A chemical reaction can look dramatic: a metal burns, a gas bubbles out of solution, or a clear liquid suddenly turns cloudy. It may seem as if matter is appearing or disappearing. Yet one of the most powerful ideas in chemistry is that matter is not lost in ordinary chemical reactions. The atoms are simply rearranged into new combinations. When we use mathematics to count atoms and compare masses, the hidden order of the reaction becomes clear.
Chemistry is full of changes, but not all parts of a substance change equally. Bonds break, new bonds form, and particles collide and reorganize. What stays constant is the total number of each kind of atom. Because atoms are conserved, total mass is also conserved. This is why chemists rely on equations, coefficients, and mass calculations to test whether a reaction description makes sense.
In a chemical reaction, substances called reactants change into substances called products. As [Figure 1] illustrates, the atoms in the reactants are not destroyed and new atoms are not created. Instead, existing atoms separate from old partners and join new ones. If a reaction starts with a certain number of hydrogen, oxygen, carbon, or iron atoms, it must end with those same numbers of those same atoms.
This idea is called the law of conservation of mass. In ordinary chemical reactions, the total mass before the reaction equals the total mass after the reaction. Since mass comes from the matter present, and matter is made of atoms, the conservation of mass follows from conservation of atoms.
Conservation of mass means that in a chemical reaction, the total mass of the reactants equals the total mass of the products.
Conservation of atoms means that the number of atoms of each element stays the same before and after a chemical reaction.
A simple reaction shows this clearly. Hydrogen gas reacts with oxygen gas to form water:
