Your phone can receive a text message through radio waves, your kitchen can heat food with microwaves, and your eyes can detect visible light from stars that may be hundreds of years away. These seem like completely different experiences, yet they all involve the same basic phenomenon: electromagnetic radiation. One of the most powerful ideas in physics is that this radiation can be described in two different but complementary ways. Sometimes it acts like a wave spreading through space. Sometimes it acts like tiny packets of energy called photons.
Electromagnetic radiation includes radio waves, microwaves, infrared radiation, visible light, ultraviolet radiation, X-rays, and gamma rays. The differences among them are real, but they are not different substances. They are all forms of energy carried by the same kind of physical process.
That is why a car radio, a microwave oven, a heat lamp, sunlight, a hospital X-ray machine, and radiation from distant galaxies all belong to one broad family. What changes from one type to another are properties such as wavelength, frequency, and energy.
Waves are repeating disturbances that transfer energy from place to place. Earlier studies of waves may have included water waves or sound waves. Electromagnetic waves are different because they do not need matter such as water or air in order to travel.
Unlike sound, which requires a medium such as air, electromagnetic radiation can move through empty space. That fact is essential to astronomy: sunlight reaches Earth through space, even though space is mostly a vacuum.
As shown in [Figure 1], a electric field is a region where electric forces act on charged particles, and a magnetic field is a region where magnetic forces act. Electromagnetic radiation consists of changing electric and magnetic fields that support each other and move outward through space. The changing electric field produces a changing magnetic field, and the changing magnetic field produces a changing electric field.
In an electromagnetic wave, the electric field and magnetic field are perpendicular to each other, and both are perpendicular to the direction the wave travels. This is why electromagnetic radiation is called a transverse wave. The fields oscillate while the wave as a whole moves forward.
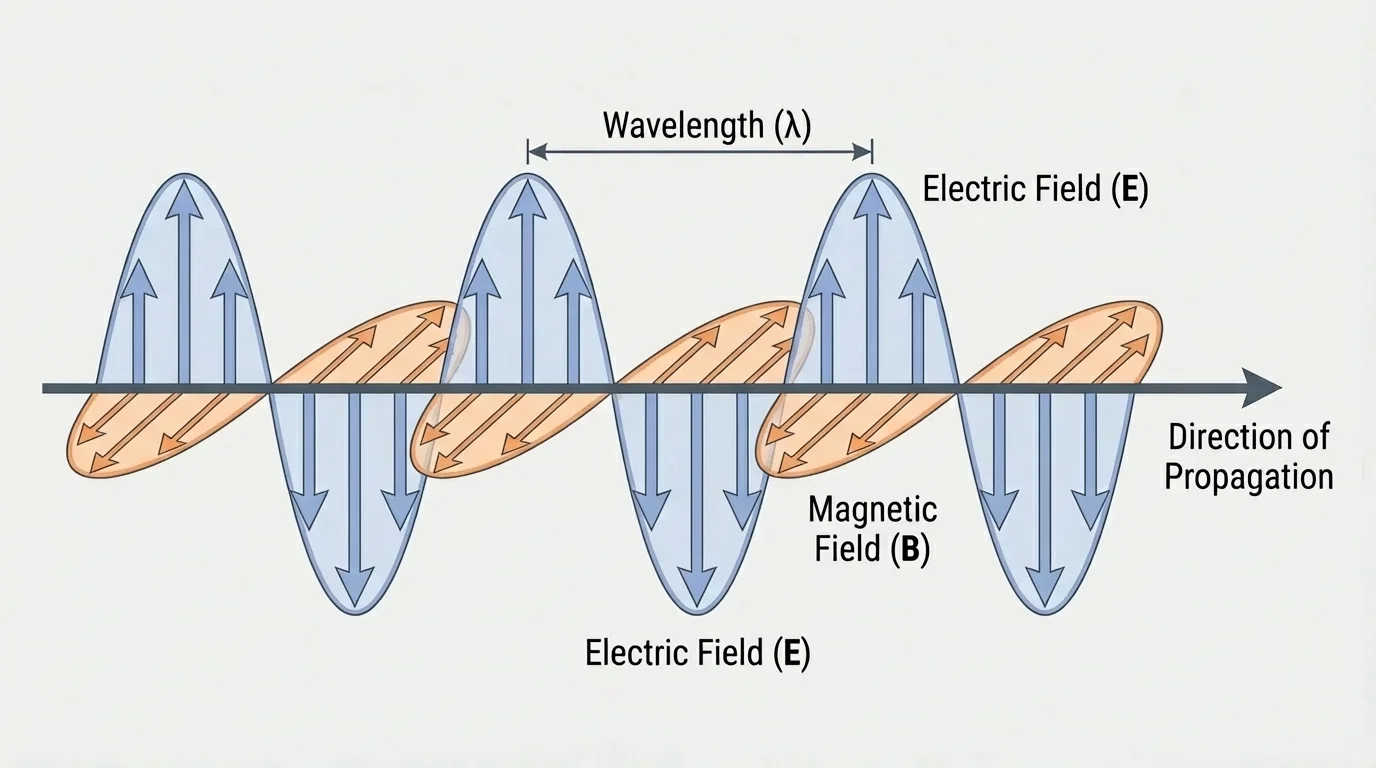
Scientists developed this picture in the nineteenth century, especially through the work of James Clerk Maxwell. Maxwell's equations showed that electric and magnetic fields are deeply connected and that waves made of these fields should travel at a speed that matched the measured speed of light. This led to a major insight: light itself is electromagnetic radiation.
