A smartphone can play music, light up a screen, get warm in your hand, and send signals through the air—all from the same battery. That is not four different "substances" being released. It is one physical quantity, energy, appearing in different ways. This idea is one of the deepest unifying ideas in science: very different events can be described by tracking a single conserved quantity.
Whenever something changes, energy is involved. A runner speeds up, a speaker vibrates, a lamp glows, ice melts, a car engine heats up, a plant captures sunlight, and a star shines. In each case, energy is transferred, transformed, or both. Because of this, energy gives scientists and engineers a powerful way to describe systems that might otherwise seem unrelated.
Energy is not a material you can hold like a rock or pour like water. Instead, it is a quantitative property of a system. That means it can be measured and assigned a value. A system might be a moving bicycle, a cup of hot tea, Earth's atmosphere, a chemical reaction, or an atom. Once a system is defined, we can ask how much energy it has, how that energy is stored, and how it changes.
Energy is a measurable property of a system that depends on the motion of matter and radiation and on the interactions and arrangement of particles within the system.
Conservation of energy means the total energy of an isolated system remains constant, even though energy may transfer between objects or change from one form to another.
This is why energy matters so much in physics, chemistry, biology, Earth science, and engineering. If you can identify the system and track its energy, you can explain what is happening in a clear and consistent way.
One of the most important ideas in physics is that energy is a single quantity that can be tracked across many kinds of change, as [Figure 1] illustrates with a system boundary and energy flows. You may observe motion, heat, light, or sound, but science treats these not as unrelated phenomena, but as different manifestations of the same conserved quantity.
Suppose a skateboarder rolls down a ramp. At the top, the system has more energy associated with position in Earth's gravitational field. As the skateboarder moves downward, that energy becomes more associated with motion. Some energy is also transferred to the wheels, the ramp, and the air, often ending up as thermal energy and sound. If we choose the system carefully, the total energy can still be tracked. Nothing has disappeared; it has changed form or moved elsewhere.
To talk precisely, scientists distinguish between energy transfer and energy transformation. Transfer means energy moves from one object or region to another. For example, a hot pan transfers energy to a cooler egg. Transformation means energy changes from one form to another within a system. For example, in a flashlight, chemical energy in the battery becomes electrical energy, then light and thermal energy.
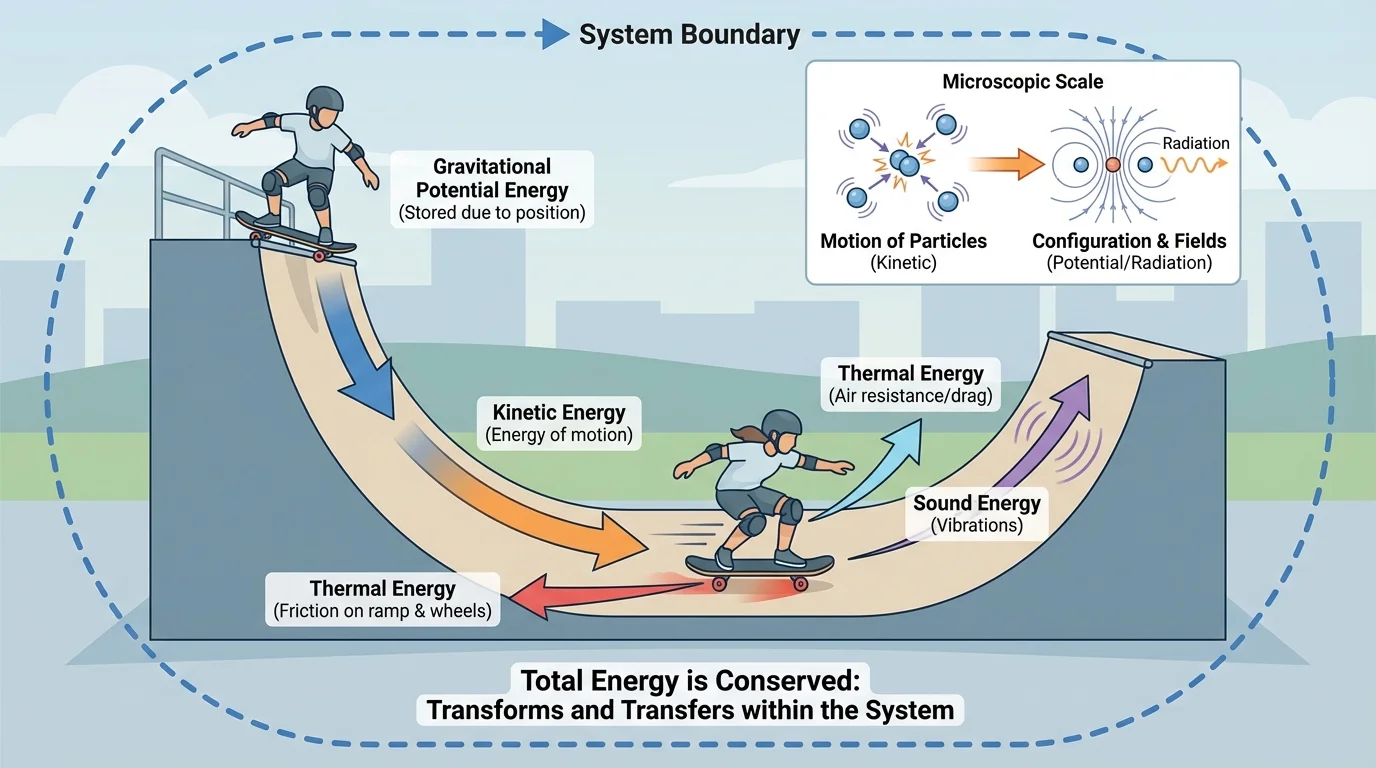
The law of conservation of energy does not mean every form stays constant. The speed of an object can change, the temperature can change, and light can be emitted. What stays constant is the total energy of an isolated system. This idea is so reliable that when scientists encounter an apparent energy loss, they assume they have not yet tracked all of the energy pathways.
"Energy cannot be created or destroyed, only transformed from one form to another."