\[2\textrm{H}_2 + \textrm{O}_2 \rightarrow 2\textrm{H}_2\textrm{O}\]
On the left side, the number of atoms equals the coefficient multiplied by the subscript count. For hydrogen, there are \(2 \times 2 = 4\) hydrogen atoms. For oxygen, there are \(1 \times 2 = 2\) oxygen atoms. On the right side, the coefficient \(2\) in front of water means there are \(2 \times 2 = 4\) hydrogen atoms and \(2 \times 1 = 2\) oxygen atoms. The counts match exactly.
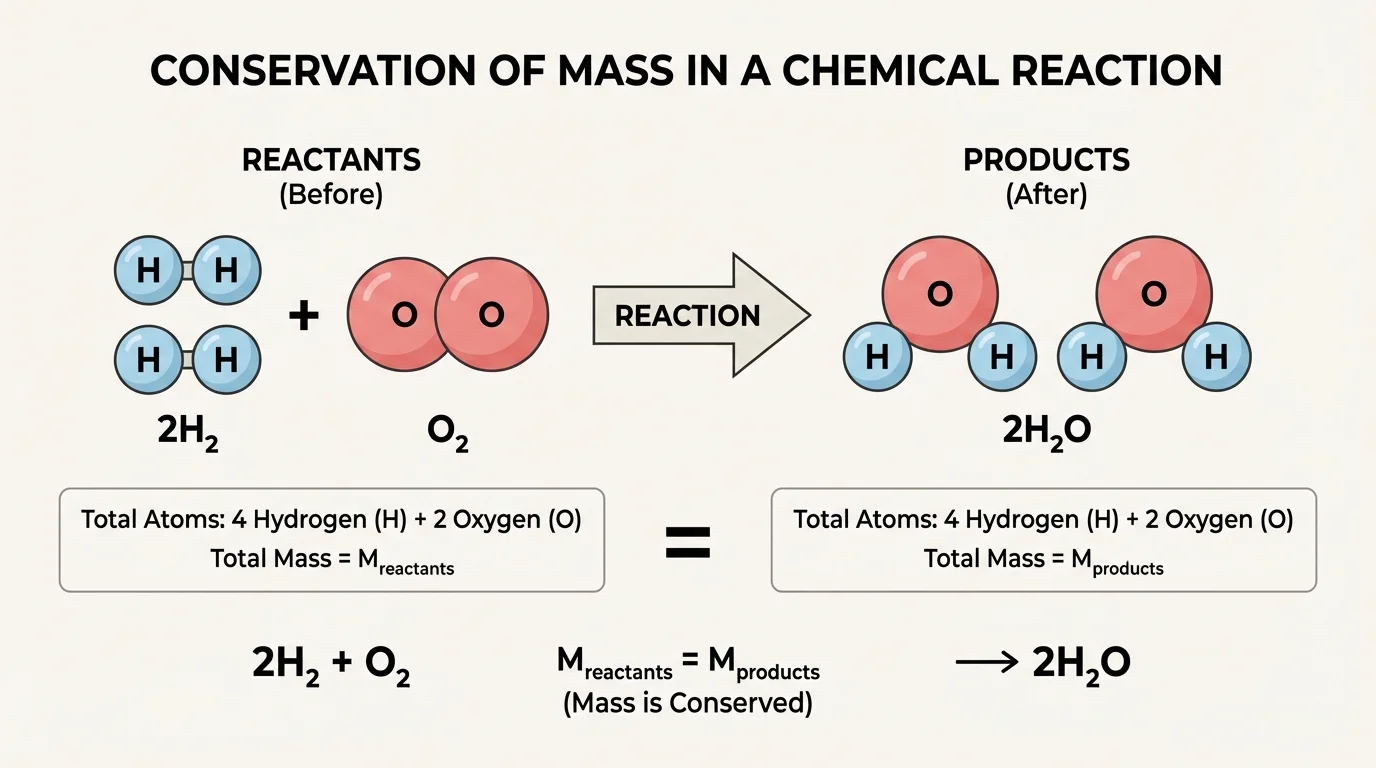
The reaction may look different at the particle level before and after, but the count of atoms is unchanged. That is the core claim that mathematical representations support. We use numbers, formulas, and equations to prove that what appears to be a transformation is really a rearrangement.
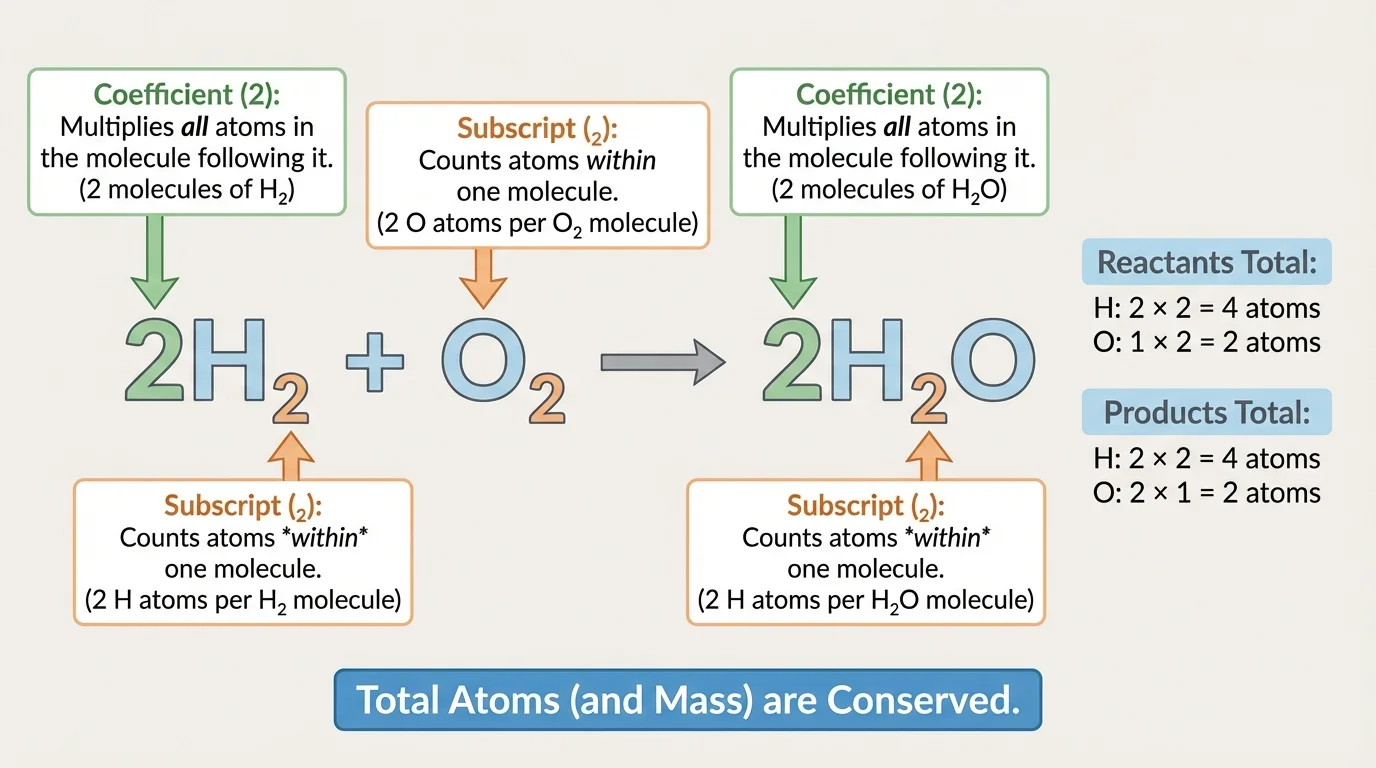
A chemical equation is more than a sentence about substances reacting. As [Figure 2] shows, it is also a mathematical counting statement. The formulas tell which atoms are present, the subscripts tell how many of each atom are in one particle, and the coefficients tell how many particles are involved.
Students often confuse coefficients and subscripts. A subscript belongs to the formula itself. In \(\textrm{H}_2\textrm{O}\), the subscript \(2\) means one water molecule contains \(2\) hydrogen atoms. A coefficient sits in front of the formula and multiplies the entire formula. In \(3\textrm{H}_2\textrm{O}\), the coefficient \(3\) means there are \(3\) water molecules, so the total number of hydrogen atoms is \(3 \times 2 = 6\), and the total number of oxygen atoms is \(3 \times 1 = 3\).
Consider the equation
\[2\textrm{Na} + \textrm{Cl}_2 \rightarrow 2\textrm{NaCl}\]
