When a phone battery dies, a car stops after braking, or a light bulb gets hot, it can feel as if energy has simply vanished. But it has not. One of the most important ideas in science is also one of the most surprising: energy does not disappear. Instead, it changes form, and very often some of it ends up as warming in the surrounding environment. That is why machines heat up, why no engine is perfectly efficient, and why controlling waste heat is one of the central challenges of modern engineering.
Energy is involved in nearly everything you do: charging a laptop, cooking food, riding a bicycle, playing sports, and using transportation. In each case, energy is transferred and converted. The difficult part is not just getting energy, but getting it in a form that is useful for a purpose. A phone battery stores chemical energy that can be converted into electrical energy for the screen and processor. But during use, some of that energy also becomes thermal energy, warming the phone and the air around it.
This distinction between having energy and having energy in a useful form matters in science, engineering, economics, and environmental policy. A power plant may release huge amounts of energy, but if much of it leaves as low-temperature heat to the surroundings, only part of the original energy input becomes useful electrical energy.
Energy is the ability to cause change. It can appear in forms such as kinetic, gravitational potential, elastic, chemical, electrical, radiant, nuclear, and thermal energy.
Energy transfer means energy moves from one object or system to another. Energy conversion means energy changes from one form to another.
Thermal energy is associated with the motion and arrangement of particles in matter. When energy becomes more spread out as thermal energy in the surroundings, it is usually less useful for doing organized work.
A key reason this topic is so important is that real systems are never perfectly ideal. In textbook diagrams, a machine may seem to convert one form of energy neatly into another. In the real world, some energy almost always spreads into the environment through heating, sound, or vibration.
The law of conservation of energy states that energy cannot be created or destroyed. In a closed system, the total amount of energy stays constant. What changes is the form the energy takes and where that energy is located.
For example, when a roller coaster climbs a hill, energy from a motor is transferred to the coaster, increasing its gravitational potential energy. As the coaster descends, that stored energy is converted mostly into kinetic energy. But the conversion is not perfectly clean. Some energy is also transferred as sound, and some becomes thermal energy because of friction in the wheels and air resistance.
This can be expressed very simply: if a system starts with a certain total energy, it ends with the same total energy, even if that energy is now divided among several forms. If a falling object loses gravitational potential energy by an amount of \(200 \textrm{ J}\), then the total gain in other forms must also add up to \(200 \textrm{ J}\). It might gain \(150 \textrm{ J}\) of kinetic energy, while \(50 \textrm{ J}\) becomes thermal energy and sound.
"Energy cannot be created or destroyed, only transformed."
— First Law of Thermodynamics
Notice what this principle does not say. It does not say all forms of energy are equally valuable for every task. A moving turbine, an electric current, and gasoline in a tank can be very useful because they can drive organized processes. Warm air scattered through a room still contains energy, but that energy is more difficult to collect and direct into a specific task.
One energy input often splits into multiple outputs, as [Figure 1] illustrates for a machine that produces motion but also warms up. The total energy is still conserved, but only part of it may be useful for the job we want done.
Suppose an electric motor receives \(500 \textrm{ J}\) of electrical energy. If it produces \(380 \textrm{ J}\) of kinetic energy in a fan blade, then \(120 \textrm{ J}\) must have gone elsewhere, usually into thermal energy in the motor coils, bearings, and surrounding air, with perhaps a little sound as well. The energy is not lost in the sense of disappearing; it is lost only in the sense of becoming less useful for the intended purpose.