Electromagnetic radiation is energy that travels through space as changing electric and magnetic fields and can also be described as photons.
Wavelength is the distance from one point on a wave to the corresponding point on the next cycle, such as crest to crest.
Frequency is the number of wave cycles that pass a point each second.
Photon is a particle-like packet of electromagnetic energy.
The speed of electromagnetic radiation in a vacuum is commonly represented by the symbol speed of light, or simply \(c\), and its value is approximately \(3.0 \times 10^8\, \textrm{m/s}\). That enormous speed is why communication by radio and light seems almost instantaneous over everyday distances.
The wave model treats electromagnetic radiation as a wave with measurable properties. Three especially important properties are amplitude, wavelength, and frequency. Amplitude is related to how strong the fields are and is connected to intensity, or how much energy arrives per unit area. Wavelength and frequency describe the spacing and timing of the wave.
For electromagnetic waves in a vacuum, wavelength and frequency are linked by a simple relationship:
\[c = f\lambda\]
In this equation, \(c\) is the speed of light, \(f\) is frequency, and \(\lambda\) is wavelength. If frequency increases, wavelength must decrease, because \(c\) stays constant in a vacuum.
Numeric example: finding wavelength from frequency
A radio station broadcasts at \(100 \times 10^6\, \textrm{Hz}\). Find the wavelength in air, assuming \(c = 3.0 \times 10^8\, \textrm{m/s}\).
Step 1: Start with the wave equation.
\(\lambda = \dfrac{c}{f}\)
Step 2: Substitute the values.
\(\lambda = \dfrac{3.0 \times 10^8}{100 \times 10^6}\)
Step 3: Simplify.
\(\lambda = 3.0\, \textrm{m}\)
The radio wave has a wavelength of about \(3.0\, \textrm{m}\).
The wave model helps explain why different electromagnetic waves bend, spread, reflect, and combine the way they do. It also explains why antennas are designed with sizes related to wavelength and why lenses and mirrors can direct light.
Even though all electromagnetic waves move at the same speed in a vacuum, they interact differently with matter because of their different wavelengths and frequencies. Long radio waves can pass around buildings more effectively than visible light, while X-rays can pass through soft tissue more easily than visible light.