On the reactant side, sodium atoms total \(2\), and chlorine atoms total \(2\). On the product side, the coefficient \(2\) in front of \(\textrm{NaCl}\) means there are \(2\) sodium atoms and \(2\) chlorine atoms. The equation is balanced because each element has the same count on both sides.
This is why changing a subscript to balance an equation is not allowed. If you changed \(\textrm{H}_2\textrm{O}\) to \(\textrm{H}_2\textrm{O}_2\), you would be changing the identity of the substance, not just the number of particles. Balancing means adjusting coefficients only, so that the reaction represents the same substances while preserving atom counts.
From earlier chemistry work, remember that element symbols represent types of atoms, formulas represent substances, and subscripts are part of a substance's identity. Balancing an equation never changes the formula of a substance.
To balance an equation means to choose coefficients so that the number of atoms of each element is equal on both sides. As [Figure 3] illustrates, this process is like solving a counting puzzle. You compare totals, adjust coefficients, and check again until all atom counts match.
Take the unbalanced equation for forming water:
\[\textrm{H}_2 + \textrm{O}_2 \rightarrow \textrm{H}_2\textrm{O}\]
Hydrogen is balanced at first glance because there are \(2\) hydrogen atoms on both sides. Oxygen is not balanced: there are \(2\) oxygen atoms on the left but only \(1\) on the right. Put a coefficient of \(2\) before water:
\[\textrm{H}_2 + \textrm{O}_2 \rightarrow 2\textrm{H}_2\textrm{O}\]
Now oxygen is balanced because the right side has \(2 \times 1 = 2\) oxygen atoms, but hydrogen is no longer balanced because the right side has \(2 \times 2 = 4\) hydrogen atoms. Put a coefficient of \(2\) before hydrogen gas:
\[2\textrm{H}_2 + \textrm{O}_2 \rightarrow 2\textrm{H}_2\textrm{O}\]
Now hydrogen atoms are \(4\) on both sides, and oxygen atoms are \(2\) on both sides. The equation is balanced.