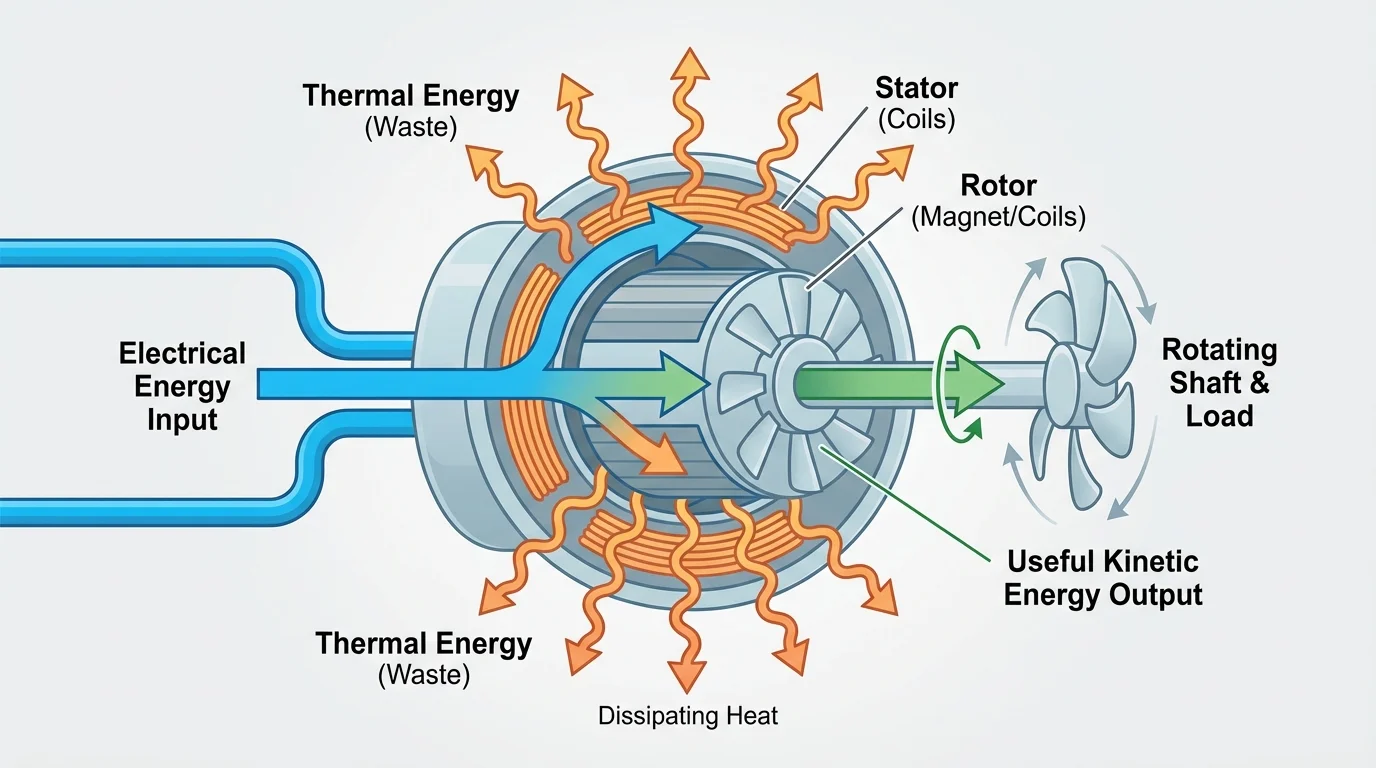
In physics, people sometimes talk about energy quality. This does not mean "good" or "bad" energy. It means how easily energy can be used to produce a desired effect. Electrical energy is high quality for many purposes because it can be transferred and controlled precisely. Thermal energy spread through the environment is lower quality because it is dispersed and harder to channel into organized motion or electrical output.
A bright lamp gives another clear example. The useful output might be light, but an incandescent bulb also becomes very hot. Much of the electrical energy turns into thermal energy rather than visible light. That is why incandescent bulbs are much less efficient for lighting than LEDs. The same idea from [Figure 1] applies: one input energy becomes several output forms, and only some outputs match the intended goal.
Useful does not mean conserved better
All energy is conserved equally well. The word useful refers to whether a form of energy can be easily used for a specific purpose. A car engine, a battery, and an electric motor do not destroy energy when they heat up. They convert some of the input into forms that are harder to use again without additional processes.