— First law of thermodynamics
At the high school level, it is useful to think about energy in terms of a basic question: What changes are possible, and where does the ability to produce change come from? A stretched spring can launch a cart. A moving ball can break a window. Sunlight can power a solar panel. In each case, energy helps explain the capacity for change.
At the scale of everyday life, energy appears in forms we can observe directly. These are often called macroscopic forms because they describe large-scale behavior. Common examples include motion, sound, light, and thermal energy.
Motion is associated with the movement of objects. A thrown baseball, a falling rock, and a speeding train all have energy because they are moving. This is called kinetic energy. For an object of mass \(m\) moving with speed \(v\), the kinetic energy is
\[E_k = \frac{1}{2}mv^2\]
If a \(2 \textrm{ kg}\) ball moves at \(3 \textrm{ m/s}\), then its kinetic energy is \(E_k = \dfrac{1}{2}(2)(3^2) = 9 \textrm{ J}\). The unit \(\textrm{J}\), or joule, is the standard SI unit of energy.
Sound is energy carried by vibrations through matter. A guitar string vibrates, setting nearby air molecules into motion. Those vibrations travel to your ear. Sound is easy to notice, but it is often only a small fraction of the total energy involved in a process. For example, when a basketball hits the floor, most energy does not become sound; much of it becomes thermal energy in the ball and floor.
Light is energy carried by electromagnetic radiation. Visible light is only one part of the electromagnetic spectrum. Infrared radiation warms your skin near a heater, ultraviolet radiation from the Sun can damage cells, and X-rays can pass through soft tissue to create medical images.
Thermal energy is related to the microscopic motion and interactions of particles in matter. A hot object and a cold object both contain energy, but the hotter object generally has a greater average particle motion. Thermal energy is not exactly the same as temperature, though the two are related. Temperature measures the average kinetic energy of particles, while thermal energy depends on both temperature and the amount of matter present.
A bathtub of warm water contains more thermal energy than a cup of boiling water, even though the boiling water has a higher temperature. The larger amount of matter makes a big difference.
Some textbooks also discuss chemical, electrical, nuclear, elastic, and gravitational energy as distinct forms. These labels are useful, but they are best understood as special cases of a deeper microscopic picture involving particle motion and particle arrangement.
The microscopic view is where the idea of energy becomes especially powerful. Different large-scale forms can be modeled using particles and interactions, as [Figure 2] shows by comparing motion and spacing in solids, liquids, and gases. At this scale, energy is mainly associated with two broad features: the motion of particles and the configuration, or relative positions, of particles.
Particles in matter are always moving unless they are at absolute zero, a theoretical limit. In gases they move freely and rapidly. In liquids they move past one another. In solids they vibrate around fixed positions. Faster microscopic motion usually means greater kinetic energy at the particle level. This is a major part of thermal energy.
