains the same.
A model is a simplified representation of something that helps us understand it. In chemistry, a particle model uses symbols, drawings, or objects to stand for atoms and molecules. As [Figure 1] shows, a model lets us track individual atoms as they move from reactants to products.
Suppose we use white circles for hydrogen atoms and red circles for oxygen atoms. Before a reaction, we might draw hydrogen molecules as pairs of white circles, \(\textrm{H}_2\), and oxygen molecules as pairs of red circles, \(\textrm{O}_2\). After the reaction, we draw water molecules, \(\textrm{H}_2\textrm{O}\), with two white circles attached to one red circle. The picture changes, but the total number of white and red circles does not.
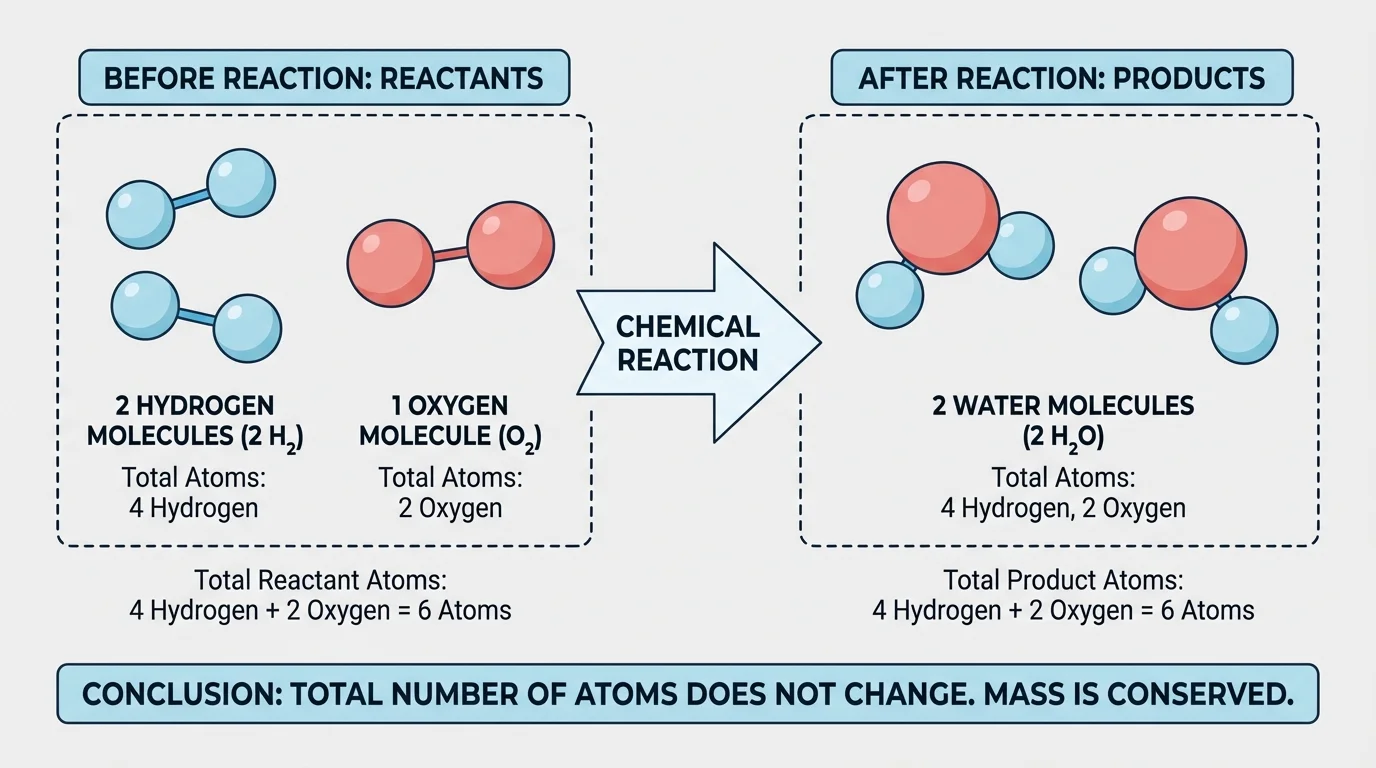
This kind of model is powerful because you do not need to know the masses of individual atoms to understand conservation. You simply count how many atoms of each type are present before and after.
For example, if you start with four hydrogen atoms and two oxygen atoms, you can rearrange them into two water molecules. Each water molecule has two hydrogen atoms and one oxygen atom, so two water molecules contain four hydrogen atoms and two oxygen atoms. Nothing is lost.
Model example: making water
Look at the reaction in words: hydrogen reacts with oxygen to form water.
