Every calcium atom in your bones, every oxygen atom you inhale, and much of the iron in your blood has a history that began long before Earth existed. Those atoms were not present at the start of the universe in their current form. They were built inside stars. That idea is one of the most powerful in science: stars are not just bright objects in the sky, but cosmic factories that create many of the elements that make planets and life possible.
To communicate scientific ideas clearly about stars, we need to connect three big ideas: stars change over time, their changing cores make different elements, and astronomers use evidence from starlight to figure this out. The goal is not just to memorize stages and events such as the red giant phase or a supernova, but to explain how scientists know what stars are made of and how those stars contribute matter to the universe.
At the beginning of the universe, the main elements were hydrogen and helium, with only tiny amounts of a few others. Yet Earth contains carbon, oxygen, silicon, calcium, iron, and many more. A major scientific question is: where did those heavier elements come from? The answer is tied to the life cycles of stars.
Stars spend most of their lives converting lighter elements into heavier ones through nuclear processes in their cores. The exact details can become very advanced, but the core idea is manageable: stars start mainly with hydrogen, fuse it into helium, and later, depending on their mass, can produce heavier elements such as carbon, oxygen, silicon, and iron. When stars die, especially in powerful explosions, many of those elements are spread into space, where they can later become part of new stars, planets, and living organisms.
The gold in jewelry and the iodine in your body were produced by extreme cosmic events. The atoms around you are a record of ancient astrophysical processes.
This topic also shows how science works. We cannot travel to a star's core, but we can still build strong explanations by using observations, models, and physical laws. Astronomers analyze light, compare stars of different masses and ages, and test predictions against evidence.
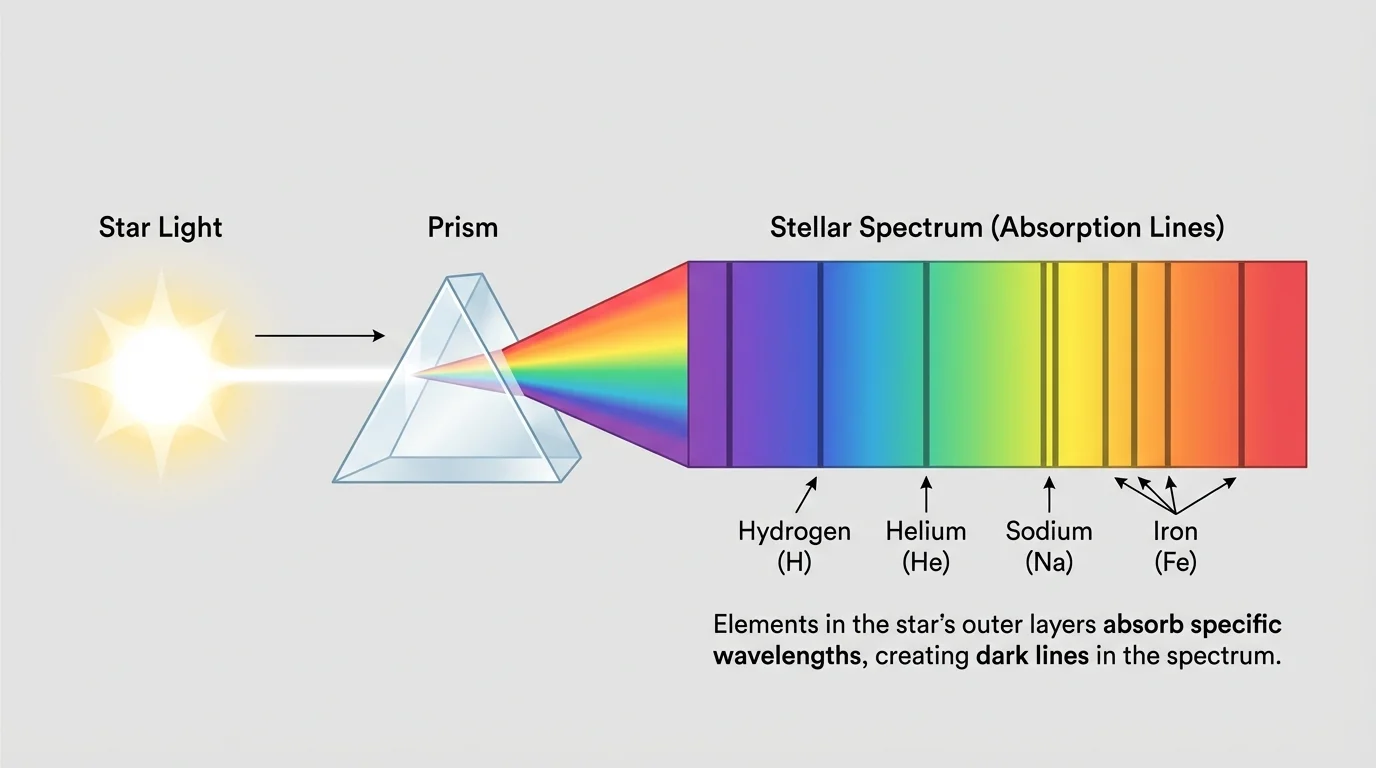
[Figure 1] illustrates a key tool in astronomy: the spectrum, which reveals what a star is made of through the pattern of light separated into colors and lines. When starlight is spread out, it forms a band of colors with specific lines at particular wavelengths. These lines act like fingerprints for elements.