The second major idea is configuration energy, which depends on how particles are arranged relative to one another and how they interact. A compressed spring has energy because its atoms are displaced from their preferred spacing. Water behind a dam has energy because of position in Earth's gravitational field. In chemistry, atoms in molecules have different energies depending on bond arrangements, such as in \(\textrm{CO}_2\), \(\textrm{H}_2\textrm{O}\), or \(\textrm{C}_6\textrm{H}_{12}\textrm{O}_6\).
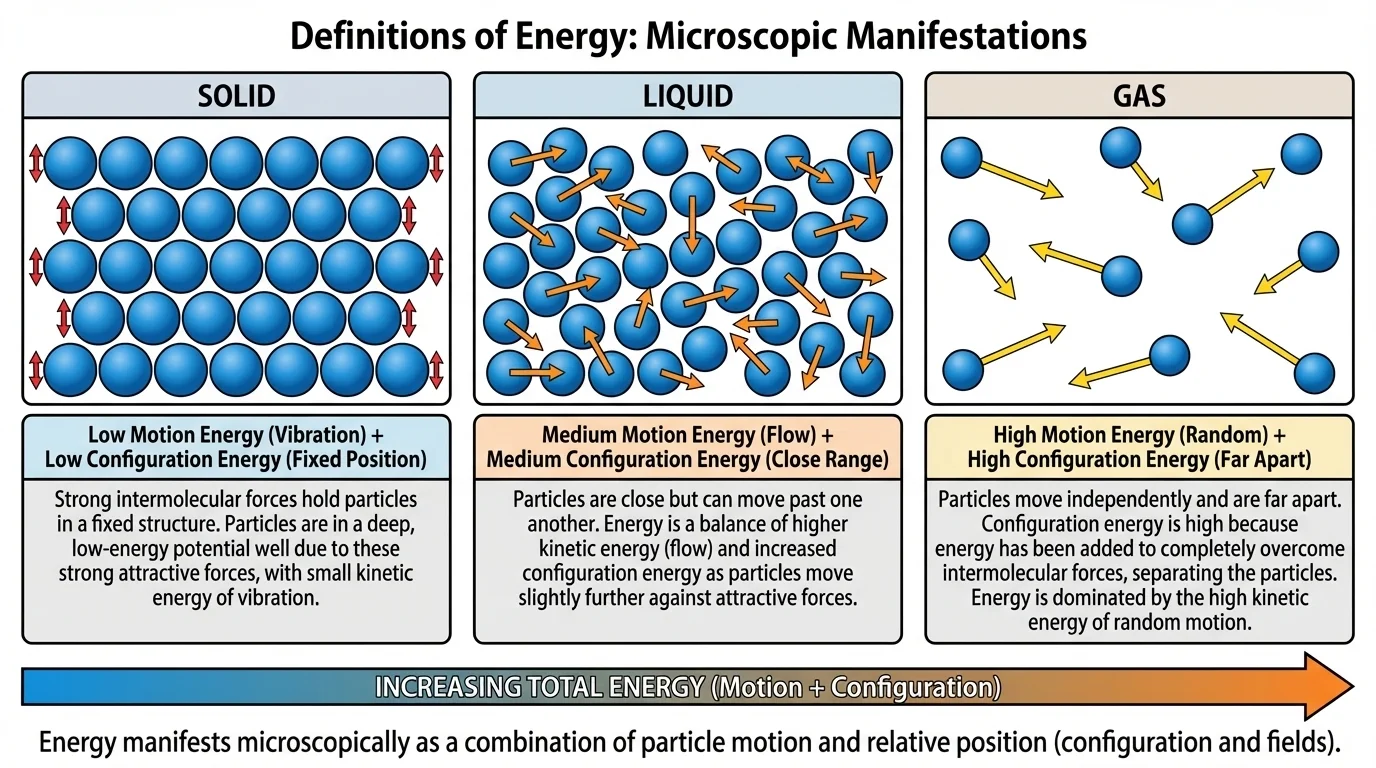
This microscopic approach explains why the many "forms" of energy are really connected. Thermal energy mostly reflects random particle motion and interactions. Elastic energy comes from displaced particle positions in a material. Chemical energy depends on electron arrangements and atomic bonding. Even sound can be seen microscopically as organized vibrations passed from particle to particle.
