A speeding roller coaster, a phone battery running low, and a car heating up when it brakes all look like very different events. Yet the same rule explains them all: energy never simply vanishes. If the energy of a system changes, that change must be matched by energy transferred into the system, out of the system, or both. This is one of the most powerful ideas in science because it works across mechanics, chemistry, electricity, and even living systems.
Conservation of energy is not just a rule for solving physics problems. It is a way of thinking. Engineers use it to design engines and power grids. Athletes rely on it whenever chemical energy in muscles turns into motion and heat. Climate scientists use it to track how Earth gains energy from sunlight and loses energy by infrared radiation. Whenever you ask, "Where did the energy go?" you are using the conservation idea.
At first, this idea can feel strange because in everyday language people say energy is "used up." A battery is said to die, fuel is burned up, and a moving ball "loses energy." But scientifically, energy is not destroyed. Instead, it is transferred or transformed. The challenge is not proving that energy exists somewhere; the challenge is tracking where it moved and what form it took.
Conservation of energy means that the total change of energy in any system is always equal to the total energy transferred into or out of the system.
System means the object or group of objects you choose to study. Everything outside it is the surroundings.
Energy transfer happens when energy crosses the boundary of the system, often by heating, mechanical work, electrical work, or radiation.
To use this idea well, you must be clear about what your system is. If your system is a falling ball alone, its gravitational potential energy decreases while its kinetic energy increases, and some energy may leave as sound or thermal energy due to air resistance. If your system is the ball plus Earth, then gravity is an internal interaction within the system, not an external transfer.
[Figure 1] helps show how scientists often picture a system as something with a boundary. The boundary may be real, like the walls of a container, or imagined, like a line drawn around a skateboarder and board. Once the boundary is chosen, we can ask two key questions: What energy is stored inside the system, and what energy crosses the boundary?
Energy can cross a boundary in several common ways. Heating transfers energy because of a temperature difference. Mechanical work transfers energy when a force acts through a distance. Electrical work transfers energy through moving charges in a circuit. Radiation transfers energy by electromagnetic waves, such as visible light or infrared.
