A remarkable fact about the universe is that the same kind of mathematical pattern helps describe both planets orbiting stars and tiny charged particles interacting inside matter. Gravity holds moons, planets, and galaxies together, while electric forces shape lightning, electronics, and the structure of atoms. These forces can act even when objects are not touching. That sounds strange at first, but physics explains this through fields.
In everyday life, many forces seem to require contact: a hand pushes a door, shoes push on the floor, and a bat hits a ball. But some forces act at a distance. A dropped object falls toward Earth because of gravity. A charged balloon can attract bits of paper because of electric force. These are examples of interactions that happen through space rather than through direct contact.
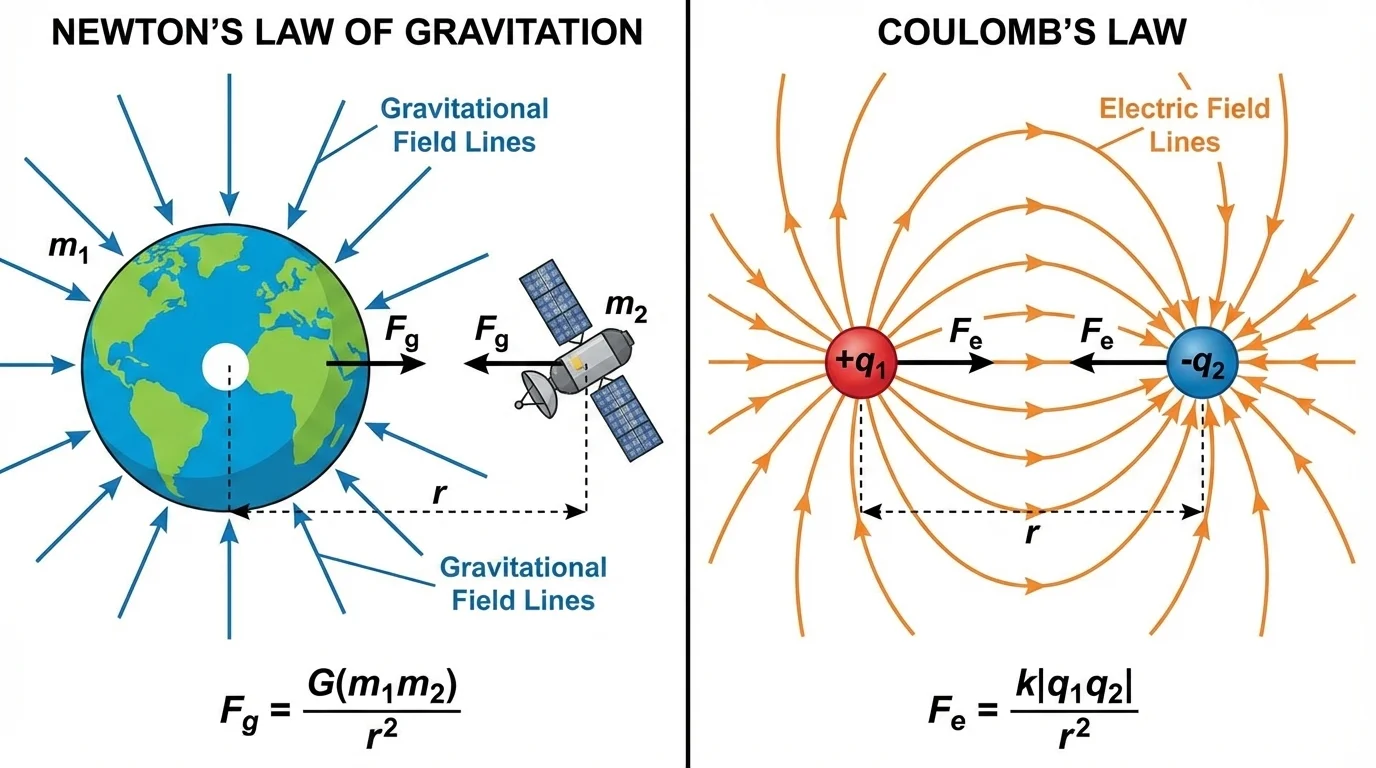
Scientists describe this idea using a field. As [Figure 1] shows, a field is a region around an object where another object can experience a force. A massive object creates a gravitational field. A charged object creates an electric field. The force depends on the properties of the interacting objects and on how far apart they are.