This idea helps explain why engineers care so much about heating, friction, and insulation. Their goal is often not to "save energy from being destroyed," which is impossible because energy is already conserved, but to keep more of it in a form that remains useful.
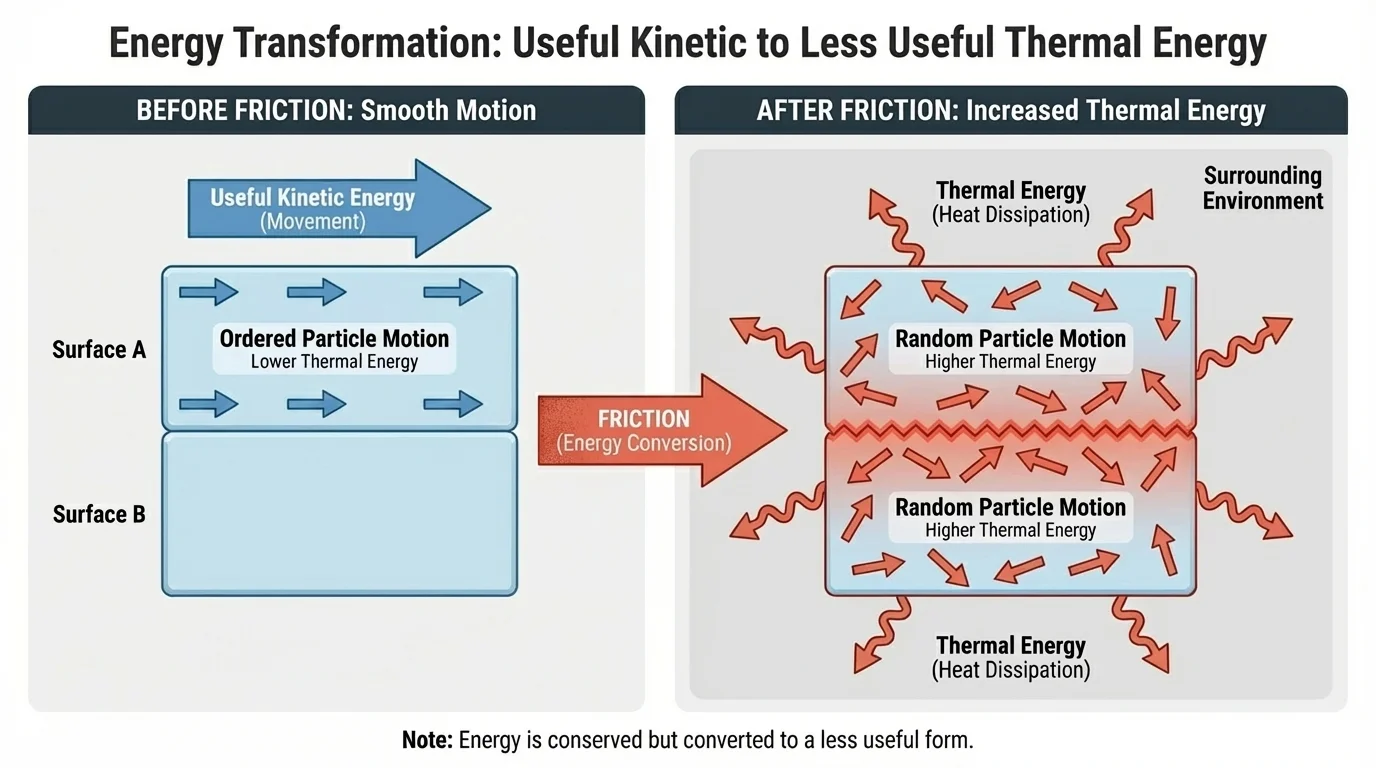
Why does thermal energy appear so often? The answer involves the behavior of matter at the particle level, as [Figure 2] shows. Organized motion, like a sliding box or spinning wheel, can be converted into disorganized particle motion when surfaces rub, objects deform, or electric charges encounter resistance.