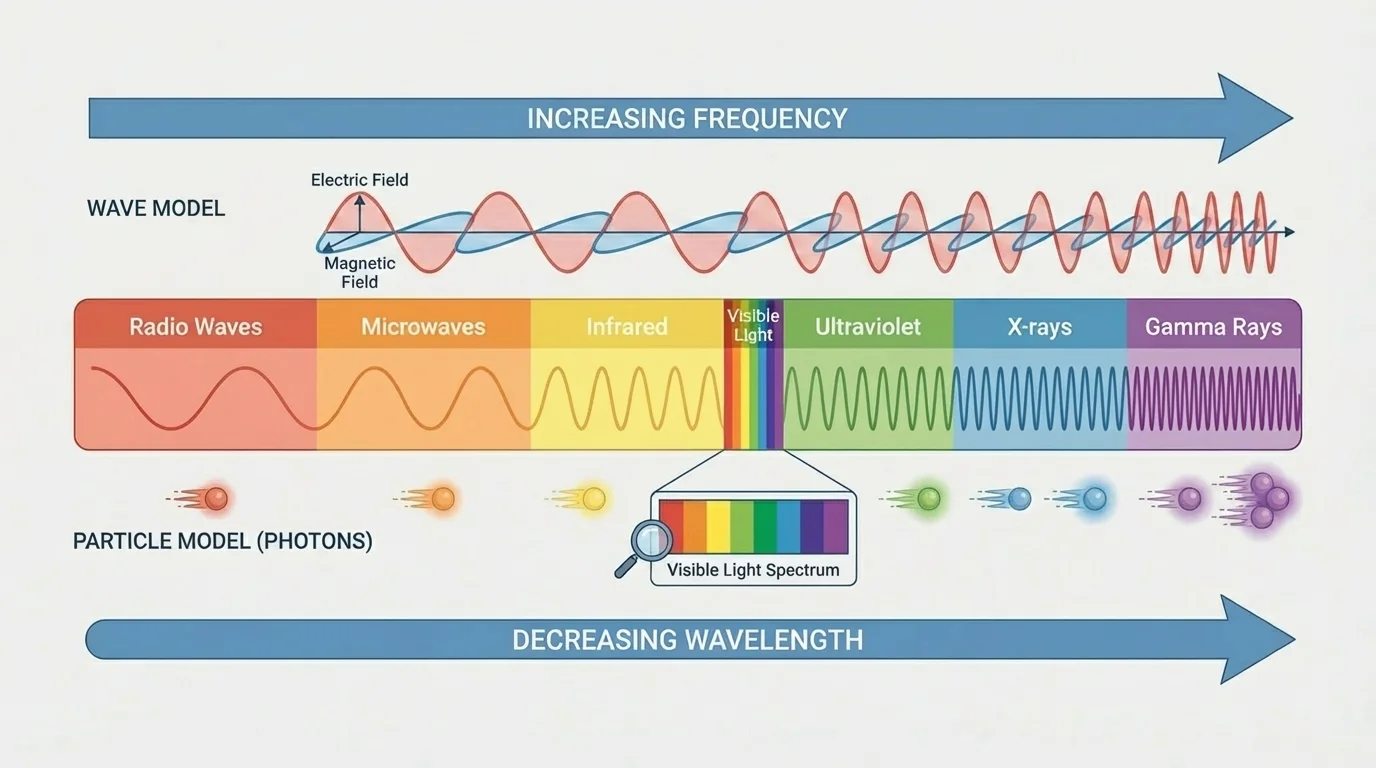
As [Figure 2] illustrates, all electromagnetic radiation fits into a continuous range called the electromagnetic spectrum. The spectrum is usually arranged from long wavelength and low frequency at the radio-wave end to short wavelength and high frequency at the gamma-ray end.