For this topic, we focus only on systems with two objects. That means every calculation and prediction will involve one object interacting with one other object. This keeps the math clear and matches the assessment boundary.
Thinking in terms of fields helps explain why these forces can transfer energy across space. A satellite gains or loses gravitational potential energy depending on its position relative to Earth. Charged particles can also gain or lose electric potential energy as they move in an electric field.
You already know that a force is an interaction that can change an object's motion. You also know that forces are measured in newtons, written as \(\textrm{N}\), and that forces have both magnitude and direction.
Gravity and electric force are not random. They follow precise mathematical laws, and those laws allow us to describe and predict how strongly two objects interact.
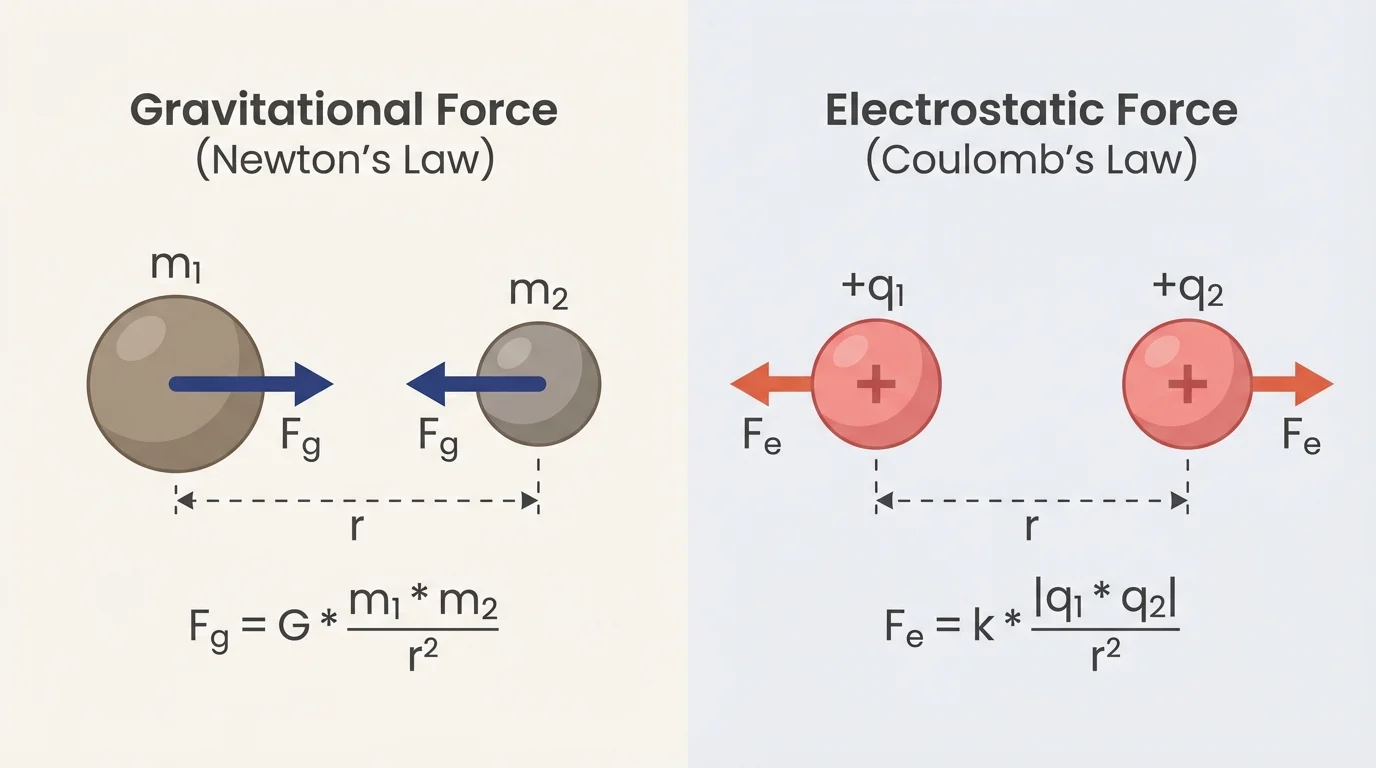
Newton's Law of Gravitation states that every pair of masses attracts each other with a force. The basic two-object geometry involves two masses, a center-to-center distance, and equal forces pulling each object toward the other.
