Imagine you could shrink down smaller than a grain of sand, smaller than a speck of dust, smaller even than a cell in your body. 🔬 At that tiny scale, you would see that everything around you is built from unbelievably small building blocks called atoms. These atoms combine in many ways to make all the different substances in the universe—from the air you breathe to the metal in your phone.
Matter is anything that has mass and takes up space. Your desk, water, air, your own body—these are all forms of matter. All matter is made from atoms.
An atom is the smallest piece of an element that still has the properties of that element. There are a little over 100 different kinds of atoms, called elements. Each element has its own type of atom.
Atoms are extremely small. Millions of atoms can fit across the width of a single human hair. Yet the way these atoms are arranged and combined decides what substance you get.
The idea that everything is made of atoms helps explain why substances have different properties, like hardness, color, melting point, or whether they dissolve in water.
The basic structure of an atom, with a central nucleus and electrons around it, is shown in [Figure 1].
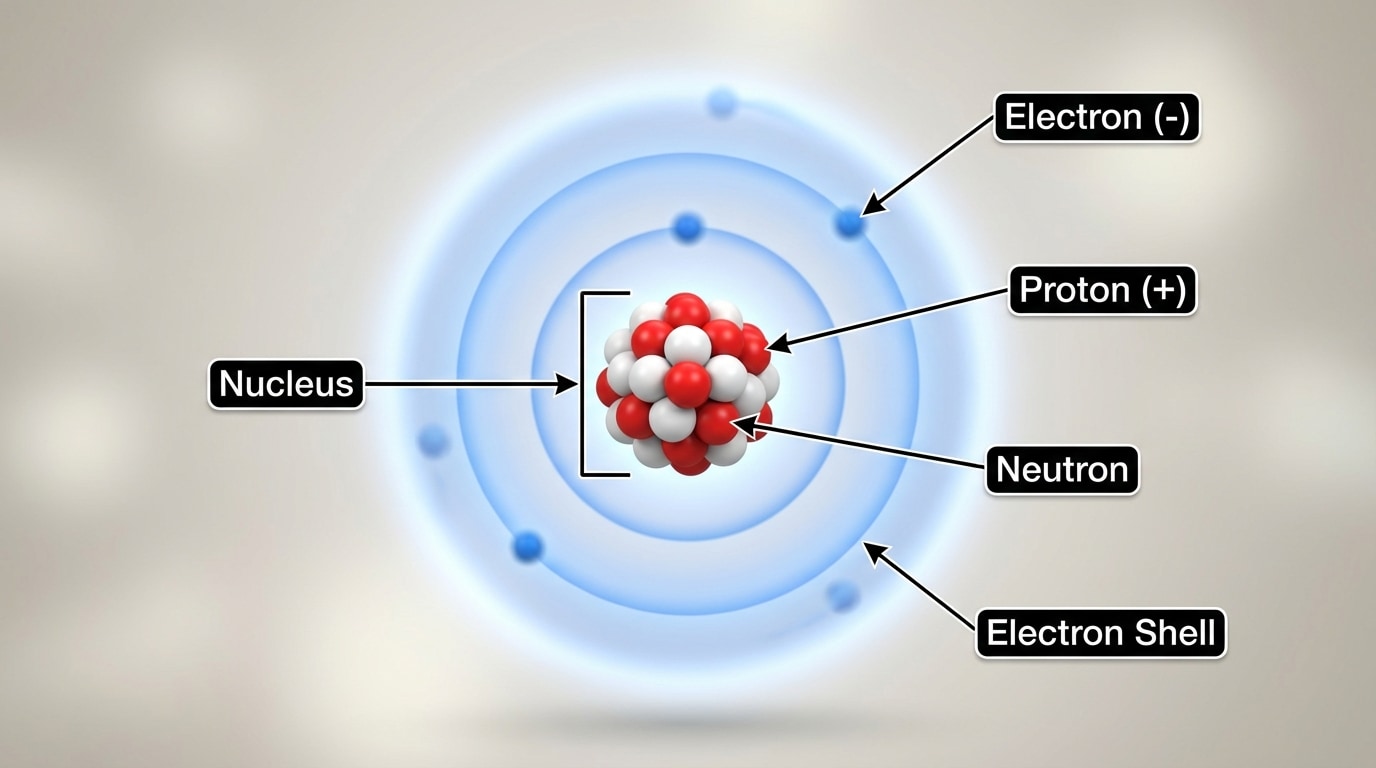
Each type of atom is identified by the number of protons in its nucleus. This number is called the atomic number.