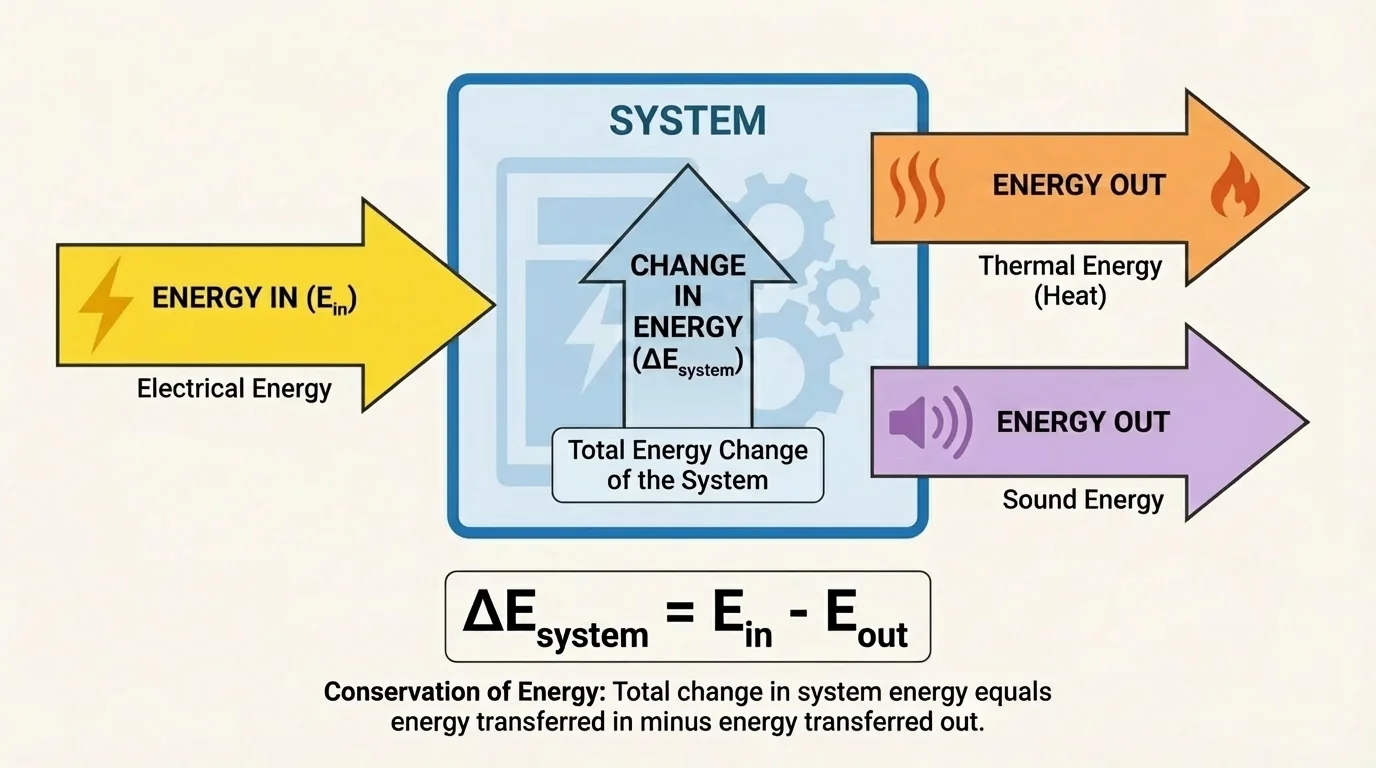
This energy accounting approach is extremely useful because it works whether the system is simple or complicated. A lamp receives electrical energy, converts part of it to light, and transfers much of it to the surroundings as thermal energy. A human body takes in chemical energy from food, transfers some to motion, and releases much as heat. A power plant converts chemical or nuclear energy into electrical energy, but some is always transferred away as unwanted heating.
When we say "total change of energy," we mean the sum of all energy changes inside the chosen system. That total may involve many forms at once. For example, in a bouncing ball, kinetic energy, gravitational potential energy, elastic energy during deformation, sound energy, and thermal energy may all be involved. The law still works because it applies to the total, not just one form.
From earlier physical science, remember that motion, position, temperature, and chemical arrangement can all be linked to energy. Conservation does not say each form stays constant. It says the total is accounted for.
The main accounting statement can be written as
\[\Delta E_{system} = E_{in} - E_{out}\]
Here, \(\Delta E_{system}\) is the change in energy of the system, \(E_{in}\) is the total energy transferred in, and \(E_{out}\) is the total energy transferred out. If more energy enters than leaves, the system's total energy increases. If more leaves than enters, the system's total energy decreases.
This statement is closely related to the first law of thermodynamics, which extends conservation of energy to all physical processes, including heating and internal energy changes. In thermodynamics, internal energy includes the microscopic kinetic and potential energies of particles within matter.
Notice that conservation of energy is not a guess based on one experiment. It is a deeply tested principle. From planetary motion to electric circuits to chemical reactions, every successful model of nature must obey it.
"Energy cannot be created or destroyed, only transferred or transformed."
— Fundamental principle of physics
Energy appears in many forms, and physical situations often involve several at once. A moving object has kinetic energy. An object raised above the ground has gravitational potential energy. A stretched spring has elastic potential energy. A warm object has thermal energy associated with random particle motion. Bonds in fuels and food store chemical energy. Moving charges carry electrical energy, and light carries radiant energy.