The magnitude of the gravitational force is given by
\[F_g = G\frac{m_1m_2}{r^2}\]
In this equation, \(F_g\) is the gravitational force, \(G\) is the gravitational constant, \(m_1\) and \(m_2\) are the two masses, and \(r\) is the distance between their centers.
[Figure 2] helps visualize three major ideas in this equation. First, if either mass increases, the force increases. Second, if both masses increase, the force increases even more because the masses are multiplied together. Third, if distance increases, the force decreases according to an inverse-square law. That means distance matters a lot.
For example, if the distance between two objects doubles from \(r\) to \(2r\), the force becomes
\[F_g' = G\frac{m_1m_2}{(2r)^2} = G\frac{m_1m_2}{4r^2} = \frac{1}{4}F_g\]
So doubling the distance makes the gravitational force only one-fourth as large. If the distance triples, the force becomes \(\dfrac{1}{9}\) of the original value.
Gravity is always attractive. Two masses always pull toward each other. There is no gravitational repulsion at this level of physics. As shown earlier in [Figure 2], each object pulls on the other with equal magnitude and opposite direction.
Numerical example: gravitational force between two students
Suppose two students each have a mass of \(60 \textrm{ kg}\), and their centers of mass are about \(1.0 \textrm{ m}\) apart. Use \(G = 6.67 \times 10^{-11} \textrm{ N}\cdot\textrm{m}^2/\textrm{kg}^2\).
Step 1: Write the formula.
\(F_g = G\dfrac{m_1m_2}{r^2}\)
Step 2: Substitute the values.
\(F_g = 6.67 \times 10^{-11} \cdot \dfrac{(60)(60)}{(1.0)^2}\)
Step 3: Calculate.
\(60 \cdot 60 = 3600\), so \(F_g = 6.67 \times 10^{-11} \cdot 3600 = 2.4012 \times 10^{-7} \textrm{ N}\)
The force is approximately \(2.4 \times 10^{-7} \textrm{ N}\). This is extremely small, which is why we do not notice gravitational attraction between ordinary objects very easily.
Although the force between small everyday objects is tiny, gravity becomes enormous when at least one mass is very large, such as Earth, the Moon, or the Sun.
Coulomb's Law describes the electric force between two charged objects. The main interaction patterns include opposite charges attracting and like charges repelling.
The magnitude of the electrostatic force is given by
\[F_e = k\frac{|q_1q_2|}{r^2}\]
Here, \(F_e\) is the electric force, \(k\) is Coulomb's constant, \(q_1\) and \(q_2\) are the two charges, and \(r\) is the distance between them. The absolute value signs around \(q_1q_2\) mean that this equation gives the magnitude of the force.
[Figure 3] shows that, just as with gravity, the force increases if the size of either charge increases. And just as with gravity, increasing distance decreases the force by the square of the distance.