Visible light is only a tiny part of this spectrum. Human eyes detect a narrow band, roughly from red light with longer wavelengths to violet light with shorter wavelengths. Much of the universe is therefore invisible to our eyes unless we use instruments that detect other parts of the spectrum.
| Type | Relative wavelength | Relative frequency | Common uses or sources |
|---|---|---|---|
| Radio waves | Longest | Lowest | Broadcasting, communication, astronomy |
| Microwaves | Very long | Low | Cooking, radar, wireless networks |
| Infrared | Longer than visible | Below visible | Thermal imaging, remote controls, heat transfer |
| Visible light | Middle range | Middle range | Vision, photography, fiber optics |
| Ultraviolet | Shorter than visible | Higher than visible | Sterilization, tanning, some chemical reactions |
| X-rays | Very short | Very high | Medical imaging, security scanning |
| Gamma rays | Shortest | Highest | Nuclear processes, cancer treatment, astrophysics |
Table 1. Major regions of the electromagnetic spectrum with their relative wavelengths, frequencies, and common applications.
As the spectrum moves toward higher frequency, each unit of radiation carries more energy. This is why gamma rays and X-rays can damage cells more readily than radio waves. It is also why ultraviolet radiation from the Sun can cause sunburn, while visible light usually does not.
Many animals detect parts of the electromagnetic spectrum that humans cannot. Some snakes sense infrared radiation from warm prey, and many insects can see ultraviolet patterns on flowers that guide them to nectar.
The spectrum is not divided by sharp natural borders. The named regions are useful categories, but the changes are gradual. A wave just beyond the visible red region is infrared; just beyond visible violet is ultraviolet.
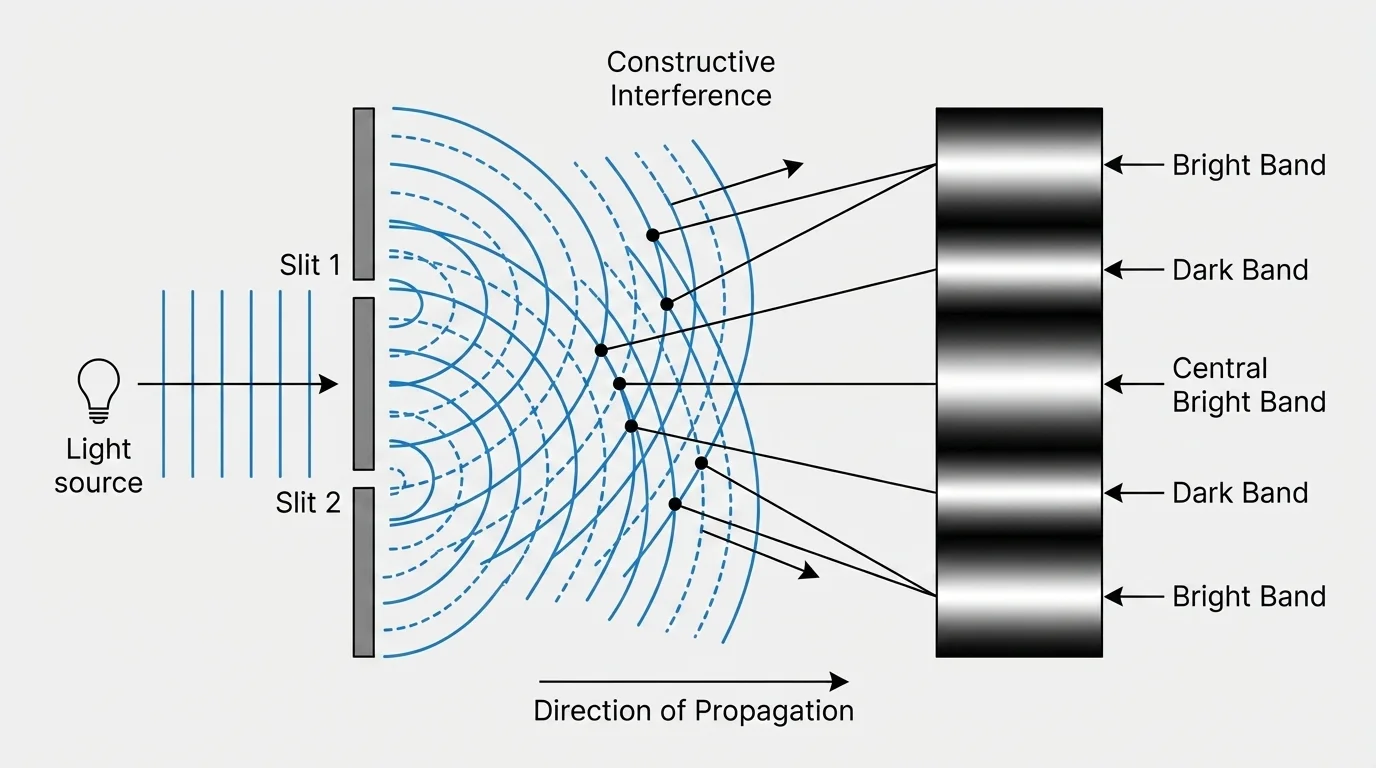
The wave model is especially powerful when radiation shows behaviors typical of waves. One example is interference, where waves overlap and combine. If crests line up with crests, the result is stronger, called constructive interference. If crests line up with troughs, they can cancel, called destructive interference.