For example, hydrogen produces one pattern of spectral lines, helium another, and iron another. If those lines appear in a star's light, astronomers can infer that those elements are present in the star's outer layers. This is one of the strongest pieces of evidence that stars contain the same kinds of elements found on Earth.
Brightness also matters. A star's apparent brightness is how bright it appears from Earth, and it depends on both the star's intrinsic luminosity and its distance. By comparing brightness and spectral information, astronomers can estimate a star's temperature, composition, size, and stage in its life cycle.
Another clue comes from motion. If spectral lines shift slightly toward the red or blue end of the spectrum, astronomers can tell whether a star is moving away or toward Earth. This is not directly about element production, but it helps scientists study stellar systems, supernova remnants, and the movement of matter enriched by stars.
Spectrum is the spread of light into its component wavelengths or colors. Spectral lines are specific bright or dark lines in a spectrum that identify particular elements because atoms absorb or emit only certain wavelengths.
Because each element has its own set of spectral lines, evidence from starlight allows scientists to communicate claims about composition with precision. This is why astronomy is not guesswork. It is evidence-based science.
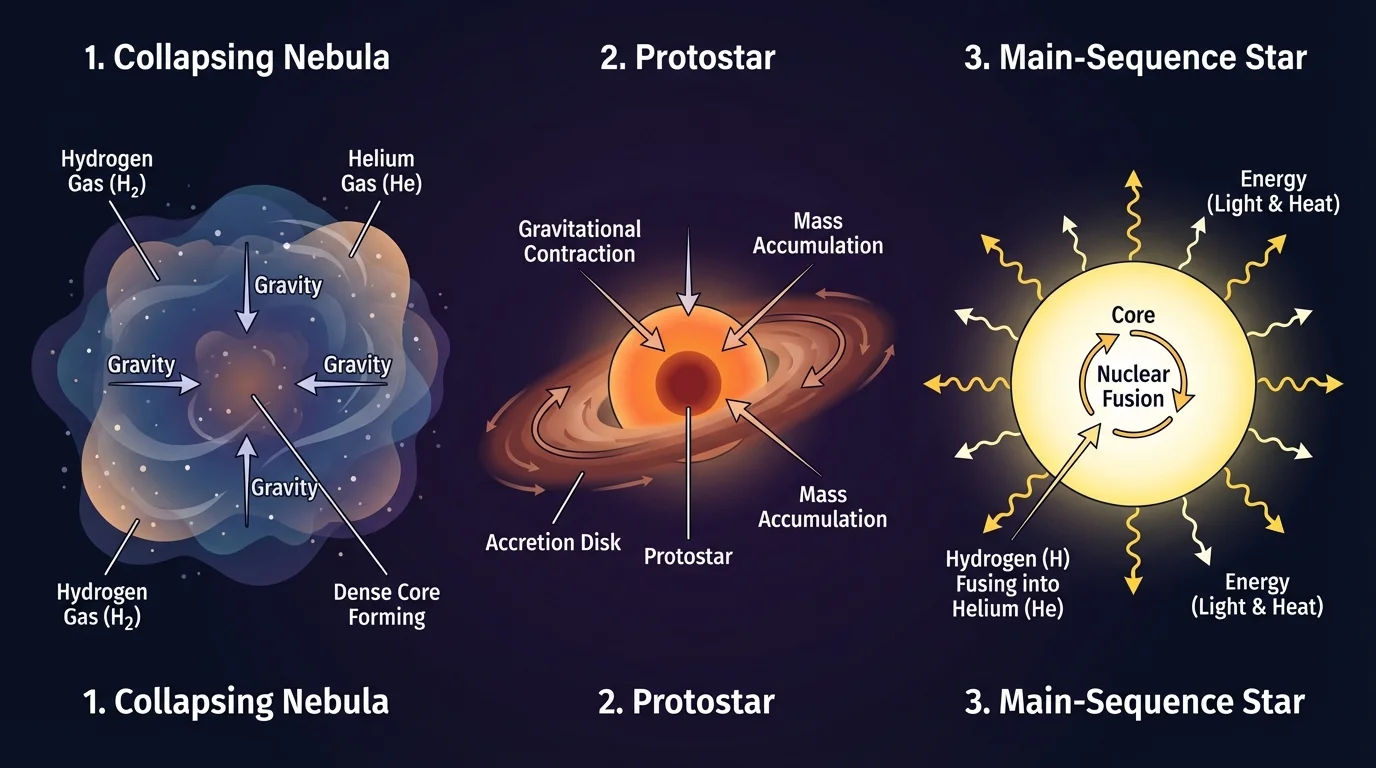
[Figure 2] helps show the early life cycle of a star. A star begins in a huge cloud of gas and dust called a nebula. Gravity pulls matter inward, causing the cloud to contract. As it shrinks, the center becomes hotter and denser. This forming object is called a protostar.
If enough mass gathers, the temperature in the core becomes high enough for nuclear fusion to begin. At that point, a stable star is born. Most stars then enter the longest phase of their lives, the main-sequence star stage.
In a main-sequence star, gravity pulls inward while pressure from hot gas and energy production pushes outward. This balance keeps the star stable for a long time. Our Sun is currently in this stage.
The amount of mass a star has at birth is extremely important. Mass affects core temperature, pressure, lifespan, and which elements a star can eventually produce. A more massive star burns fuel faster, lives a shorter life, and can make heavier elements than a low-mass star.