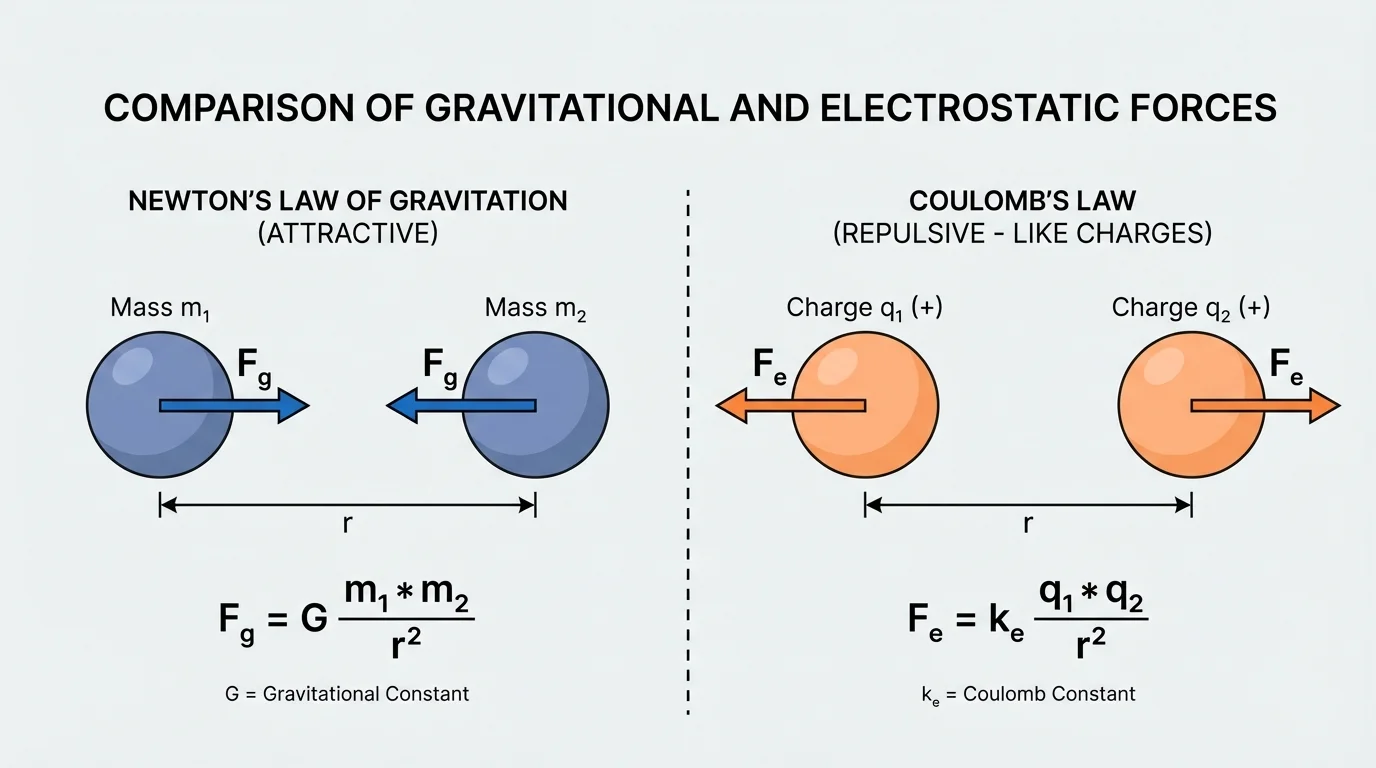
The key difference is direction. Electric force can be either attractive or repulsive. If the charges have opposite signs, such as \(+\) and \(-\), the force is attractive. If they have the same sign, such as \(+\) and \(+\) or \(-\) and \(-\), the force is repulsive. That directional behavior is one reason electric interactions are so important in matter.
For example, electrons and protons attract each other, but two electrons repel. This is consistent with the interaction patterns shown in [Figure 3].
Mass is a measure of the amount of matter in an object and determines how strongly it participates in gravitational interactions.
Charge is a property of matter that causes electric interactions. Charges can be positive or negative.
Attraction means two objects pull toward each other, while repulsion means they push away from each other.
Because electric forces can repel as well as attract, they can sometimes balance or overcome other forces in a system. This helps explain why matter has structure instead of collapsing under gravity at small scales.
Numerical example: electric force between two charges
Two small objects carry charges of \(2.0 \times 10^{-6} \textrm{ C}\) and \(3.0 \times 10^{-6} \textrm{ C}\). They are \(0.50 \textrm{ m}\) apart. Use \(k = 8.99 \times 10^9 \textrm{ N}\cdot\textrm{m}^2/\textrm{C}^2\).
Step 1: Write the formula.