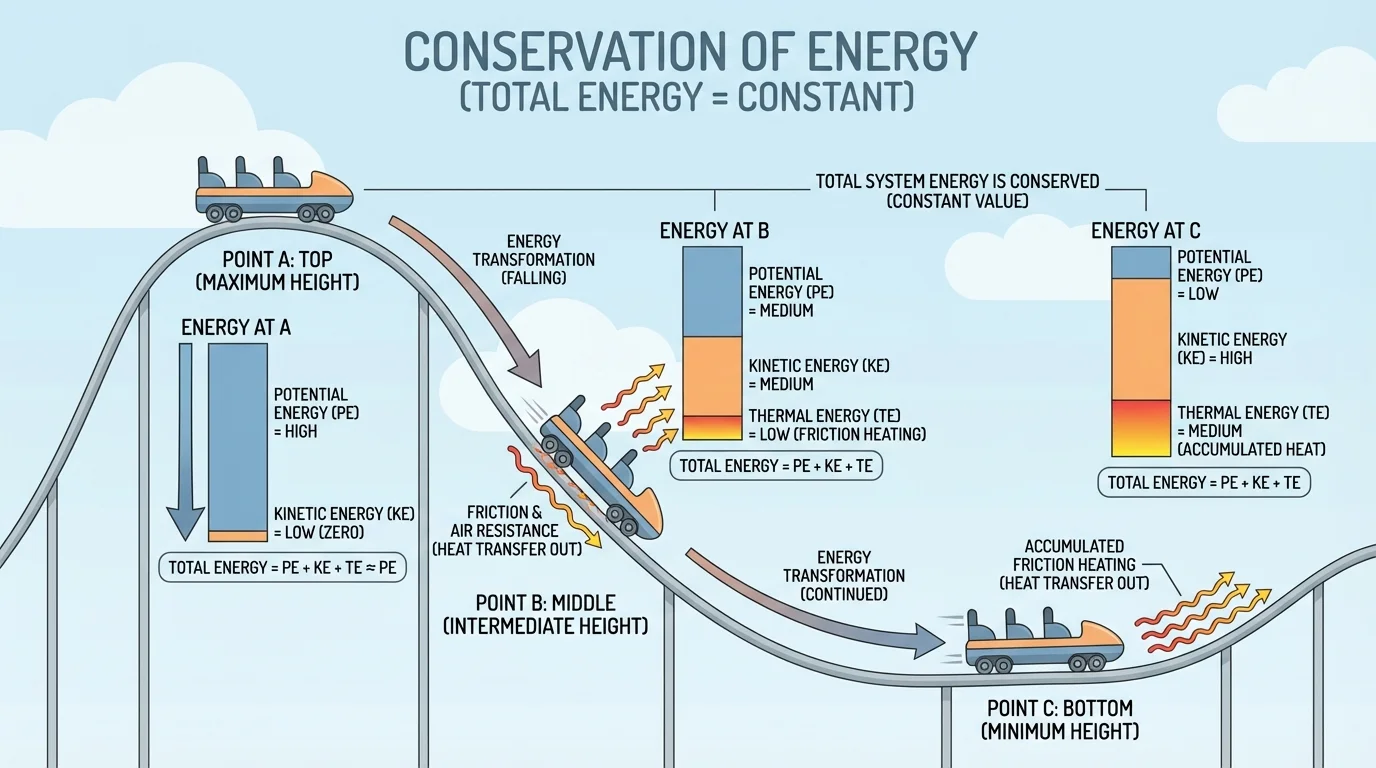
[Figure 2] shows these transformations in a roller coaster. At the top of a hill, the coaster has a large amount of gravitational potential energy. As it descends, that stored energy changes mostly into kinetic energy, so the coaster speeds up. Because of friction in the wheels and air resistance, some energy is also transferred away as thermal energy and sound.
Suppose a coaster car of mass \(500 \textrm{ kg}\) is at a height of \(20 \textrm{ m}\). Its gravitational potential energy relative to the bottom is
\[U_g = mgh = (500)(9.8)(20) = 98{,}000 \textrm{ J}\]
If friction is small, much of that \(98{,}000 \textrm{ J}\) can become kinetic energy at the bottom. If friction and air resistance are significant, the coaster still obeys conservation of energy; some of the original gravitational potential energy ends up warming the wheels, track, and air instead of increasing speed.