When you rub your hands together, friction converts some of the kinetic energy of motion into thermal energy. On the microscopic scale, atoms and molecules in the skin and the air move and interact. Their motion becomes more random, and the temperature rises. Similar processes happen in bicycle brakes, engine parts, and machine bearings.
Electrical resistance is another major source of thermal energy. In a wire, moving charges collide with atoms in the material. These interactions transfer energy to the lattice of particles in the wire, causing it to warm up. This is useful in devices like toasters and electric heaters, but unwanted in phone chargers, power lines, and computers, where heating represents energy converted away from the main purpose.
Collisions also matter. When a ball hits the floor and rebounds to a lower height, some of its mechanical energy has been converted into sound and thermal energy during the impact. The floor and ball deform slightly, and the particles inside them vibrate more vigorously. The ball's lower rebound height is evidence that less of the original energy remains in the organized form of upward motion.
This is why many everyday processes are effectively one-way. It is easy for the organized motion of a car to become heat in brake pads, but much harder for randomly moving warm particles in the brake pads and air to spontaneously reorganize and push the car forward again. The energy still exists, but it has become spread out.
Spacecraft engineers worry intensely about waste heat. In space, there is no air to carry heat away by convection, so satellites must use carefully designed radiators to release thermal energy and prevent equipment from overheating.
The more spread out energy becomes, the more difficult it is to use for doing work. That is one reason the surroundings matter in energy analysis. A machine does not operate in isolation; energy often flows into the environment, especially as heat.
The efficiency of a device tells us what fraction of the input energy becomes useful output energy. It is often written as
\[\eta = \frac{\textrm{useful energy output}}{\textrm{total energy input}} \times 100\%\]
If a machine takes in \(1000 \textrm{ J}\) and produces \(250 \textrm{ J}\) of useful output, then
\[\eta = \frac{250}{1000} \times 100\% = 25\%\]
This means \(75\%\) of the input energy was converted into forms that were not useful for the intended purpose, often thermal energy in the surroundings.
Worked example: electric kettle
An electric kettle takes in \(84{,}000 \textrm{ J}\) of electrical energy. If \(71{,}400 \textrm{ J}\) goes into heating the water, what is the efficiency?
Step 1: Use the efficiency formula.
\(\eta = \dfrac{\textrm{useful output}}{\textrm{total input}} \times 100\%\)
Step 2: Substitute the values.
\(\eta = \dfrac{71{,}400}{84{,}000} \times 100\%\)
Step 3: Calculate.
\(\eta = 0.85 \times 100\% = 85\%\)
The kettle is \(85\%\) efficient. The remaining \(15\%\) warms the kettle body and nearby air.
Efficiency can never exceed \(100\%\) because useful output cannot be greater than total input. In real devices, efficiency is always below \(100\%\). Even highly advanced systems experience some unwanted transfers to heat, sound, or vibration.
Efficiency is not always about avoiding thermal energy. Sometimes thermal energy is the useful output. A room heater may be inefficient at producing light but very efficient at heating a room. What counts as "useful" depends on the intended function.
Energy conversion occurs in familiar objects, but the same principles also govern large engineered systems. A gasoline car converts chemical energy in fuel into thermal energy in the engine, then into mechanical motion of pistons, then into kinetic energy of the car. Along the way, a large amount becomes thermal energy in the engine block, exhaust, tires, and surrounding air.