Step 1: Count the atoms in the reactants.
Two hydrogen molecules contain \(4\) hydrogen atoms total, and one oxygen molecule contains \(2\) oxygen atoms total.
Step 2: Build the products from the same atoms.
Two water molecules, \(2\textrm{H}_2\textrm{O}\), contain \(4\) hydrogen atoms and \(2\) oxygen atoms.
Step 3: Compare before and after.
Before: hydrogen \(= 4\), oxygen \(= 2\). After: hydrogen \(= 4\), oxygen \(= 2\).
The atoms are rearranged, so the new substance is different, but the total number of atoms of each element stays the same.
The same idea works whether the reaction is simple or more complicated. A good model always helps you answer one question: are all the atoms accounted for?
When students first learn this topic, they sometimes focus on how different the products look. A gas can become a liquid, a clear liquid can become a cloudy solid, or a black solid can become invisible gases. But appearance can be misleading. As [Figure 2] illustrates, the important check is to count each type of atom separately.
Consider a reaction involving methane, the main gas in many stoves. Methane is \(\textrm{CH}_4\). When it reacts with oxygen, the products can be carbon dioxide, \(\textrm{CO}_2\), and water, \(\textrm{H}_2\textrm{O}\). If we use a particle model, we can count the carbon, hydrogen, and oxygen atoms before and after the reaction and see that the totals match when all particles are included.
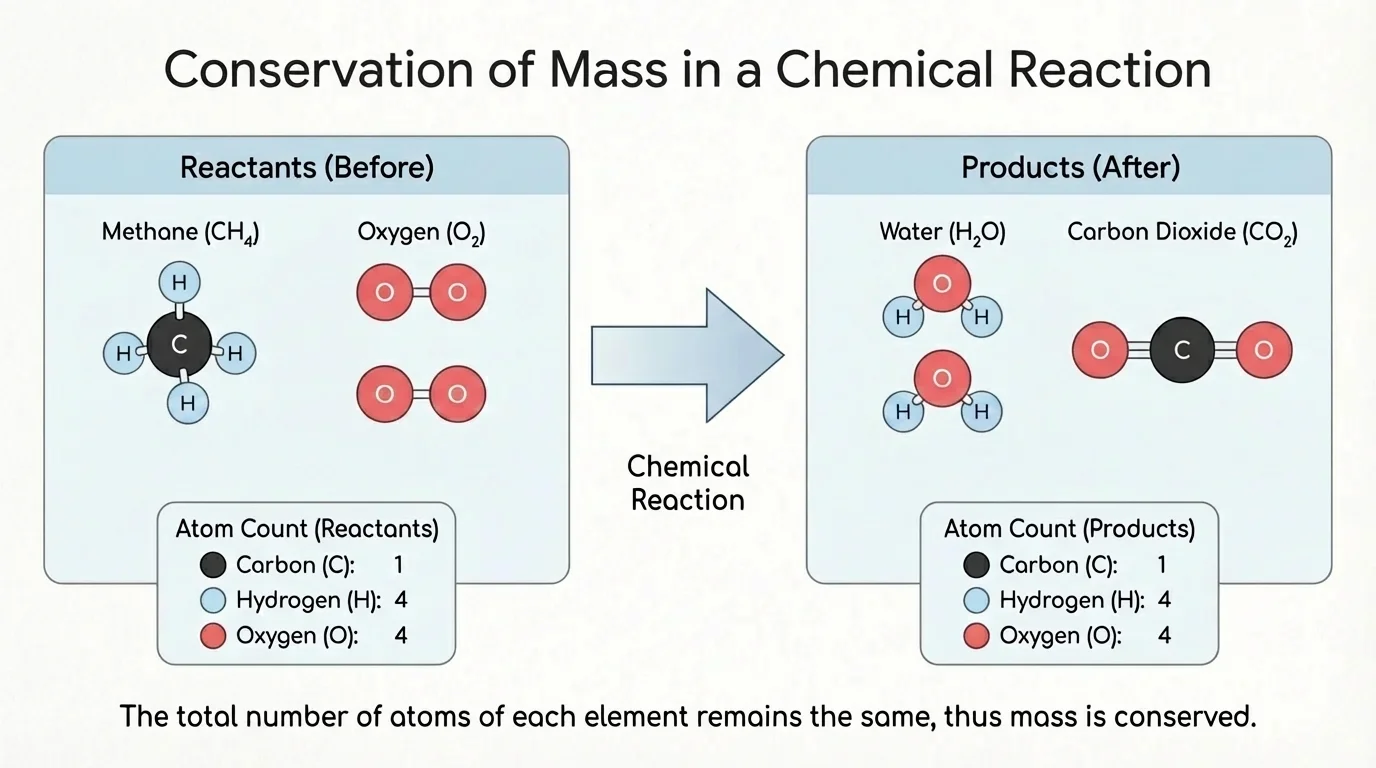
Notice that we are not focusing on balancing symbolic equations here. Instead, we are using a model and counting atoms to show conservation. This is exactly the level of thinking scientists use first: identify the particles, track them, and compare totals.