Other examples work the same way. In a flashlight, chemical energy in the battery becomes electrical energy in the circuit, then radiant energy and thermal energy in the bulb or LED. In photosynthesis, plants use radiant energy from sunlight to build chemical energy in molecules such as \(\textrm{C}_6\textrm{H}_{12}\textrm{O}_6\). In cellular respiration, organisms transfer some of that chemical energy into useful biological work and some into thermal energy.
| Energy form | What it depends on | Common example |
|---|---|---|
| Kinetic | Motion | A moving bicycle |
| Gravitational potential | Position in a gravitational field | A book on a shelf |
| Elastic potential | Deformation of an elastic object | A stretched rubber band |
| Thermal | Random particle motion and interactions | Hot soup cooling |
| Chemical | Arrangement of atoms and bonds | Fuel, food, batteries |
| Electrical | Charges and electric fields | Current in a circuit |
| Radiant | Electromagnetic waves | Sunlight |
| Sound | Vibrations traveling through matter | A speaker playing music |
Table 1. Common forms of energy, what they depend on, and typical examples.
The way energy is tracked depends on the type of system chosen. In an open system, both matter and energy can cross the boundary. An uncovered pot of boiling water is open because steam and energy both leave. In a closed system, energy can cross the boundary but matter does not. A sealed bottle warming in sunlight is approximately closed. In an isolated system, neither matter nor energy crosses the boundary. Perfectly isolated systems are idealizations, but they are useful in theory.
This classification matters because the phrase "energy of the system" only refers to energy stored inside the defined boundary. If you choose a wider boundary, some transfers that were previously external may become internal rearrangements.
Why the boundary matters
If a hot metal spoon is placed in cool water, energy is transferred by heating from the spoon to the water. If the system is just the spoon, its energy decreases. If the system is just the water, its energy increases. If the system is spoon plus water together, then the transfer between them is internal, and the total energy of the combined system remains constant if no energy leaves to the surroundings.
Several formulas help describe specific energy forms. For translational kinetic energy,
\[K = \frac{1}{2}mv^2\]
For gravitational potential energy near Earth's surface,
\(U_g = mgh\)
In these formulas, \(m\) is mass, \(v\) is speed, \(g \approx 9.8 \textrm{ m/s}^2\), and \(h\) is height relative to a chosen reference level. These equations are not the whole law of conservation of energy. They are tools for calculating parts of the total energy.
For example, if a \(2 \textrm{ kg}\) object moves at \(3 \textrm{ m/s}\), then its kinetic energy is
\[K = \frac{1}{2}(2)(3^2) = 9 \textrm{ J}\]
If the same object is \(5 \textrm{ m}\) above the ground, its gravitational potential energy is
\[U_g = (2)(9.8)(5) = 98 \textrm{ J}\]
Together, these values help describe the object's mechanical energy. But if friction, heating, or sound are involved, you must include those changes too. A complete energy analysis tracks all significant transfers, not just the easiest ones to calculate.
Now consider how the accounting principle is used in actual situations.
Example 1: Falling object
A \(1.5 \textrm{ kg}\) ball falls from rest from a height of \(10 \textrm{ m}\). Ignore air resistance. What is its kinetic energy just before it hits the ground?
Step 1: Identify the starting energy.
At the top, the ball is at rest, so its kinetic energy is \(0\). Its gravitational potential energy is \(U_g = mgh = (1.5)(9.8)(10) = 147 \textrm{ J}\).
Step 2: Apply conservation of energy.
If air resistance is ignored, the loss in gravitational potential energy becomes kinetic energy. So the kinetic energy just before impact is \(147 \textrm{ J}\).
The ball's speed changes, but the total energy is accounted for throughout the fall.
This example is simple because almost all the energy stays within mechanical forms. Many real systems are less tidy because some energy spreads into thermal energy and sound.
Example 2: Lifting with an electric motor
An electric motor transfers \(600 \textrm{ J}\) of energy to lift a box. If \(480 \textrm{ J}\) becomes gravitational potential energy of the box, how much energy leaves in other forms such as heating and sound?
Step 1: Use the energy accounting idea.
The total energy transferred in is \(600 \textrm{ J}\). The useful increase in the box's stored energy is \(480 \textrm{ J}\).
Step 2: Subtract to find the rest.
Other energy transferred out is \(600 - 480 = 120 \textrm{ J}\).
The missing \(120 \textrm{ J}\) is not destroyed; it is transferred mainly as thermal energy and sound.
Engineers often care about how much of the input becomes the desired output. That leads to the idea of efficiency.
\[\textrm{efficiency} = \frac{\textrm{useful energy output}}{\textrm{total energy input}}\]
For the motor above, the efficiency is \(\dfrac{480}{600} = 0.80\), or \(80\%\).