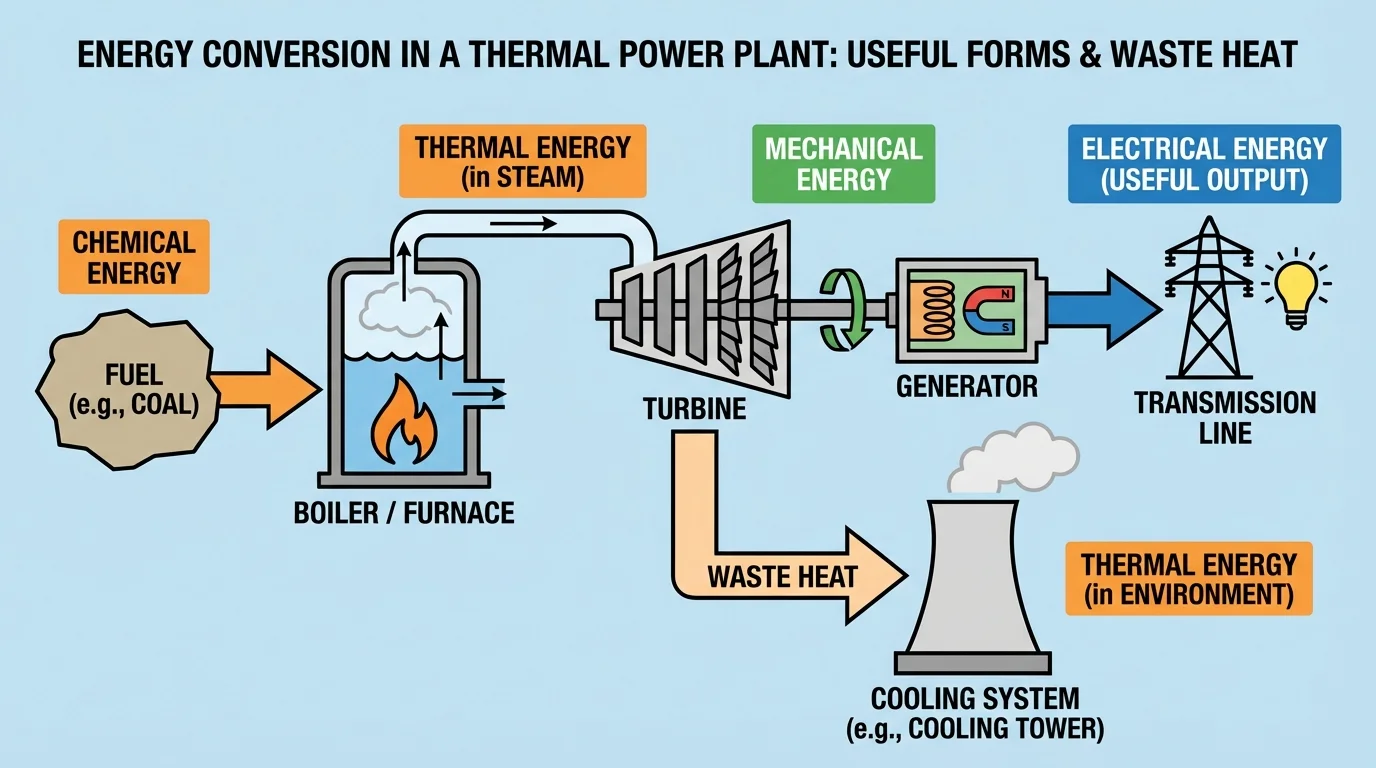
[Figure 3] Large power stations make these energy pathways especially clear. In a fossil-fuel power plant, chemical energy in fuel is converted into thermal energy by combustion. That thermal energy heats water into steam, the steam spins a turbine, and the turbine drives a generator that produces electrical energy. At each stage, some energy spreads into the environment as heat.
Even renewable systems involve similar ideas. In a wind turbine, kinetic energy in moving air becomes rotational kinetic energy in the blades, then electrical energy in the generator. Friction in moving parts and electrical resistance in wires still convert some energy into thermal energy.
Human bodies are also energy-conversion systems. During exercise, chemical energy from food is converted into kinetic energy for movement, but much of it becomes thermal energy. That is why your body temperature rises during intense activity and why sweating helps remove thermal energy. Like machines, biological systems are not \(100\%\) efficient.