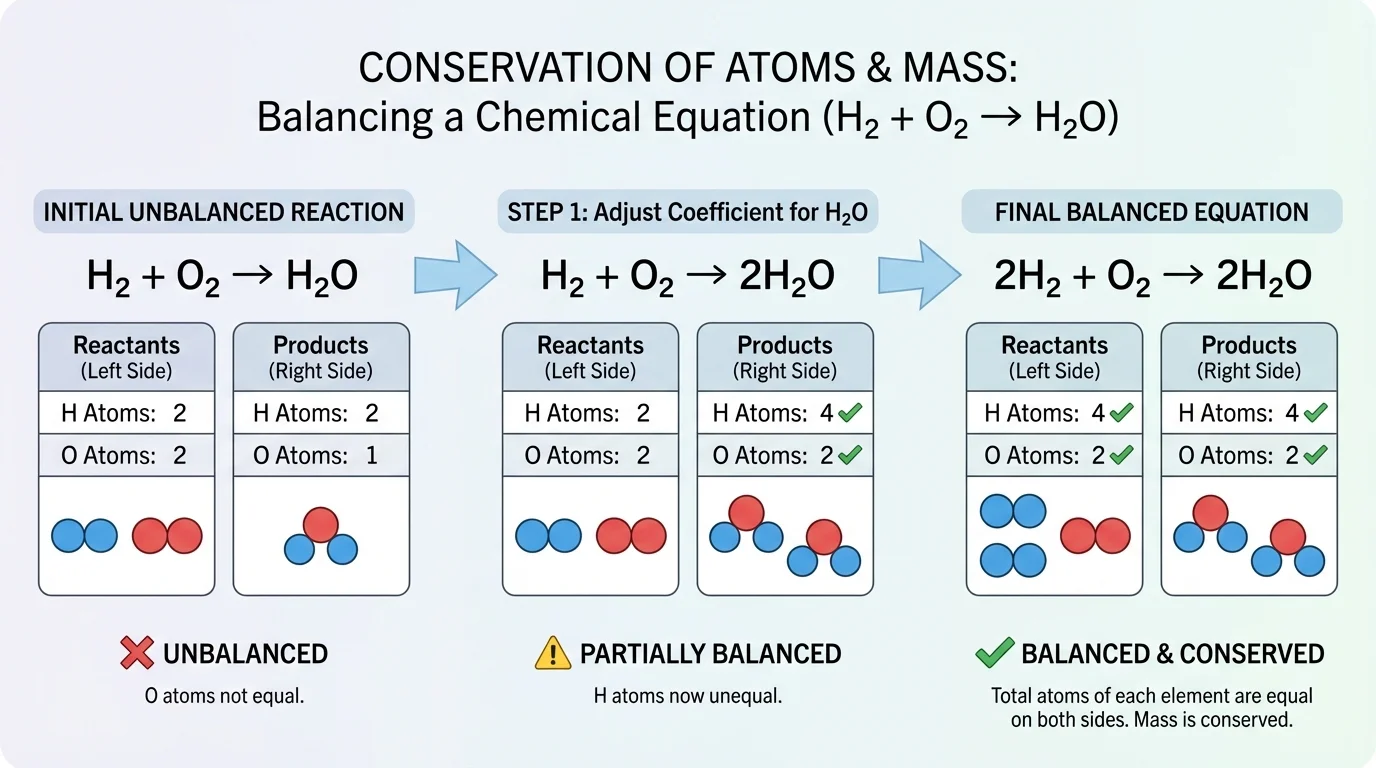
A useful strategy is to count one element at a time and leave any element that appears in its pure form, such as \(\textrm{O}_2\) or \(\textrm{H}_2\), for later if possible. Another strategy is to start with the compound that contains the most different elements. For this topic, simple reactions are enough to show the principle clearly.