Example 3: Phone battery and flashlight
A small battery delivers \(90 \textrm{ J}\) of chemical energy to a flashlight circuit. If \(15 \textrm{ J}\) is emitted as light, where does the remaining energy go?
Step 1: Identify the total input and useful output.
Total chemical energy transferred from the battery is \(90 \textrm{ J}\). Light energy output is \(15 \textrm{ J}\).
Step 2: Account for the remainder.
The remaining energy is \(90 - 15 = 75 \textrm{ J}\).
That \(75 \textrm{ J}\) is mostly transferred as thermal energy to the bulb, wires, and surrounding air. The flashlight works because energy changes form, not because energy is created.
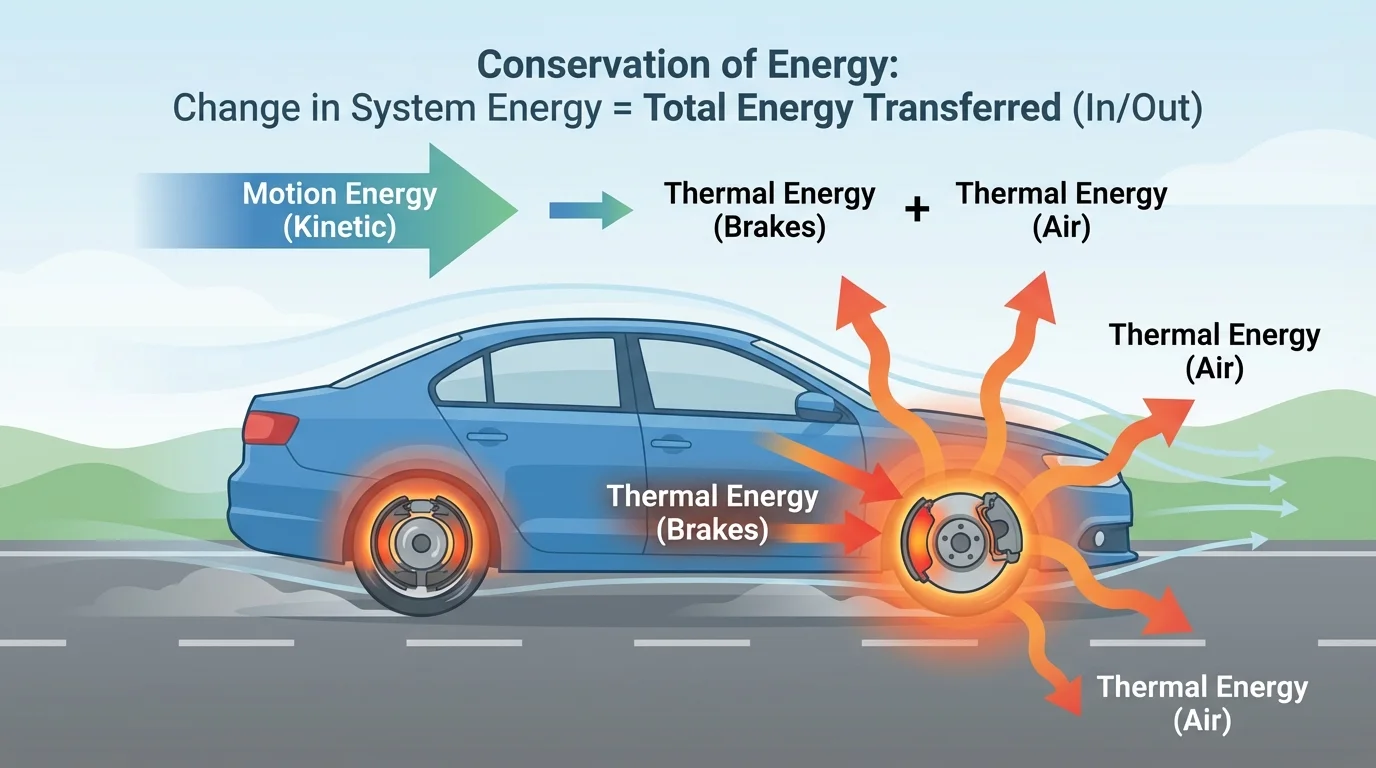
[Figure 3] shows a common example of an important idea at this level: energy can be conserved and still become less useful. This is called dissipation. When organized energy, such as the motion of a car, spreads out into the random microscopic motion of particles, it becomes harder to gather back into a useful form. The energy still exists, but it is less available for doing the particular job we want.