Here is another example in words. Iron can react with oxygen to form rust. The rust looks very different from shiny iron metal, but the iron atoms are still there. Oxygen atoms from the air join with the iron atoms to make a new substance. The total number of iron atoms and oxygen atoms is conserved.
| Substance | Atoms in one particle or unit |
|---|---|
| Water, \(\textrm{H}_2\textrm{O}\) | \(2\) hydrogen, \(1\) oxygen |
| Carbon dioxide, \(\textrm{CO}_2\) | \(1\) carbon, \(2\) oxygen |
| Oxygen gas, \(\textrm{O}_2\) | \(2\) oxygen |
| Methane, \(\textrm{CH}_4\) | \(1\) carbon, \(4\) hydrogen |
Table 1. Examples of common substances and how many atoms each particle contains.
Using a table like this helps you build and check models carefully. The exact appearance of the particles may change, but the count of atoms of each element must remain constant.
The idea that atoms are conserved leads directly to conservation of mass. If the same atoms are present before and after a reaction, then the total mass stays the same, as long as no matter escapes and no extra matter enters.
You can think of it this way: each atom contributes to the total mass. If none of the atoms disappear and no new ones appear, the total mass cannot suddenly become larger or smaller just because the atoms have been rearranged into new molecules.
Why mass stays the same
A chemical reaction changes the way atoms are connected, not the total amount of matter present. Because the same atoms are still in the system, the total mass before the reaction equals the total mass after the reaction.
This is true even when the products look very different from the reactants. A fizzing reaction might produce bubbles, a flame might produce hot gases, and a colorless gas might form from a solid. The look changes; the matter does not vanish.
Sometimes people think mass was not conserved because they only measured part of the system. As [Figure 3] shows, an open container can let gases escape into the air or allow gases from the air to enter. If you do not include those gases, it may look as if mass changed.
For example, when a fizzy tablet reacts in an open cup, bubbles of gas leave the cup and float away. If you weigh only the cup and what remains in it, the measured mass may go down. But the gas still exists. It is just no longer in the cup. If you could collect all the gas too, the total mass would match the starting mass.
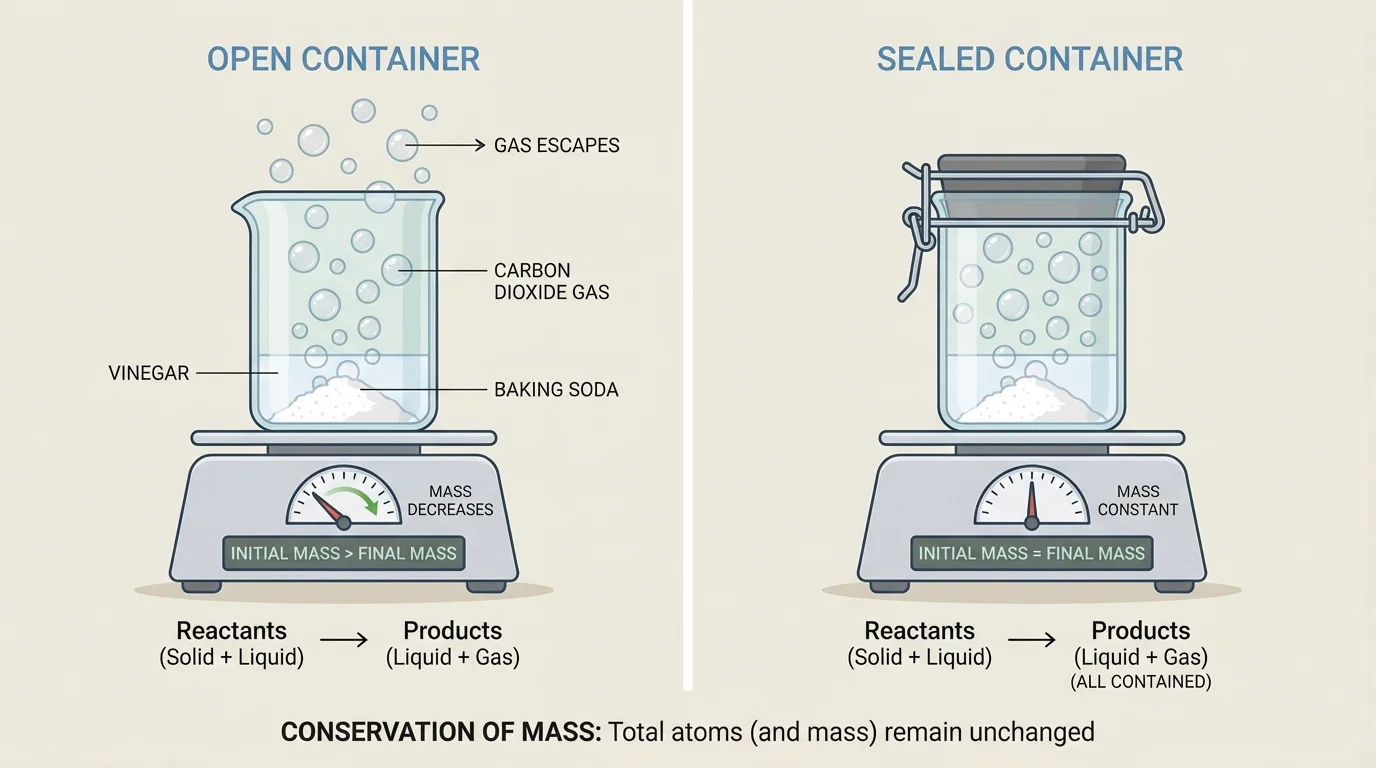
Burning can cause similar confusion. A candle seems to get smaller as it burns, but wax is reacting with oxygen in the air to form gases such as carbon dioxide and water vapor. Much of the matter leaves as gas, so the candle alone loses mass. If you include the surrounding air and all the products, mass is conserved.
Rusting shows the opposite effect. A piece of iron can gain mass as it rusts because oxygen atoms from the air join the iron. If you measured only the iron object, it seems to gain matter. But the added mass came from oxygen in the air, so the total matter in the larger system is still conserved.