Why balancing works
Balancing does not force nature to conserve atoms; it reveals that nature already does. A correct balanced equation is a mathematical model of what happens in the reaction: the same atoms appear before and after, only rearranged into different substances.
Atom counts show that the same atoms are present before and after a reaction. We can go further and use mass values to show that the total mass also stays constant. Chemists use relative atomic mass values from the periodic table to calculate the mass of reactants and products.
For example, in the balanced equation
\[2\textrm{H}_2 + \textrm{O}_2 \rightarrow 2\textrm{H}_2\textrm{O}\]
take the approximate atomic masses of hydrogen to be \(1\) and oxygen to be \(16\). One molecule of \(\textrm{H}_2\) has mass \(2\), so \(2\textrm{H}_2\) has total mass \(2 \times 2 = 4\). One molecule of \(\textrm{O}_2\) has mass \(2 \times 16 = 32\). Total reactant mass is \(4 + 32 = 36\).
On the product side, one molecule of water has mass \((2 \times 1) + 16 = 18\). Since there are \(2\) water molecules, total product mass is \(2 \times 18 = 36\). The masses are equal, so the calculation supports conservation of mass.
| Substance | How mass is found | Total mass |
|---|---|---|
| \(2\textrm{H}_2\) | \(2 \times (2 \times 1)\) | \(4\) |
| \(\textrm{O}_2\) | \(2 \times 16\) | \(32\) |
| Total reactants | \(4 + 32\) | \(36\) |
| \(2\textrm{H}_2\textrm{O}\) | \(2 \times ((2 \times 1) + 16)\) | \(36\) |
Table 1. Mass comparison for hydrogen reacting with oxygen to form water.
These masses are based on relative atomic masses rather than the mass of one single atom measured directly on a balance, but the relationship is the same. If atom counts are conserved, and each kind of atom keeps its own mass, then total mass must remain constant.
The best way to see the connection between atom conservation and mass conservation is to work through complete examples carefully.
Worked example 1: Water formation
Show that atoms and mass are conserved in \(2\textrm{H}_2 + \textrm{O}_2 \rightarrow 2\textrm{H}_2\textrm{O}\).
Step 1: Count atoms in the reactants.
Hydrogen atoms: \(2 \times 2 = 4\)
Oxygen atoms: \(1 \times 2 = 2\)
Step 2: Count atoms in the products.
Hydrogen atoms: \(2 \times 2 = 4\)
Oxygen atoms: \(2 \times 1 = 2\)
Step 3: Calculate total mass on each side using hydrogen \(= 1\) and oxygen \(= 16\).
Reactants: \((2 \times 2) + (1 \times 32) = 4 + 32 = 36\)
Products: \(2 \times 18 = 36\)
Both atom counts and total mass are equal, so the equation supports the claim.
This example is simple, but it contains the full logic of conservation. Count atoms first. Then compare masses. If the equation is balanced, the mass totals match.
Worked example 2: Methane combustion
Show conservation in \(\textrm{CH}_4 + 2\textrm{O}_2 \rightarrow \textrm{CO}_2 + 2\textrm{H}_2\textrm{O}\).
Step 1: Count atoms in the reactants.
Carbon: \(1\)
Hydrogen: \(4\)
Oxygen: \(2 \times 2 = 4\)
Step 2: Count atoms in the products.
From \(\textrm{CO}_2\): carbon \(= 1\), oxygen \(= 2\)
From \(2\textrm{H}_2\textrm{O}\): hydrogen \(= 2 \times 2 = 4\), oxygen \(= 2 \times 1 = 2\)
Total oxygen in products: \(2 + 2 = 4\)
Step 3: Compare masses using carbon \(= 12\), hydrogen \(= 1\), oxygen \(= 16\).