\(F_e = k\dfrac{|q_1q_2|}{r^2}\)
Step 2: Substitute the values.
\(F_e = 8.99 \times 10^9 \cdot \dfrac{(2.0 \times 10^{-6})(3.0 \times 10^{-6})}{(0.50)^2}\)
Step 3: Multiply the charges.
\((2.0 \times 10^{-6})(3.0 \times 10^{-6}) = 6.0 \times 10^{-12}\)
Step 4: Square the distance and calculate.
\((0.50)^2 = 0.25\)
So \(F_e = 8.99 \times 10^9 \cdot \dfrac{6.0 \times 10^{-12}}{0.25} = 8.99 \times 10^9 \cdot 2.4 \times 10^{-11}\)
\(F_e = 0.21576 \textrm{ N}\)
The force is approximately \(0.22 \textrm{ N}\). If both charges are positive, the force is repulsive. If one is positive and the other is negative, the force is attractive.
Notice how much larger this electric force is than the earlier gravitational force between two students. Electric forces can be very strong even for tiny amounts of charge.
Newton's Law of Gravitation and Coulomb's Law have a strikingly similar mathematical form. Both depend on the product of two object properties and both decrease with the square of distance.
| Feature | Gravitational Force | Electric Force |
|---|---|---|
| Law | \(F_g = G\dfrac{m_1m_2}{r^2}\) | \(F_e = k\dfrac{|q_1q_2|}{r^2}\) |
| Object property | Mass | Charge |
| Distance effect | Inverse square | Inverse square |
| Direction | Always attractive | Attractive or repulsive |
| Relative strength in everyday small objects | Usually very weak | Can be strong |
Table 1. Comparison of the main features of gravitational and electrostatic force laws for two-object systems.
These similarities are not just mathematical coincidences. They reveal a deeper pattern in nature: the arrangement of objects and the distance between them strongly affect how forces are transferred through fields.
A small static electric shock after walking across carpet is caused by electric forces between charges that built up on your body and another object. The same basic law that describes that shock also helps describe how charged particles interact in matter.
There is also an important difference in signs. Mass is always positive in the situations studied here, so gravity only pulls. Charge can be positive or negative, so electric interactions can either pull together or push apart.