Atoms are made of smaller particles, including protons in the nucleus. The identity of an element depends on the number of protons in its nucleus, so when nuclear processes in stars change nuclei, new elements can form.
This is one of the biggest differences between chemical and nuclear changes. Burning wood rearranges atoms in molecules, but fusion in stars changes the nuclei themselves.
The central process in a main-sequence star is nuclear fusion. Fusion joins light nuclei together to form heavier nuclei. In stars like the Sun, the most important overall result is that hydrogen nuclei ultimately combine to make helium nuclei, releasing enormous amounts of energy.
We can represent the basic idea in a simplified way as
\[4\textrm{H} \rightarrow \textrm{He} + \textrm{energy}\]
This equation is simplified, but it communicates the key idea: four hydrogen nuclei are transformed into one helium nucleus, and energy is released. That energy eventually escapes the star and reaches Earth as light and other radiation.
The release of energy is related to mass being converted into energy, described by Einstein's relationship
\(E = mc^2\)
where \(E\) is energy, \(m\) is mass, and \(c\) is the speed of light. Even a tiny amount of mass can produce a huge amount of energy because \(c^2\) is very large.
Numeric example: why a small mass loss produces huge energy
Step 1: Use Einstein's equation
If a reaction converts \(m = 0.001 \textrm{ kg}\) of mass into energy, then \(E = mc^2\).
Step 2: Substitute the value of \(c\)
Using \(c = 3.0 \times 10^8 \textrm{ m/s}\), we get \(E = 0.001 \times (3.0 \times 10^8)^2\).
Step 3: Calculate
\[E = 0.001 \times 9.0 \times 10^{16} = 9.0 \times 10^{13} \textrm{ J}\]
This is an enormous amount of energy from just \(0.001 \textrm{ kg}\) of mass.
Main-sequence stars therefore do two things at once: they shine because fusion releases energy, and they slowly change the composition of their cores by turning hydrogen into helium. Spectral evidence from stars at different temperatures and ages helps astronomers compare these stages, linking observation to theory.
As seen earlier in [Figure 1], the composition of a star is not guessed from color alone. Spectral lines provide direct evidence for which elements are present in the star's atmosphere, while models of the core explain which fusion processes are producing energy inside.