Reactants: methane mass \(= 12 + 4 = 16\); oxygen mass \(= 2 \times 32 = 64\)
Total reactants: \(16 + 64 = 80\)
Products: carbon dioxide mass \(= 12 + 32 = 44\); water mass \(= 2 \times 18 = 36\)
Total products: \(44 + 36 = 80\)
The total mass is \(80\) mass units on both sides, and every atom count matches.
Combustion in engines and burners follows this same principle. Fuel does not vanish; its atoms become part of new substances such as carbon dioxide and water.
Worked example 3: Decomposition of hydrogen peroxide
Show conservation in \(2\textrm{H}_2\textrm{O}_2 \rightarrow 2\textrm{H}_2\textrm{O} + \textrm{O}_2\).
Step 1: Count atoms in the reactants.
Hydrogen: \(2 \times 2 = 4\)
Oxygen: \(2 \times 2 = 4\)
Step 2: Count atoms in the products.
From \(2\textrm{H}_2\textrm{O}\): hydrogen \(= 4\), oxygen \(= 2\)
From \(\textrm{O}_2\): oxygen \(= 2\)
Total oxygen: \(2 + 2 = 4\)
Step 3: Compare masses using hydrogen \(= 1\) and oxygen \(= 16\).
One \(\textrm{H}_2\textrm{O}_2\) has mass \(2 + 32 = 34\), so \(2\textrm{H}_2\textrm{O}_2\) has mass \(68\).
Products: \(2\textrm{H}_2\textrm{O}\) has mass \(36\), and \(\textrm{O}_2\) has mass \(32\).
Total products: \(36 + 32 = 68\)
The reaction conserves both atoms and mass.
Hydrogen peroxide decomposition is useful because the production of oxygen gas can make mass seem to decrease if the gas escapes. That does not break the law of conservation of mass; it only affects what is measured.
Many students think mass is not conserved because they observe a reaction and the measured mass changes. As [Figure 4] shows, this usually happens because the reaction occurs in an open system, so gas can enter or leave the container. The missing or extra mass is not destroyed or created; it has simply moved.
For example, if a gas-producing reaction happens in an open flask, the flask may weigh less afterward because some product gas escapes into the air. If the same reaction occurs in a sealed container, the total measured mass remains the same. The chemistry is identical; only the measurement setup changes.
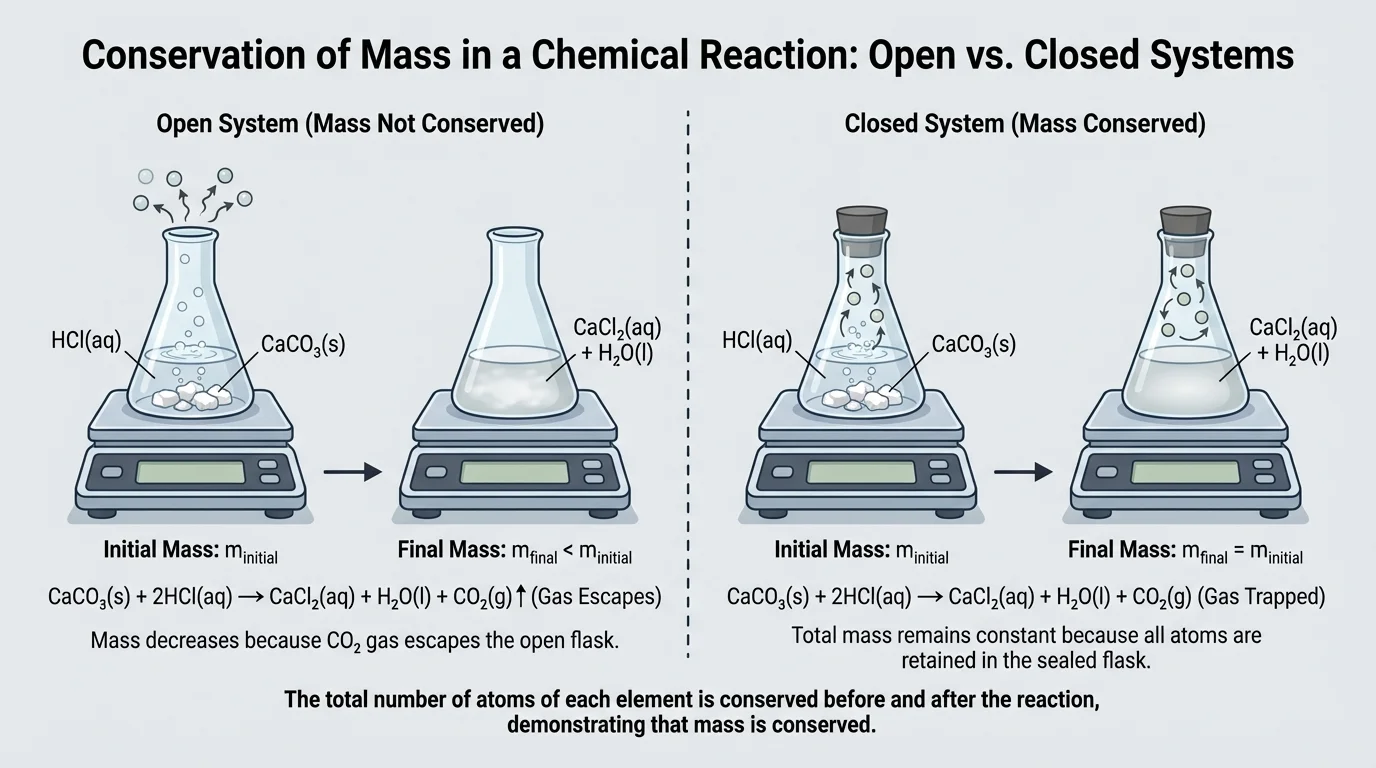
This matters in laboratory work. If a student mixes vinegar and baking soda in an open cup, the carbon dioxide gas can leave, so the measured mass of the cup and contents decreases. In a sealed bag or flask, the total mass before and after stays constant because all matter remains inside the system.