Car brakes are a common example. A moving car has kinetic energy. When the brakes are applied, friction converts much of that energy into thermal energy in the brake pads, wheels, and nearby air. Some energy also becomes sound. The car slows down, but the total energy of the wider system is still accounted for.
The same thing happens with a bouncing ball. Each bounce is lower than the previous one because some mechanical energy is dissipated as thermal energy and sound during each impact. Students often say the ball "loses energy," but a more accurate statement is that some of its mechanical energy is transferred to the surroundings and to internal thermal energy.
As we saw earlier in the roller coaster example in [Figure 2], friction does not break the conservation law. It simply shifts energy into forms that are less useful for maintaining motion. Likewise, the system-boundary view from [Figure 1] helps explain why a device may cool down or heat up depending on whether thermal energy is crossing the chosen boundary.
Modern regenerative braking in electric vehicles reduces dissipation by converting some of the car's kinetic energy back into electrical energy, which recharges the battery instead of wasting as much energy as heat.
Conservation of energy helps explain power generation. In a fossil-fuel power plant, chemical energy in fuel is released by combustion and used to heat water into steam. The steam turns a turbine, converting thermal energy into mechanical energy, and a generator converts that into electrical energy. At each stage, some energy is transferred away as heating to the environment, so no plant is \(100\%\) efficient.
Biology also depends on energy conservation. In the human body, chemical energy from food is transferred into muscle contraction, nerve signaling, and maintaining body temperature. During exercise, not all energy intake becomes motion; much becomes thermal energy, which is why athletes sweat. If a runner climbs stairs, some chemical energy increases gravitational potential energy, while some is dissipated as heat.
Earth's climate can also be viewed as an energy system. The planet receives radiant energy from the Sun and emits radiant energy back into space. If incoming and outgoing energy are balanced on average, Earth's average temperature stays relatively stable. If more energy enters than leaves over long periods, the climate system warms.
Even chemistry fits this same principle. In a chemical reaction such as
\[2\textrm{H}_2 + \textrm{O}_2 \rightarrow 2\textrm{H}_2\textrm{O}\]
energy is not created from nothing. The total energy depends on the bonds broken and the bonds formed. If the products have lower chemical potential energy than the reactants, energy is transferred to the surroundings, often as heat and light.
A major misconception is that energy is "used up." What is really used up is often a concentrated, useful form of energy, such as the chemical energy in gasoline. After combustion, the energy is more spread out, mostly as thermal energy, and much harder to convert back into the original form.
Another misconception is to confuse energy with force. Force is an interaction that can change motion. Energy is a property that can be stored and transferred. They are related, but they are not the same thing.
Students also sometimes think that if an object stops moving, it has no energy at all. But a stopped object may still have thermal energy, chemical energy, gravitational potential energy, or elastic energy. Motion is only one way energy can appear.
You can observe energy conservation in everyday life without advanced equipment. Rub your hands together quickly. Your hands slow down unless your muscles keep supplying energy, and they become warmer because mechanical energy is being transferred into thermal energy. Drop a ball and listen for the sound at impact. Notice that it does not bounce back to its original height, which shows that some mechanical energy is transferred into other forms.
A pendulum is another powerful example. At the highest points, gravitational potential energy is greatest and kinetic energy is least. At the lowest point, kinetic energy is greatest. Over time the swing becomes smaller because air resistance and friction dissipate some energy as heat and sound. The pattern matches the same principle used for roller coasters, brakes, and motors.
Once you begin viewing events through energy accounting, many processes become easier to understand. Instead of asking whether energy disappeared, ask what system you are studying, what energy stores changed, and what transfers crossed the boundary. That question works from the scale of atoms to the scale of planets.