A star does not stay on the main sequence forever. When much of the hydrogen in the core has been used, the balance between gravity and outward pressure changes. The core contracts and heats up, while the outer layers expand. The star becomes a giant star. In stars like the Sun, this stage is called a red giant. In more massive stars, it becomes a red supergiant.
[Figure 3] shows that as a massive star evolves, its interior becomes more complex, with layered shells where different fusion reactions occur at different depths. Once the core gets hot enough, helium can fuse to make heavier elements. Among the most important products are carbon and oxygen.
This is a major turning point in stellar element production. A star is no longer making only helium. It is now creating new elements with more protons in their nuclei. In more massive stars, this process continues in stages, producing elements that can include neon, magnesium, silicon, sulfur, and eventually iron in the core.
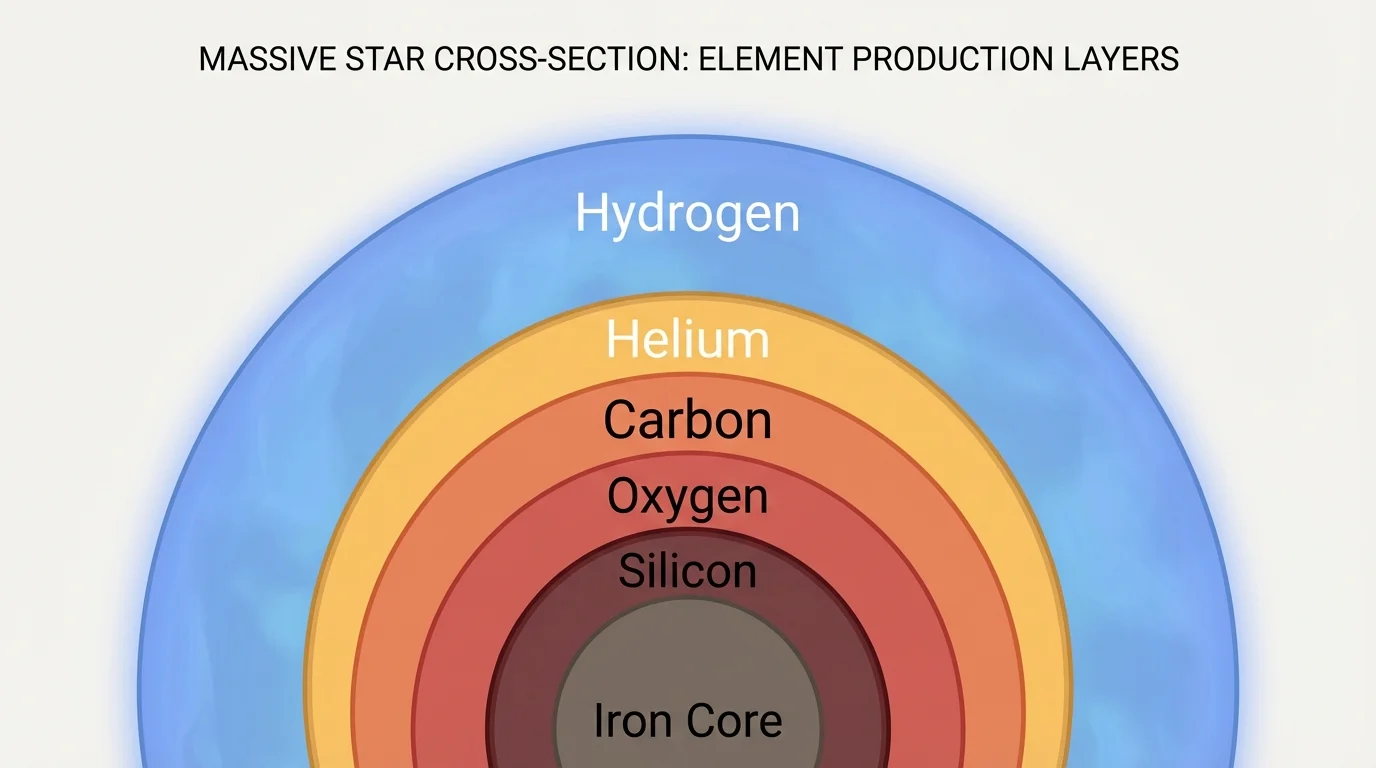
It is important to keep the mass difference in mind. Low-mass stars do not reach the extreme core temperatures needed to build the heaviest elements that massive stars can form. They still play a major role by producing and releasing helium, carbon, and some oxygen.