As shown in [Figure 3], interference helps explain the colorful patterns seen in soap bubbles, oil films, and some insect wings. Those colors are not always due only to pigments. Sometimes they are caused by light waves reflecting from different thin layers and interfering with each other.
Another wave behavior is diffraction, the spreading of waves when they pass through an opening or around an edge. Diffraction is more noticeable when the opening size is similar to the wavelength. This is one reason radio waves can bend around obstacles better than visible light in many situations.
Reflection and refraction are also well described by the wave model. Reflection occurs when waves bounce from a surface, such as light from a mirror. Refraction occurs when waves change speed as they enter a new medium and bend as a result, such as light entering water from air.
The wave model also explains polarization, which is possible because electromagnetic waves are transverse. Polarizing filters allow waves vibrating in one direction to pass more easily than others. Polarized sunglasses reduce glare because reflected light is often partially polarized.
Why wave behavior matters
When scientists observe diffraction, interference patterns, or polarization, they are seeing evidence that electromagnetic radiation has wave-like properties. These effects depend on repeated oscillations and on how different parts of a wave line up in space and time.
Lasers provide a useful real-world example. Laser light is highly ordered, and its wave properties allow it to be focused precisely and used in barcode scanners, fiber-optic communication, surgery, and distance measurement. The same wave ideas help engineers design antennas, telescopes, and imaging devices.
Later, when considering photons, it is still important to remember the wave effects in [Figure 3]. Light does not stop being wave-like just because particle language is useful for another problem.
The particle description of electromagnetic radiation says that energy is carried in discrete packets called photons. The first time many students meet this idea, it seems strange. How can something be a wave and also come in packets? The key is that models are tools. A model highlights the features that matter most for a given question.
In the photon model, each photon has energy determined by its frequency. The relationship is
\(E = hf\)
Here, \(E\) is the energy of one photon, \(h\) is Planck's constant, and \(f\) is frequency. Higher-frequency radiation has higher-energy photons.
Numeric example: comparing photon energies
Suppose one kind of light has frequency \(5.0 \times 10^{14}\, \textrm{Hz}\) and another has frequency \(1.0 \times 10^{15}\, \textrm{Hz}\). Since \(E = hf\), the second photon has twice the energy of the first because its frequency is twice as large.
This means blue or violet light photons carry more energy than red light photons, and X-ray photons carry far more energy than visible-light photons.
This idea is called quantized energy. Instead of being transferred in any arbitrary amount, electromagnetic energy can be exchanged in specific packets when photons interact with matter.
As [Figure 4] shows, some experiments cannot be explained adequately by the wave model alone. A famous example is the photoelectric effect. In this effect, light striking a metal can eject electrons from the surface, but only if the light has high enough frequency.
This result is surprising from a purely classical wave viewpoint. If light energy were spread continuously like a classical wave, then making the light more intense should eventually eject electrons regardless of color. But experiments showed that below a certain frequency, no electrons are emitted, no matter how intense the light is.