A sealed container is often the best way to test conservation of mass in the classroom because it keeps gases from escaping. That makes the before-and-after masses easier to compare fairly.
Later, when we think back to [Figure 3], the big lesson is that the choice of system matters. Science measurements are only meaningful when you know exactly what is included.
Chemical reactions do more than rearrange atoms. They can also involve energy changes. Some reactions release energy to the surroundings. Others absorb energy from the surroundings.
A campfire, a candle flame, and many fuel reactions release energy as heat and light. Some cold packs used in sports absorb energy from their surroundings, which makes them feel cold. Even though energy changes occur, the atoms are still conserved.
"In a chemical reaction, atoms are rearranged, not destroyed."
This is an important idea because students sometimes mix up conservation of matter with energy changes. Matter and energy are related in science, but for the reactions you study at this level, the key point is simple: the number of atoms stays the same, and the total mass is conserved when the system includes all the matter.
The model of rearranging atoms still works whether a reaction feels hot, cold, bright, slow, or fast. Energy change does not mean atoms disappear.
Understanding conserved atoms helps scientists and engineers in many real situations. In car engines, fuel reacts with oxygen to form gases. Engineers must know what substances go in and what substances come out to design efficient engines and reduce pollution.
Environmental scientists also use this idea when tracking carbon through the atmosphere, oceans, plants, and animals. Carbon atoms move from one substance to another, but they do not vanish. When fuels burn, carbon atoms in the fuel often become carbon dioxide in the air.
In cooking, browning bread or frying an egg involves chemical reactions that create new substances with new smells, flavors, and colors. The atoms are rearranged into new molecules. In batteries, chemical reactions move matter and energy in useful ways to provide electrical power.
The particle picture from [Figure 1] is not just a classroom idea. Scientists use models of particles every day to understand reactions in medicine, industry, climate science, and materials engineering.
A classic way to test atom conservation is to compare a gas-producing reaction in an open setup and a closed setup. As [Figure 4] illustrates, vinegar and baking soda can react to make a gas. If the gas is trapped, the system keeps all of its matter.
In a closed setup, students can place baking soda in a bottle and vinegar in a balloon or separate area, then combine them without letting gas escape. The balloon inflates as gas forms, but the total mass of the sealed system stays the same. This gives strong evidence that the particles were rearranged, not lost.