Why mass matters
Mass controls how strongly gravity compresses a star's core. Greater compression leads to higher core temperature and pressure, which allows more advanced stages of fusion. This is why massive stars can build heavier elements and have more dramatic endings.
A useful scientific explanation often traces a chain of cause and effect: more mass leads to higher core pressure, higher pressure leads to higher temperature, higher temperature enables additional fusion stages, and those stages produce heavier elements.
[Figure 4] helps clarify that stars do not all die the same way. A low-mass star and a high-mass star may begin in similar ways, but mass determines both the ending and the elements released into space.
For a low-mass star such as the Sun, the outer layers drift away into space after the red giant phase. This glowing shell of gas is called a planetary nebula, even though it has nothing to do with planets. The hot leftover core becomes a white dwarf. The white dwarf no longer sustains nuclear fusion, but the material shed earlier enriches space with the elements the star produced.
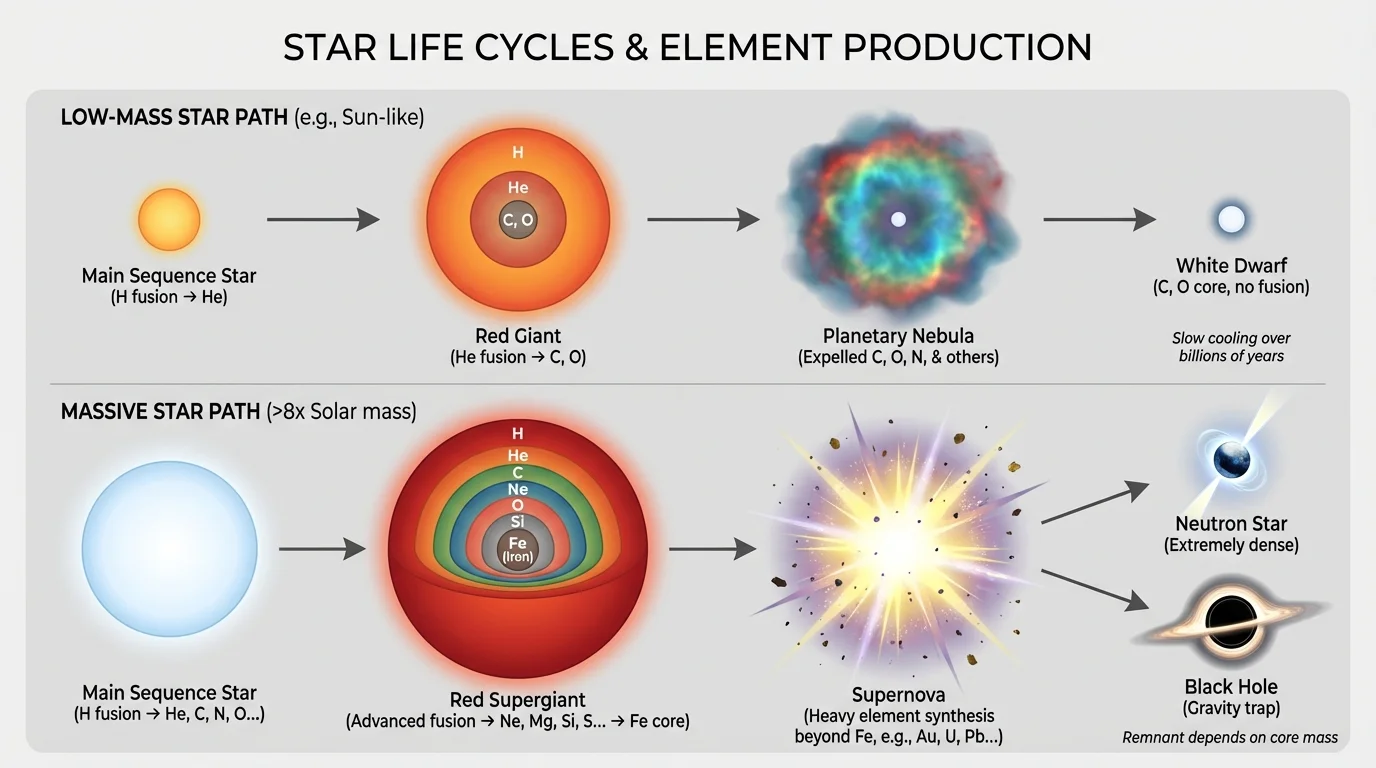
A massive star follows a far more violent path. After building up heavier elements in its interior, it eventually develops an iron-rich core. Iron is a crucial limit because fusion of iron does not release energy in the same way that earlier fusion stages do. Without enough outward energy pressure, the core collapses rapidly.