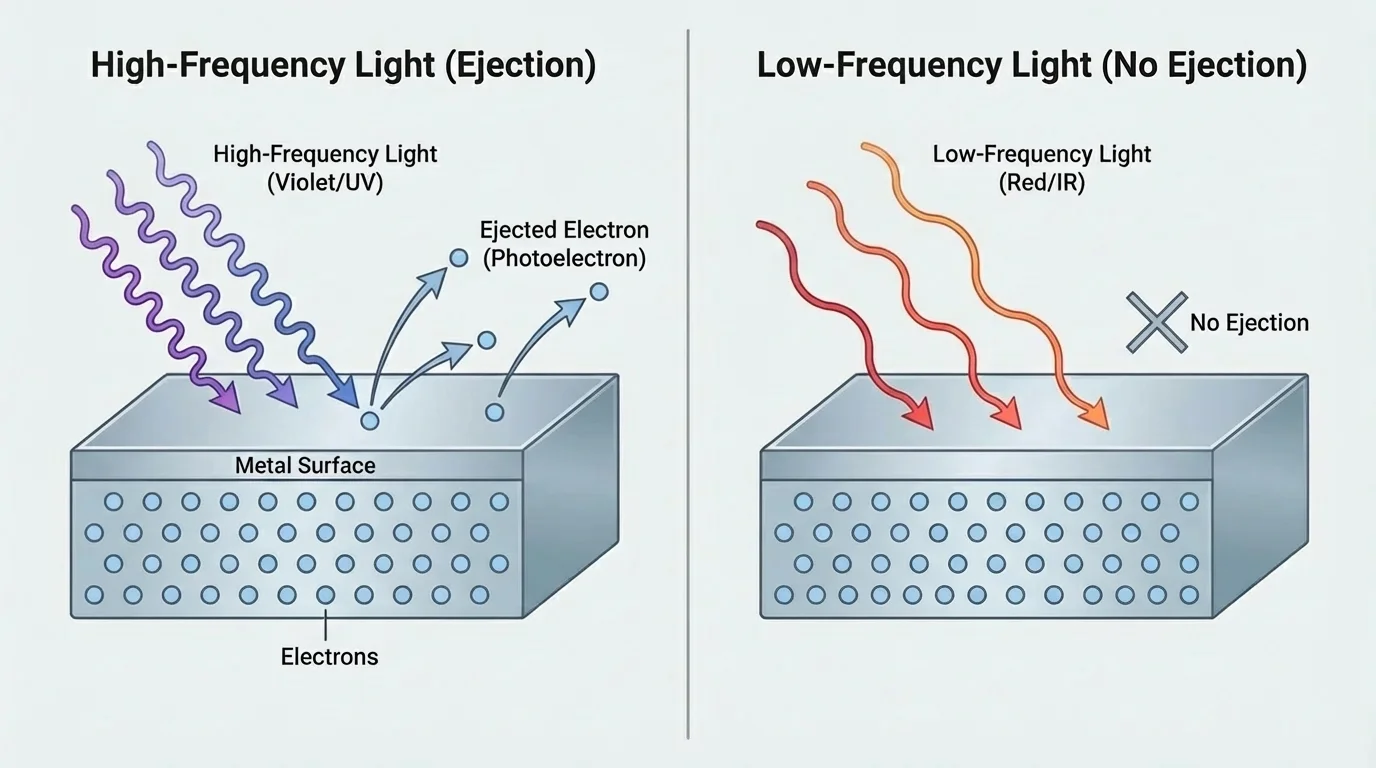
The photon model explains this neatly. One photon interacts with one electron. If the photon energy is large enough, the electron can escape. If the photon energy is too small, increasing the number of low-energy photons does not help. This was one of the key discoveries that helped launch quantum physics.
Solar cells also make sense in photon terms. Sunlight contains photons with different energies. When photons with suitable energy strike the semiconductor material in a solar panel, they can free charges and produce electric current. Digital camera sensors and many light detectors work by similar principles.
Medical imaging and radiation treatment also depend on photon energy. X-ray photons are energetic enough to pass through soft tissues but are more strongly absorbed by denser materials such as bone, creating contrast in an image. In cancer treatment, carefully controlled high-energy radiation can damage the DNA of cancer cells.