Many problems can be solved by reasoning from the equation instead of plugging in every number. This is often the fastest way to make predictions.
[Figure 4] shows that if one mass in the gravitational formula doubles while everything else stays the same, the force doubles. If both masses double, the force becomes four times as large because \((2m_1)(2m_2) = 4m_1m_2\).
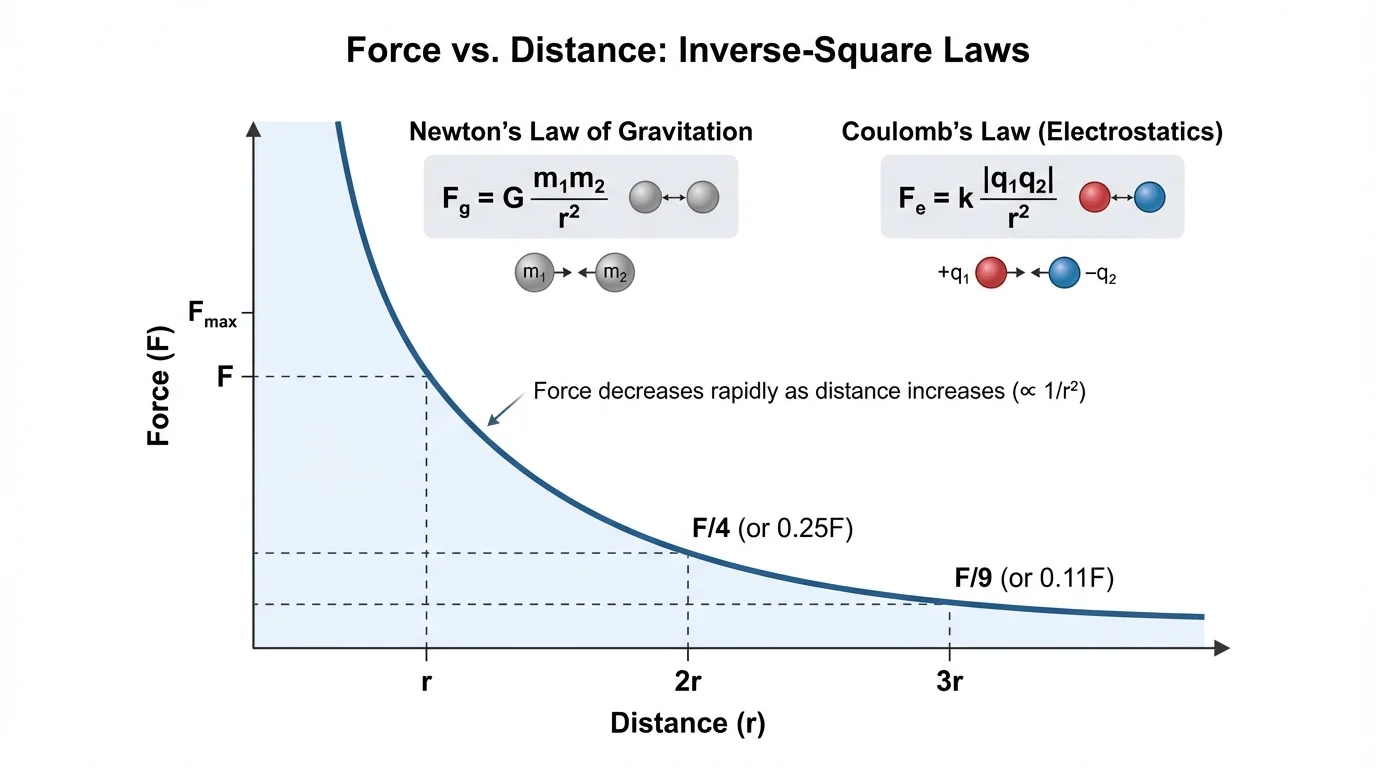
The same kind of reasoning works for charge. If one charge triples, the electric force triples. If the distance doubles, the force becomes one-fourth. If the distance is cut in half, the force becomes four times larger because
\[\frac{1}{(r/2)^2} = \frac{1}{r^2/4} = \frac{4}{r^2}\]
This inverse-square behavior is why small changes in distance can have big effects. The curve in [Figure 4] shows that the force drops quickly as objects move farther apart.
Prediction example without full calculation
A pair of charged objects exerts a force of \(12 \textrm{ N}\) when separated by distance \(r\). What happens if the distance becomes \(3r\) and the charges stay the same?
Step 1: Use the inverse-square idea.
When distance changes from \(r\) to \(3r\), the force changes by a factor of \(\dfrac{1}{3^2} = \dfrac{1}{9}\).
Step 2: Apply the factor.
\(F' = 12 \cdot \dfrac{1}{9} = \dfrac{12}{9} = 1.33 \textrm{ N}\)
The new force is \[F' \approx 1.3 \textrm{ N}\]
This style of reasoning is especially useful in science and engineering because it lets you estimate quickly before using a calculator.
Now consider another gravitational calculation involving an object near Earth. These calculations matter in orbital motion, satellite design, and space exploration.
Gravitational example: Earth and a satellite
A satellite of mass \(500 \textrm{ kg}\) is \(7.0 \times 10^6 \textrm{ m}\) from Earth's center. Earth's mass is \(5.97 \times 10^{24} \textrm{ kg}\). Find the gravitational force.
Step 1: Write the formula.
\(F_g = G\dfrac{m_1m_2}{r^2}\)
Step 2: Substitute the values.
\(F_g = 6.67 \times 10^{-11} \cdot \dfrac{(5.97 \times 10^{24})(500)}{(7.0 \times 10^6)^2}\)
Step 3: Simplify the numerator.
\((5.97 \times 10^{24})(500) = 2.985 \times 10^{27}\)
Step 4: Simplify the denominator.
\((7.0 \times 10^6)^2 = 4.9 \times 10^{13}\)
Step 5: Complete the calculation.