The collapse triggers a supernova, an extremely powerful stellar explosion. In that event, large amounts of matter are blasted into space. The remnant core may become a neutron star or, if the star was massive enough, a black hole.
| Star type | Typical later stage | Elements mainly produced before death | Final outcome |
|---|---|---|---|
| Low-mass star | Red giant | Helium, carbon, some oxygen | Planetary nebula and white dwarf |
| High-mass star | Red supergiant | Many elements up to iron | Supernova, then neutron star or black hole |
Table 1. Comparison of how low-mass and high-mass stars differ in later stages, element production, and final outcomes.
The comparison in [Figure 4] shows why "all stars make elements" is true but incomplete. The fuller statement is that different stars make different ranges of elements and release them into space in different ways.
For grade-level understanding, the most important idea is broad rather than technical: many elements heavier than iron are associated with extreme events, especially supernova explosions and other high-energy cosmic environments. These conditions allow nuclei to form that ordinary stellar fusion in a stable core cannot easily make.
You do not need to memorize the detailed pathways for each element. The central scientific claim is simpler: normal stellar fusion explains the build-up of many elements up to iron, while explosive events help account for many heavier elements.
Communicating the idea accurately
Step 1: State the pattern
Lighter elements such as helium are formed during the long stable phases of stars.
Step 2: Extend to heavier elements
Massive stars can continue producing heavier elements, eventually reaching iron.
Step 3: Add the limit
Elements heavier than iron are generally linked to extreme explosive conditions rather than ordinary steady fusion in a star like the Sun.
This kind of statement is scientifically strong because it is clear, accurate, and based on evidence without going beyond the required scope.
That distinction helps students communicate the science without getting lost in details. It is enough to know the pattern and the role of stellar mass and explosive endings.
[Figure 5] shows how the matter cycle of stars continues after stellar death, as a supernova remnant spreads enriched material into surrounding space. Gas and dust thrown out by stars mix into the interstellar medium. Later, new nebulae form from that material, and new stars and planets can develop from it.
This means the universe is chemically evolving. Early generations of stars formed mostly from hydrogen and helium. Later generations formed from gas enriched by earlier stars, so they contained more heavy elements. Our solar system formed from matter that had already been processed by older stars.
Astronomers test this idea by examining the spectra of stars of different ages and locations. Younger stars in metal-rich regions often show evidence of more heavy elements than very old stars. In astronomy, elements heavier than helium are often grouped under the term "metals," even though many are not metals in chemistry.
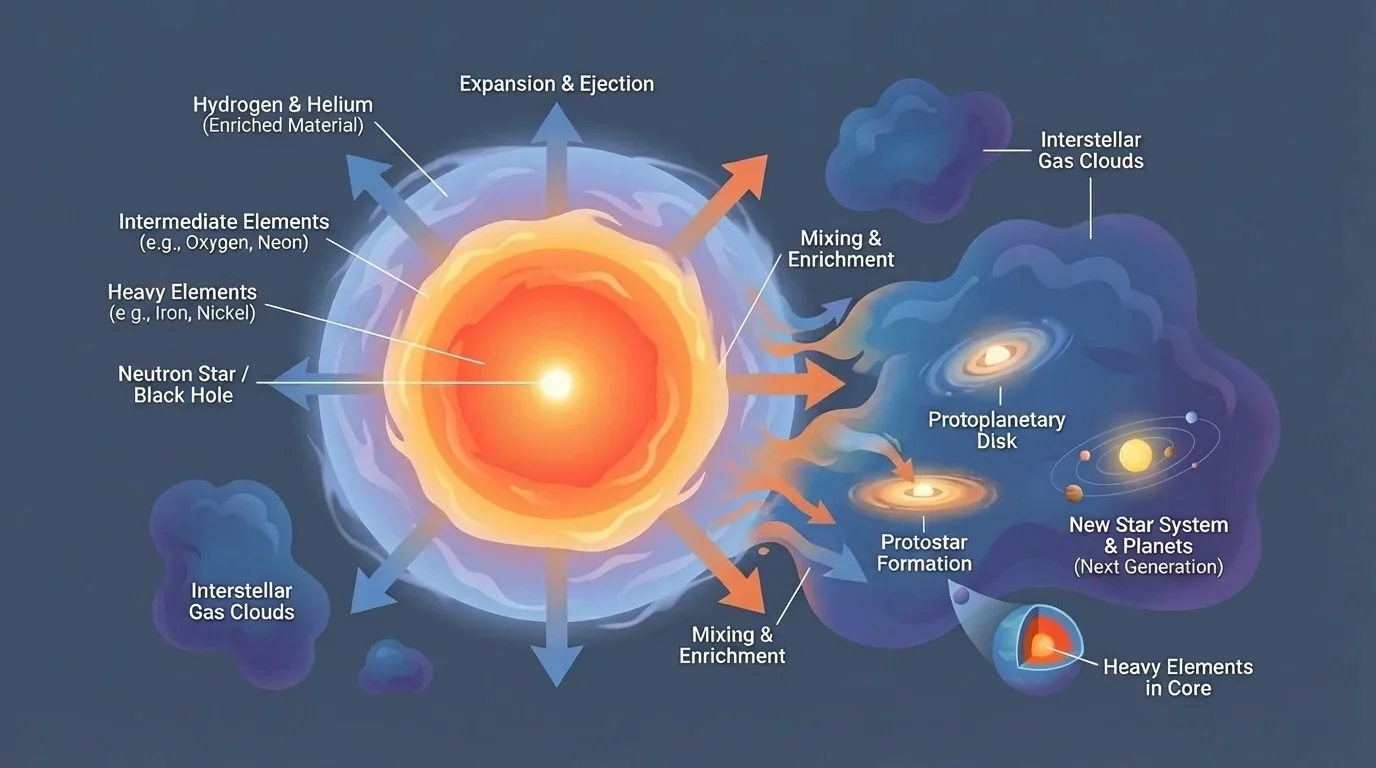