One quantity, many appearances
At the macroscopic scale, energy may appear as motion, heat, light, or sound. At the microscopic scale, these can be understood as combinations of particle motion and particle arrangement. This is why a single conservation law can describe so many different phenomena.
The particle model also helps explain phase changes. When ice melts, energy is added, but the temperature can remain constant during the melting process. Why? Because the added energy goes mainly into changing particle arrangement rather than increasing average kinetic energy. The particles become less locked into fixed positions, moving from the structured solid state toward the liquid state. The same logic applies during boiling.
Later, when discussing thermal insulation, engines, or chemical reactions, the particle view remains essential. As we saw earlier with macroscopic thermal energy, and as [Figure 2] makes clear, "hotter" matter is not just a vague condition; it reflects average microscopic motion and interaction.
Not all energy needs matter as a carrier. Some energy is associated with fields, and in some cases that energy moves across space as radiation. This idea is especially important for understanding gravity, electricity, magnetism, and light, as [Figure 3] shows.
A field is a way of describing how one object can influence another at a distance. Earth creates a gravitational field, which affects nearby masses. Charged particles create electric fields. Moving charges also create magnetic fields. When we say an object has gravitational potential energy or electrical potential energy, we are describing energy associated with position in one of these interaction fields.
Radiation is a phenomenon in which energy stored in fields moves through space. Sunlight is the most familiar example. Energy produced in the Sun travels through the vacuum of space and reaches Earth as electromagnetic radiation. That energy can warm land and water, power photosynthesis, and be converted to electricity in a solar panel.
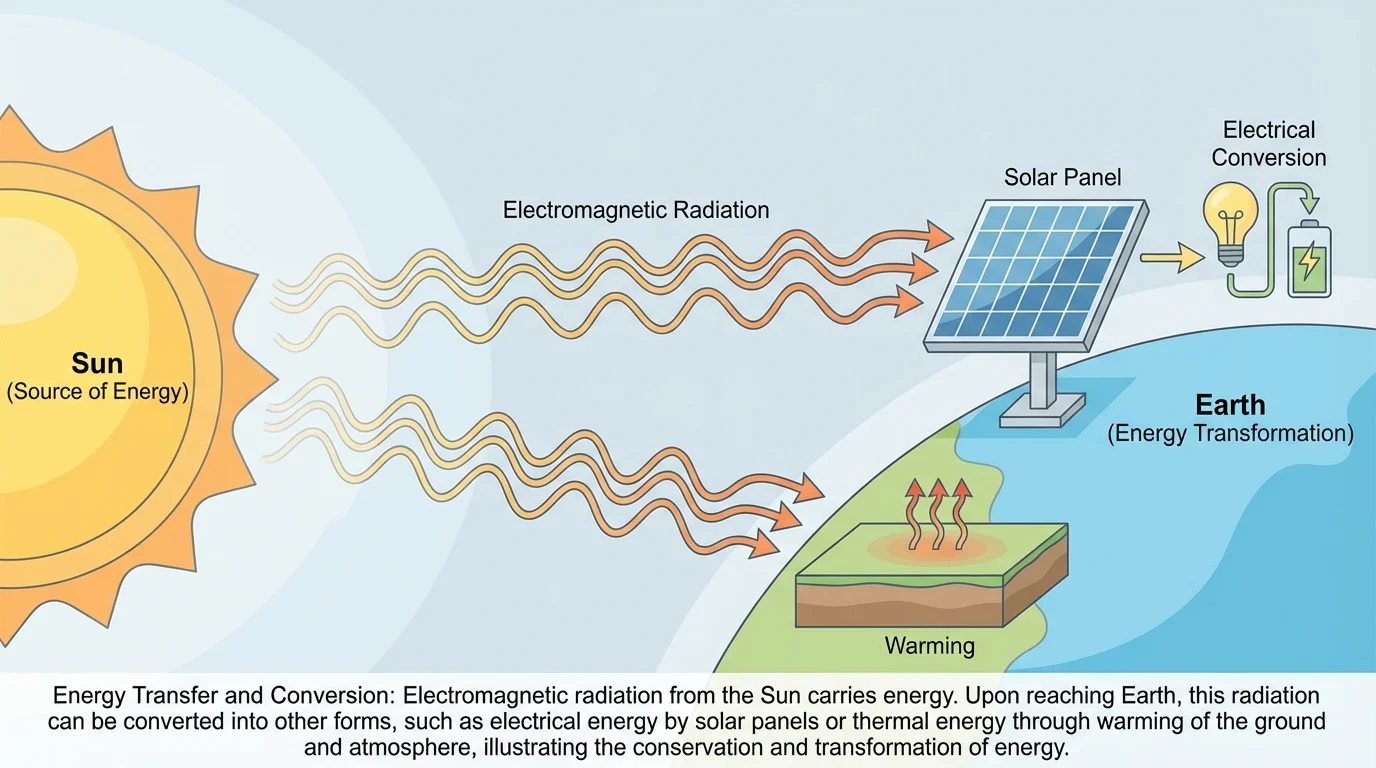
Unlike sound, which requires matter to vibrate, electromagnetic radiation can travel through empty space. This is why sunlight reaches Earth even though space is nearly a vacuum. The energy is carried by changing electric and magnetic fields.