Atoms of the same element all have the same number of protons. So every oxygen atom has 8 protons. Atoms with different numbers of protons are different elements.
The elements are shown on the periodic table, which is like a map of all the kinds of atoms that exist in nature (plus some that scientists have created in labs).
Did you know? ⭐ The oxygen in the air you breathe and the oxygen in the water you drink are made of exactly the same kind of oxygen atoms.
Atoms rarely stay alone. They tend to connect to other atoms. When they do, they form molecules or extended structures.
A molecule is a group of two or more atoms joined together by chemical bonds. These atoms can be the same kind or different kinds.
Molecules can be very simple, like \(O_2\), or extremely complex, like DNA, which stores your genetic information and contains billions of atoms in a single long molecule.
[Figure 2] illustrates how atoms can combine to form simple molecules like water and more complex solid structures like salt crystals.
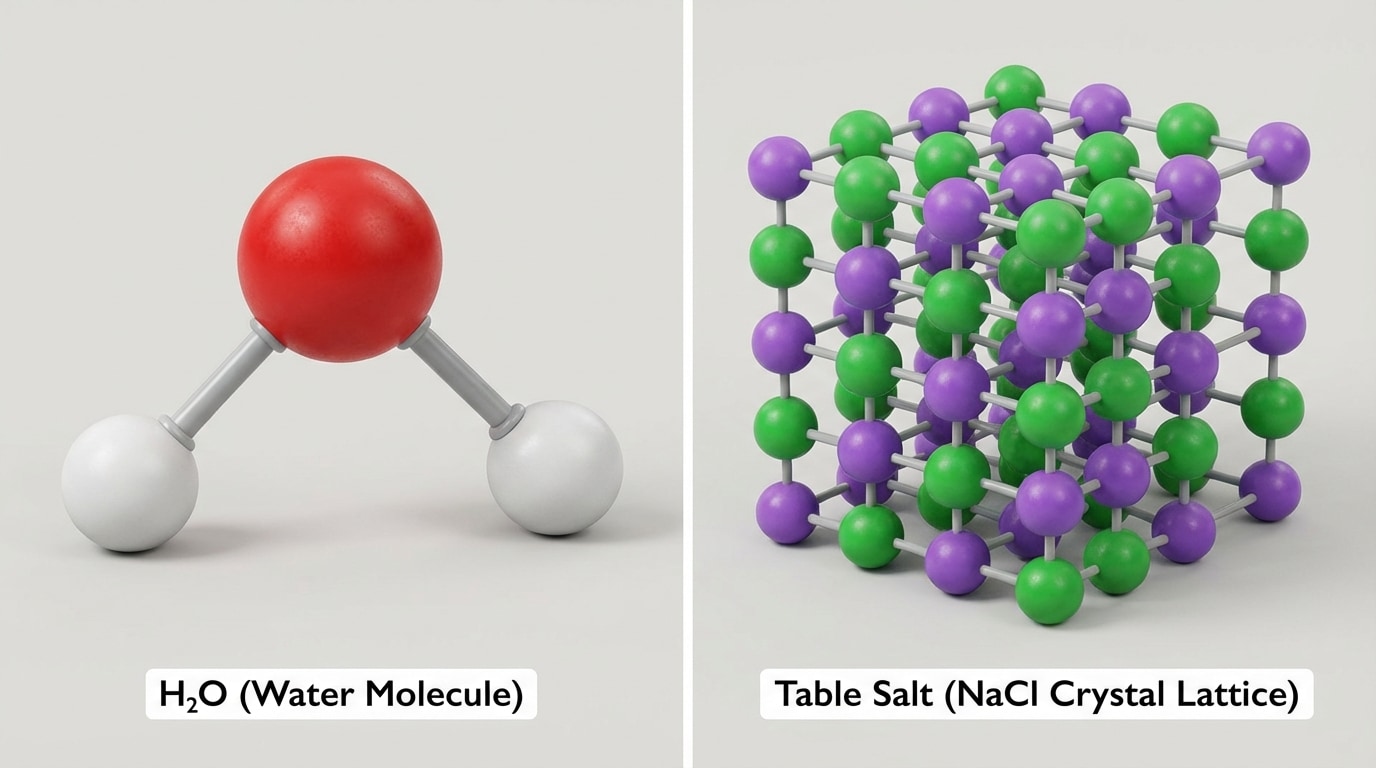
Sometimes atoms or ions do not form separate molecules. Instead, they build giant, repeating patterns called extended structures.