Consider these common examples:
| System | Input Energy | Main Useful Output | Common Less Useful Outputs |
|---|---|---|---|
| LED flashlight | Electrical | Light | Thermal energy |
| Bicycle brakes | Kinetic | Slowing the wheel | Thermal energy, sound |
| Phone charger | Electrical | Chemical energy stored in battery | Thermal energy |
| Car engine | Chemical | Kinetic energy of vehicle | Thermal energy, sound |
| Power plant | Chemical or nuclear | Electrical | Thermal energy to surroundings |
Table 1. Examples of systems in which input energy is split into useful outputs and less useful forms, especially thermal energy.
Power grids also face energy conversion limits. Electricity moving through transmission lines encounters resistance, so some electrical energy becomes thermal energy. Engineers reduce these losses by using high voltages, appropriate materials, and careful design. The basic issue is the same: the challenge is not that energy disappears, but that some of it shifts into forms that are less useful for the goal.
One of the deepest ideas in physics is that energy naturally tends to become more spread out. A concentrated, organized form of energy can easily turn into a more dispersed form. For example, electrical energy can power a speaker, and the speaker can produce sound and heat. But the reverse process, in which random thermal motion from the room spontaneously reorganizes into electrical energy in your battery, does not happen by itself in everyday conditions.
This does not violate conservation of energy. Instead, it shows that the degradation of useful energy into less useful forms is common in real processes. "Degradation" does not mean the energy is gone. It means the energy has become more diffuse and less available for doing organized work.
Temperature and thermal energy are related but not identical. Temperature measures the average kinetic energy of particles, while thermal energy depends on the total energy of particle motion and interactions in a substance.
A dropped ball illustrates this well. If it starts with gravitational potential energy, then bounces lower each time, the missing mechanical energy has been converted mostly into thermal energy and sound. To restore the ball to its original height, external energy must be added again. The process does not reverse on its own.
This is why engineers use lubricants, insulation, streamlined shapes, and low-resistance materials. These methods do not change the law of conservation of energy. They reduce how quickly useful energy degrades into dispersed thermal energy.
You can observe these ideas directly with safe, simple investigations. Rub your hands together for \(10\) to \(15\) seconds. The warming shows kinetic energy being converted into thermal energy by friction. Roll a ball across the floor and notice it eventually stops; friction and deformation transfer energy to the floor, the ball, and the air.
Another example is stretching and releasing a rubber band carefully. Some energy is stored elastically, but repeated stretching often makes the band feel warmer. Not all the energy returns to organized motion; some becomes thermal energy because the material's internal structure resists perfect recovery.
Worked example: bouncing ball
A ball has \(12 \textrm{ J}\) of gravitational potential energy before falling. Just after bouncing, it has \(8 \textrm{ J}\) of mechanical energy. How much energy was converted to other forms?
Step 1: Compare initial and remaining mechanical energy.
Energy converted to other forms is \(12 - 8\).
Step 2: Calculate the difference.
\(12 - 8 = 4\)
Step 3: State the result with units and meaning.
\[4 \textrm{ J}\]
The ball converted \(4 \textrm{ J}\) into thermal energy, sound, and internal vibrations during the bounce.
These small investigations help connect abstract energy diagrams to physical experience. Heat, sound, and slowing motion are clues that energy has changed form, not vanished.
Managing less useful energy is one of the major tasks of modern technology. Computer processors need cooling systems because electrical energy is partly converted to heat. Electric vehicles need battery-management systems to control temperature. Factories capture waste heat when possible to improve overall efficiency.
On a global scale, the issue matters for fuel use and environmental impact. If a power station is only \(35\%\) efficient, then \(65\%\) of the original input energy becomes less useful, much of it as heat released to the environment. Improving efficiency means producing the same useful output with less fuel, which can reduce costs and lower pollution.
This is also why thermal insulation matters in buildings. A heater may successfully convert electrical energy into thermal energy, but if that thermal energy quickly transfers outdoors, the useful warming of indoor space is reduced. Good insulation helps keep energy in the place where it remains useful.
Scientists and engineers therefore ask two related questions: How much energy is there? and How much of it remains useful for the task? Conservation answers the first question. Efficiency, design, and heat management help answer the second.