\(F_g = 6.67 \times 10^{-11} \cdot \dfrac{2.985 \times 10^{27}}{4.9 \times 10^{13}}\)
\(\dfrac{2.985 \times 10^{27}}{4.9 \times 10^{13}} \approx 6.09 \times 10^{13}\)
So \(F_g \approx 6.67 \times 10^{-11} \cdot 6.09 \times 10^{13} \approx 4.06 \times 10^3 \textrm{ N}\)
The gravitational force is \[F_g \approx 4.1 \times 10^3 \textrm{ N}\]
A similar approach works for electric-force problems, but direction depends on the signs of the charges.
Electrostatic example: identifying attraction or repulsion
Two charges are \(q_1 = -4.0 \times 10^{-6} \textrm{ C}\) and \(q_2 = +1.5 \times 10^{-6} \textrm{ C}\), separated by \(0.20 \textrm{ m}\). Find the magnitude of the force and state the direction.
Step 1: Use Coulomb's Law.
\(F_e = k\dfrac{|q_1q_2|}{r^2}\)
Step 2: Multiply the charge magnitudes.
\(|q_1q_2| = (4.0 \times 10^{-6})(1.5 \times 10^{-6}) = 6.0 \times 10^{-12}\)
Step 3: Square the distance.
\((0.20)^2 = 0.040\)
Step 4: Calculate the force.
\(F_e = 8.99 \times 10^9 \cdot \dfrac{6.0 \times 10^{-12}}{0.040}\)
\(\dfrac{6.0 \times 10^{-12}}{0.040} = 1.5 \times 10^{-10}\)
So \(F_e = 8.99 \times 10^9 \cdot 1.5 \times 10^{-10} = 1.3485 \textrm{ N}\)
Step 5: Determine direction.
One charge is negative and one is positive, so the force is attractive.
The result is approximately \(1.35 \textrm{ N}\), and the charges pull toward each other.
These examples show that formulas are not just symbols. They allow us to make quantitative predictions about real systems.
The law of gravitation helps engineers predict the forces acting on satellites, space stations, and moons. A small change in orbital distance changes the gravitational force, which affects speed and path. Weather satellites, communication satellites, and GPS systems all depend on accurate gravitational calculations.
Electrostatic force appears in many technologies. Laser printers and photocopiers use electric charges to attract toner particles to specific places on paper. Electrostatic paint spraying uses charge to help paint spread evenly onto surfaces. Lightning also involves large-scale electric forces between charged regions.
At the particle scale, electric force is essential to atomic structure. Protons and electrons attract each other, helping atoms exist. At the same time, electrons repel each other. Even though those systems are more complex in detail, the basic two-object rule from Coulomb's Law still explains a great deal about pairwise interactions.
Why fields matter
A field lets us describe how one object influences space around it. Once the field exists, another object entering that region experiences a force. This is a powerful scientific model because it connects object properties, distance, force, and energy transfer in one framework.
The field idea also explains why the same inverse-square pattern appears in both gravity and electrostatics. As the distance from an object increases, its influence spreads out through a larger area, so the force weakens rapidly.
One common mistake is forgetting to square the distance. In both laws, the denominator is \(r^2\), not just \(r\). This changes the result a lot.
Another mistake is using the wrong distance. In these formulas, \(r\) is the distance between the two objects. For spherical objects, that usually means center-to-center distance, just as shown earlier in [Figure 2].
Students also sometimes confuse magnitude with direction in Coulomb's Law. The formula \(F_e = k\dfrac{|q_1q_2|}{r^2}\) gives magnitude. You must then use the signs of the charges to decide whether the force is attractive or repulsive.
Finally, be careful with units and powers of ten. Mass should be in \(\textrm{kg}\), charge in \(\textrm{C}\), distance in \(\textrm{m}\), and force in \(\textrm{N}\). Scientific notation is often necessary because the constants and quantities can be very large or very small.
"Nature uses patterns. One of the most powerful is that force often depends not just on what objects are, but on how far apart they are."
When you use these equations well, you are doing more than calculating. You are connecting mathematics to how the universe is organized, from satellites circling Earth to charges interacting in matter.