The same principle explains technologies students use every day. Wi-Fi signals, radio waves, visible light from screens, infrared remote controls, and X-rays are all forms of electromagnetic radiation. They differ mainly in wavelength, frequency, and energy.
Force and energy are not the same. A force is an interaction that can change motion, while energy is a property of a system that can be transferred or transformed. Fields help connect these ideas by describing how objects interact across space.
Thinking in terms of fields also helps avoid a common mistake: imagining energy as always sitting inside objects like a fluid in a container. Sometimes that picture is useful, but often energy belongs to the whole system of interacting objects and fields. A book held above the floor has gravitational potential energy because of the book-Earth system, not because the book alone "contains" it in isolation.
Although the concept of energy is broad, a few simple equations help make it concrete. These formulas do not cover every case, but they show how energy can be measured and tracked quantitatively.
For motion, the kinetic energy formula is
\[E_k = \frac{1}{2}mv^2\]
For gravitational potential energy near Earth's surface, a useful model is
\(E_g = mgh\)
where \(m\) is mass, \(g \approx 9.8 \textrm{ m/s}^2\), and \(h\) is height above a reference level.
Example 1: Falling backpack
A \(5 \textrm{ kg}\) backpack is on a shelf \(2 \textrm{ m}\) above the floor. How much gravitational potential energy does it have relative to the floor?
Step 1: Choose the formula
Use \(E_g = mgh\).
Step 2: Substitute values
\(E_g = (5)(9.8)(2)\)
Step 3: Calculate
\(E_g = 98 \textrm{ J}\)
The backpack has \(98 \textrm{ J}\) of gravitational potential energy relative to the floor.
If the backpack falls and air resistance is small, most of that gravitational potential energy becomes kinetic energy before impact. The total energy stays consistent, even though the form changes.
Thermal energy changes are often calculated using
\[Q = mc\Delta T\]
where \(Q\) is energy transferred as heat, \(m\) is mass, \(c\) is specific heat capacity, and \(\Delta T\) is temperature change.
Example 2: Heating water
How much energy is needed to raise the temperature of \(0.50 \textrm{ kg}\) of water by \(10^\circ \textrm{C}\)? Use \(c = 4180 \textrm{ J/(kg}\cdot\textrm{ ^\circ C)}\).
Step 1: Write the formula
\(Q = mc\Delta T\)
Step 2: Substitute values
\(Q = (0.50)(4180)(10)\)
Step 3: Calculate
\(Q = 20{,}900 \textrm{ J}\)
It takes \(20{,}900 \textrm{ J}\) of energy to produce that temperature increase.
Conservation of energy often lets us connect one state of a system to another. A roller coaster at the top of a hill has more energy associated with height; lower down, that energy is more associated with speed. Friction does not destroy energy. Instead, it transfers energy into thermal energy of the track, wheels, and air. That idea connects directly back to the system diagrams in [Figure 1].
Example 3: Speed from height
A \(1.5 \textrm{ kg}\) ball falls from rest through a height of \(4.0 \textrm{ m}\). Ignore air resistance. Find its speed just before impact.
Step 1: Use conservation of energy
Initially, \(E_g = mgh\). Just before impact, \(E_k = \dfrac{1}{2}mv^2\). Set them equal: \(mgh = \dfrac{1}{2}mv^2\).
Step 2: Simplify
The mass cancels, so \(gh = \dfrac{1}{2}v^2\), giving \(v^2 = 2gh\).
Step 3: Substitute and solve
\(v^2 = 2(9.8)(4.0) = 78.4\), so \(v = \sqrt{78.4} \approx 8.9 \textrm{ m/s}\).