Supernova remnants also emit light that can be analyzed spectroscopically. Scientists detect lines from oxygen, silicon, iron, and other elements in the debris. This is direct evidence that stars not only produce elements but also release them into space.
The iron in your blood helps carry oxygen because of atomic properties shaped by processes that began in ancient stars. Human biology depends on matter forged on a cosmic scale.
When people say, "We are made of star stuff," they are expressing a scientific idea, not just a poetic one. Carbon in organic molecules, oxygen in water, calcium in bones, and iron in blood all connect everyday life to stellar evolution.
The recycling pattern first introduced with star birth in [Figure 2] and shown again through stellar debris in [Figure 5] explains how one generation of stars influences the next. Matter is reused, but its composition changes over time because stars manufacture new elements.
Understanding element production in stars is not only about distant space. Spectroscopy, the same general method used to study stars, is also used in chemistry labs, environmental testing, and forensic science. The logic is similar: identify matter by the way it interacts with light.
In astronomy, communication matters because the science depends on evidence-based claims. A strong explanation includes a claim, evidence, and reasoning. For example, a student might claim that massive stars produce heavier elements than stars like the Sun. The evidence could include observed spectra, the short lifetimes and high core temperatures of massive stars, and supernova observations. The reasoning would connect greater mass to hotter cores and more advanced fusion stages.
Model scientific explanation
Claim: Massive stars are responsible for producing many of the universe's heavier elements.
Evidence: Spectra show that stars and supernova remnants contain elements such as carbon, oxygen, silicon, and iron.
Reasoning: Massive stars reach higher core temperatures than low-mass stars, allowing more fusion stages before they die. Their supernova explosions then spread those elements into space.
This explanation is stronger than simply saying "big stars make more stuff" because it ties observation to physical cause.
The layered structure shown earlier in [Figure 3] also helps scientists explain why different elements appear in different parts of a dying massive star. Observations of the explosion debris can be compared with these models to test whether the explanation fits the evidence.
When communicating this topic, precise language matters. Stars do not "create matter from nothing." Instead, they transform existing nuclei into new elements through nuclear processes. Also, not every star makes every element. The star's mass determines how far element production can proceed.
"The nitrogen in our DNA, the calcium in our teeth, the iron in our blood, the carbon in our apple pies were made in the interiors of collapsing stars."
— Carl Sagan
That statement is memorable because it connects astronomy to ordinary human experience. It turns a distant cosmic process into a clear scientific story about origin, transformation, and evidence.