Crystals are a good example. In crystals, atoms or ions repeat in a regular 3D pattern. This gives crystals their flat faces and sharp edges.
These extended structures can contain enormous numbers of atoms, all arranged in a regular pattern. This pattern strongly affects the substance’s properties, such as hardness, melting point, and how it breaks.
A pure substance is matter that is made of only one kind of particle (all the particles are the same). It can be:
Each pure substance has its own characteristic physical and chemical properties that are the same for any bulk quantity under the same conditions (like temperature and pressure).
Physical properties can be observed or measured without changing the substance into something new.
For example:
Because these properties are consistent, scientists can use them to identify an unknown pure substance. For instance, if a liquid boils at 100 °C, freezes at 0 °C, and has a density of about 1 \(\textrm{g/cm}^3\), it is very likely water.
Chemical properties describe how a substance reacts and what new substances it can form.
For example:
These chemical properties help identify substances and predict how they will behave in chemical reactions.
The atoms or molecules in a substance can be arranged and move in different ways, depending on the state of matter (solid, liquid, or gas).
In a solid:
Solids may be made of:
This close, orderly arrangement explains why most solids are hard and difficult to compress.
In a liquid:
Examples: water, oil, milk. In liquids, the particles move more freely than in solids, which is why liquids can flow and be poured.
In a gas:
Air is a mixture of gases, mostly nitrogen and oxygen. Helium in a balloon is another example. Because gas particles are so far apart, gases are easy to compress.
[Figure 3] compares how particles are arranged and move in solids, liquids, and gases.
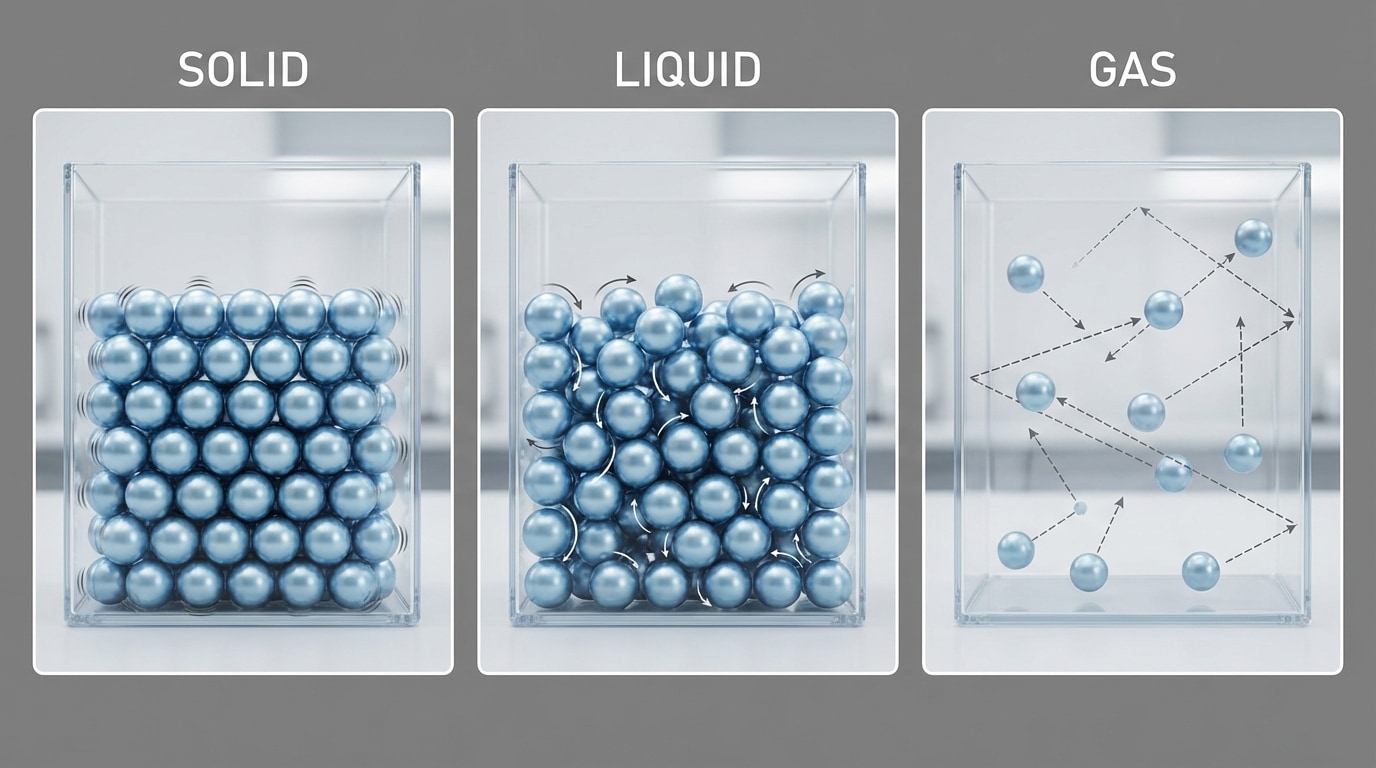
The state of matter depends on how strongly particles attract each other and how much they move. Their motion is related to temperature.
When temperature or pressure changes, substances can switch between solid, liquid, and gas. These are called changes of state or phase changes.
Example: Ice (solid water) melts into liquid water at 0 °C. Liquid water freezes into ice at the same temperature.
Example: Water boils at about 100 °C at normal pressure. Steam (water vapor) from a hot shower condenses on a cold mirror as liquid droplets.
These changes of state can be predicted and described using particle models of matter: how close particles are, how they move, and how strong their attractions are.