The ball's speed is approximately \(8.9 \textrm{ m/s}\).
These calculations are simple versions of a much broader idea: once you know how energy is stored and transferred, you can predict motion, temperature change, and system behavior.
Energy concepts are not just classroom tools. They shape technologies, industries, and environmental decisions.
In transportation, vehicle safety depends on energy transfer. Seat belts and airbags increase the time over which a passenger's motion changes, reducing force during the transfer of kinetic energy. Brakes convert kinetic energy into thermal energy through friction. In electric vehicles, regenerative braking captures part of that energy and converts it back into electrical energy stored in the battery.
In power generation, the central challenge is often converting one form of energy into another efficiently. A hydroelectric plant uses gravitational energy of elevated water, then kinetic energy of flowing water, then rotational energy of turbines, and finally electrical energy. A fossil-fuel power plant starts with chemical energy in fuels such as methane, \(\textrm{CH}_4\), which reacts with oxygen in combustion. One simplified combustion equation is
\[\textrm{CH}_4 + 2\textrm{O}_2 \rightarrow \textrm{CO}_2 + 2\textrm{H}_2\textrm{O}\]
In that reaction, energy associated with chemical configuration changes and is transferred mainly as thermal energy, which is then used to produce electricity.
In climate science, Earth's temperature depends on energy balance. Sunlight arrives mainly as visible and ultraviolet radiation, while Earth emits mostly infrared radiation. If more energy enters than leaves, the planet warms. The radiation pathway from the Sun to Earth and then into useful technologies is exactly the kind of field-based energy movement illustrated in [Figure 3].
In biology, living things constantly transform energy. Plants convert light energy into chemical energy through photosynthesis. Animals obtain chemical energy from food and transform it into motion, thermal energy, and biochemical work. Even when a person is resting, energy is being transferred and transformed inside cells.
Efficiency and wasted energy
Engineers often talk about "wasted" energy, but the energy is not gone. It has usually been transferred into forms that are less useful for a specific purpose, often thermal energy dispersed into the surroundings. A light bulb that becomes hot is still obeying conservation of energy; it is simply not converting as much electrical energy into visible light as we might want.
Medicine also relies on energy ideas. X-rays are high-energy electromagnetic radiation used for imaging. Ultrasound uses sound waves to probe tissues. Lasers deliver controlled light energy for surgery and measurement. In every case, understanding energy transfer is essential for effectiveness and safety.
One common misunderstanding is saying energy is "used up." In ordinary speech that phrase is common, but scientifically it is misleading. What usually happens is that energy becomes spread out in forms less useful for a particular task, especially as thermal energy. A battery in a phone does not destroy energy; it transforms chemical energy into electrical, light, sound, and thermal energy.
Another misunderstanding is treating energy as identical to motion. Motion is only one manifestation. An object can have energy because of position, chemical arrangement, electric interactions, or radiation. A stretched bow, a charged capacitor, and a hot cup of soup all have energy even when nothing large-scale appears to be moving.
Students also often confuse energy with power. Energy is the amount transferred or transformed. Power is the rate at which that happens. Two devices may use the same amount of energy, but one may do it faster. Mathematically, power is
\[P = \frac{E}{t}\]
If \(100 \textrm{ J}\) is transferred in \(5 \textrm{ s}\), then \(P = \dfrac{100}{5} = 20 \textrm{ W}\). The watt, \(\textrm{W}\), is a joule per second.
Finally, students sometimes think conservation of energy means every process is reversible or equally useful. That is not true. While total energy is conserved, the quality and concentration of energy matter. A fully charged battery can power a device effectively; the same total energy spread out as low-grade thermal energy in a room is much less useful for running electronics.
| Macroscopic observation | Microscopic interpretation | Typical energy idea |
|---|---|---|
| Moving car | Ordered motion of many particles together | Kinetic energy |
| Hot metal rod | Faster random particle motion and interactions | Thermal energy |
| Compressed spring | Particles displaced from lower-energy spacing | Elastic configuration energy |
| Chemical fuel | Electron and bond arrangement in molecules | Chemical energy |
| Sunlight | Energy carried by electromagnetic fields | Radiation |
Table 1. A comparison of common large-scale energy observations with their microscopic interpretations.
Seeing these connections is the real goal. Once you understand energy as a conserved property of systems, the topic becomes much more than a list of separate formulas or forms. It becomes a way of thinking that links mechanics, heat, light, electricity, chemistry, and modern technology.