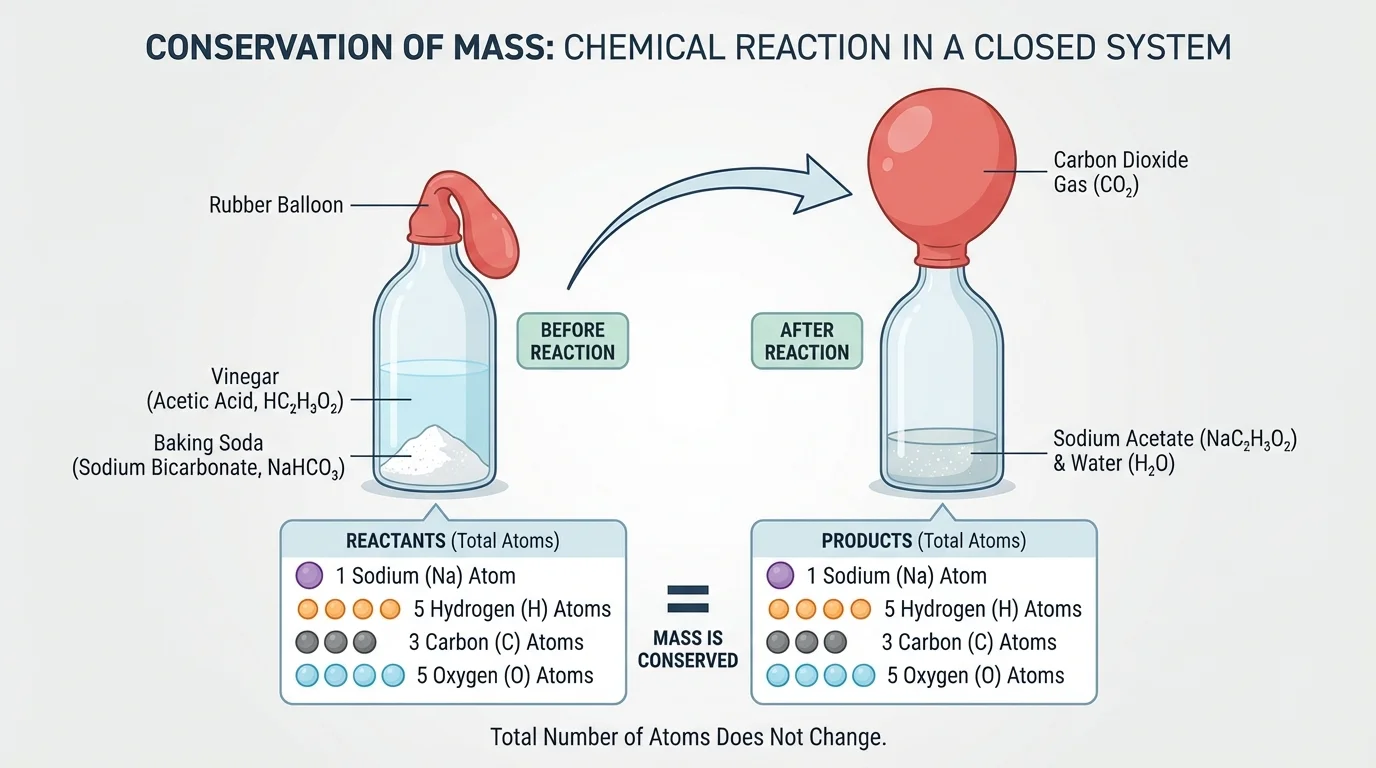
In an open setup, the gas escapes into the room. If students weigh only the cup or bottle afterward, it may seem as if mass decreased. The difference between the two setups helps make the idea of a system very concrete.
What the investigation shows
Step 1: Start with all reactants in a sealed system.
The bottle, vinegar, baking soda, and trapped air are all counted as part of the system.
Step 2: Let the reaction occur.
Bubbles form because a gas is produced, but it stays inside the balloon and bottle.
Step 3: Compare the masses.
The mass before and after is the same because the matter stayed inside the system.
This supports the idea that the total number of atoms does not change in the reaction.
When you connect that investigation to [Figure 4], it becomes easier to explain why bubbling does not mean matter is disappearing. The gas is still matter, even when it spreads out and becomes harder to see.
One misunderstanding is thinking that atoms are destroyed during burning. In ordinary chemical reactions, they are not. They are rearranged into new molecules.
Another misunderstanding is confusing chemical change with physical change. If ice melts, the water molecules are still water molecules, so that is a physical change. If hydrogen reacts with oxygen to make water, the particles change into a new substance, so that is a chemical reaction.
A third misunderstanding is assuming that if something cannot be seen, it no longer exists. Many products of reactions are gases, and gases can spread out quickly. Invisible does not mean gone.
It is also important not to confuse dissolving with reacting. When sugar dissolves in water, the sugar particles spread out, but they remain sugar. In a chemical reaction, new substances form.