When metals rust, the mass of the metal object usually increases because oxygen atoms from the air combine with the metal. A nail that rusts has not created mass; it has gained oxygen from its surroundings.
That example is a useful reminder that the system boundary matters. The same conservation law explains both apparent mass loss and apparent mass gain. You must include all reactants and all products in the system.
Conservation of atoms and mass is not just a classroom rule. Engineers use it when designing engines, because fuel and oxygen enter the system and exhaust gases leave it. Environmental scientists use it when tracking pollutants, asking where atoms of carbon, nitrogen, or sulfur move during chemical changes. Industrial chemists use balanced equations to predict how much reactant is needed and how much product can form.
Medical technology also depends on this idea. In metabolism, atoms in food molecules are rearranged through many reactions into carbon dioxide, water, and other compounds. The body does not make atoms disappear. It transforms substances through reaction pathways that still obey the same atom-counting rules seen in simple equations such as those in [Figure 1] and [Figure 3].
Even air pollution measurements depend on conservation thinking. If carbon-containing fuel burns, the carbon atoms must end up somewhere, usually in carbon dioxide or, if combustion is incomplete, in carbon monoxide or soot. Scientists can trace those products because the atoms have been rearranged, not erased.
The strongest evidence for conservation in a chemical reaction comes from mathematical representations: balanced equations, atom counts, and mass calculations. A balanced equation is a claim that the same numbers of each kind of atom exist before and after the reaction. Mass calculations based on those same atoms confirm that the total mass is unchanged.
When a reaction seems to violate this principle, check the system. Ask whether a gas escaped, whether matter from the air entered, or whether the measurement included all substances. Chemistry can look messy in the lab or in daily life, but the underlying accounting is precise.
"Chemical reactions are not about making matter vanish; they are about rearranging matter into new forms."
That is why balancing equations matters so much. It is not a formal game with numbers. It is the mathematical expression of a physical truth about matter.