Although many middle school models focus on temperature, pressure is also important for gases. Pressure in a gas comes from particles colliding with the walls of their container.
This explains things like why a basketball feels harder when it is pumped full of air, or why a spray can warns you not to heat it.
Smartphones and computers rely on understanding the structure of matter. The materials inside microchips are carefully chosen crystals (like silicon) whose properties depend on how atoms are arranged. Tiny changes in atomic structure can turn a material from an insulator into a conductor.
Batteries in your devices work because of chemical reactions that involve atoms and ions moving from one material to another. Their performance depends on the chemical properties of the substances used.
When you pump air into a bicycle tire or a basketball, you are increasing the number of gas molecules in a fixed space. They collide more often with the walls, increasing pressure so the tire or ball becomes firm.
Car engines burn fuel (a chemical reaction) where molecules of gasoline react with oxygen molecules in the air, forming new molecules (mainly carbon dioxide and water) and releasing energy.
Cooking is full of changes of state and chemical reactions:
Weather also depends on changes of state:
Experiment 1: Observing Melting and Freezing
This shows that during melting, the temperature stays nearly constant while the state changes.
Experiment 2: Condensation on a Cold Surface
This water comes from water vapor in the air that cools and condenses on the cold glass.
Experiment 3: Gas Takes Up Space 🤔
Atoms are the tiny building blocks of matter, and there are different types (elements). Atoms combine to form molecules or extended structures. Solids, liquids, and gases differ in how closely packed the particles are and how they move: solids have closely packed particles that vibrate in place, liquids have particles that stay close but slide past each other, and gases have widely spaced, fast-moving particles that only interact when they collide. Each pure substance has characteristic physical and chemical properties, like melting point, boiling point, density, and reactivity, that can be used to identify it. Changes of state—melting, freezing, evaporation, boiling, condensation, sublimation, and deposition—happen when temperature or pressure changes, and they can be described and predicted using particle models of matter. These ideas help explain and predict what happens in everyday life, from boiling water and inflating tires to the way smartphones and batteries work. 🌍