Numeric example: why ultraviolet photons are more damaging
If ultraviolet light has frequency \(1.0 \times 10^{15}\, \textrm{Hz}\) and red light has frequency \(4.0 \times 10^{14}\, \textrm{Hz}\), then by \(E = hf\) the ultraviolet photon has \(\dfrac{1.0 \times 10^{15}}{4.0 \times 10^{14}} = 2.5\) times the energy of the red photon.
That higher photon energy helps explain why ultraviolet radiation can trigger chemical changes in skin cells more easily than red visible light.
The same logic from [Figure 4] helps explain why brightness and color are not the same thing. Brightness is related to the number of photons arriving and their total energy flow, while color in visible light is tied to photon frequency.
The phrase wave-particle duality means that electromagnetic radiation shows both wave-like and particle-like behavior. This does not mean light switches back and forth between being two completely different objects. Instead, it means a full description of nature requires more than one classical picture.
If the question is about interference, diffraction, refraction, or polarization, the wave model is usually the most useful. If the question is about energy transfer to atoms, electrons leaving metals, or how detectors count light, the photon model is often more useful.
A useful way to think about it is this: the two models are not enemies. They are complementary descriptions of the same physical reality. Modern physics uses quantum theory to connect them more deeply, but at this level it is enough to recognize when each model explains the evidence best.
"Light has no single everyday analogy that captures everything it does."
— A central lesson of modern physics
This dual nature is not limited to visible light. Radio waves, microwaves, infrared radiation, ultraviolet radiation, X-rays, and gamma rays all share it. The same spectrum shown earlier in [Figure 2] can be discussed in both wave terms and photon terms.
Modern society depends on electromagnetic radiation. Wireless communication uses radio waves and microwaves to transmit information through modulation, encoding data onto waves that antennas send and receive. Fiber-optic systems use light pulses traveling through glass fibers, where reflection and refraction help guide the signals over long distances.
Microwave ovens use microwaves that interact strongly with water molecules in food, causing energy transfer that heats the food. Infrared cameras detect thermal radiation and are used in firefighting, building inspection, astronomy, and environmental monitoring.
Visible light is essential not only for vision but also for photography, microscopy, and plant growth. Ultraviolet radiation can sterilize equipment because high-energy photons can damage the genetic material of microbes. X-rays and gamma rays are used carefully in medicine because their high energies can both help and harm.
Information transfer with electromagnetic waves
Electromagnetic radiation is central to information technologies because waves can be generated, controlled, reflected, focused, and detected. Different wavelength ranges are chosen for different jobs: radio for long-distance broadcasting, microwaves for radar and wireless networks, and visible or infrared light for optical communication.
Astronomers use the full electromagnetic spectrum to study the universe. Radio telescopes detect cold gas clouds and pulsars, infrared telescopes reveal dust-hidden regions where stars are forming, visible-light telescopes show stars and galaxies, and X-ray and gamma-ray telescopes detect extreme events such as black hole accretion and supernova remnants.
Safety depends on understanding both models. Long exposure to intense ultraviolet light can damage skin and eyes. Excessive X-ray or gamma-ray exposure can harm living tissue because high-energy photons can ionize atoms or break chemical bonds. Engineers, doctors, and scientists therefore match the type of radiation to the task and control exposure carefully.
You can observe wave behavior directly with polarized sunglasses and a phone or laptop screen. As you rotate the glasses, the screen can become dimmer because the emitted light is polarized. This is a visible reminder that electromagnetic radiation has directional wave properties.
Photon behavior is less obvious in daily life, but every digital camera, solar calculator, and light sensor depends on it. When light is detected electronically, what matters is not only that a wave arrives but also that energy is absorbed in discrete interactions.
The great achievement of modern physics is not choosing one model and rejecting the other. It is learning that nature can be richer than our first intuitions. Electromagnetic radiation is one phenomenon, but to understand it well, we need both the language of waves and the language of photons.