Your phone battery, the steel in a bridge, the oxygen you breathe, and the water in a glass all look completely different at the human scale. Yet each is built from atoms, and the way those atoms are arranged and held together determines almost everything we observe. One of the most powerful ideas in science is that the hidden structure of matter explains visible properties: why some substances conduct electricity, why some react violently, why some are gases and others are solids, and why chemical reactions release or absorb energy.
All ordinary matter is made of atoms. An atom is the basic unit of an element, and each element is defined by the number of protons in its nucleus. For example, gold atoms are gold because they have the proton count that defines gold, and carbon atoms are carbon because they have the proton count that defines carbon. If the number of protons changes, the element changes.
Atoms are extremely small. A single grain of sand contains an enormous number of them, and even a drop of water contains countless molecules made from atoms. Although atoms are too small to see with the naked eye, scientists have learned a great deal about their internal structure and how that structure controls chemical behavior.
Element means a pure substance made of only one kind of atom. Atom means the smallest unit of an element that still has that element's identity. Molecule means a group of atoms bonded together as a single unit.
Not all substances are single atoms. Many substances exist as molecules, such as \(\textrm{O}_2\) in oxygen gas and \(\textrm{H}_2\textrm{O}\) in water. Others form large repeating structures, such as metals or crystals. In every case, understanding matter starts with understanding atoms.
Atoms are not solid little balls. As shown in [Figure 1], they have a nucleus, a tiny dense center containing positively charged protons and uncharged neutrons, with negatively charged electrons occupying the space around it. This internal structure can be described as a charged substructure because different parts of the atom carry different electric charges.
The proton has a charge of \(+1\), the electron has a charge of \(-1\), and the neutron has a charge of \(0\). In a neutral atom, the number of electrons equals the number of protons, so the total positive and negative charges balance. For example, a neutral carbon atom has \(6\) protons and \(6\) electrons.
Most of an atom's mass is concentrated in the nucleus because protons and neutrons are much more massive than electrons. Most of the atom's volume, however, is the region occupied by electrons. That means atoms are mostly empty space, but that phrase can be misleading: the electrical forces in that space are strong and important.
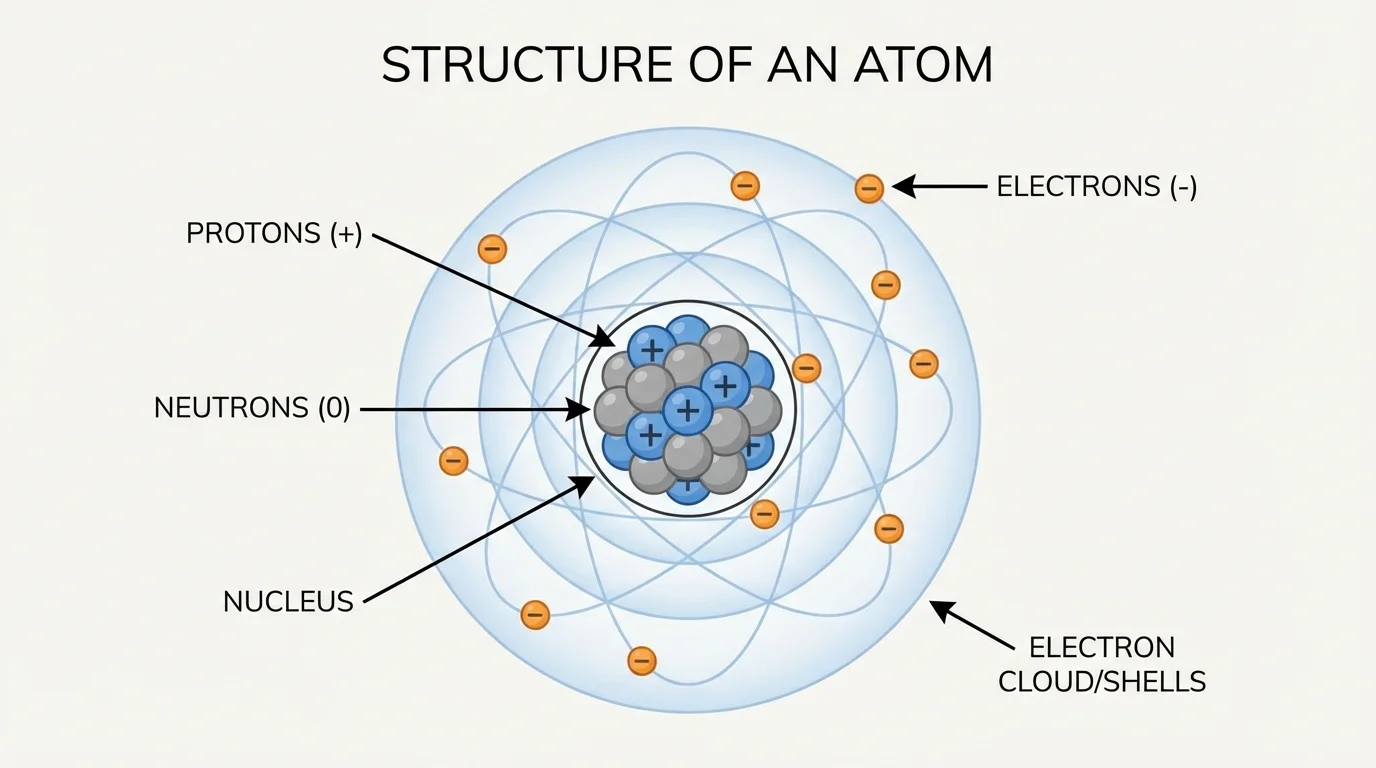
The arrangement of electrons is not random. Electrons occupy regions of space called energy levels or orbitals. At the high school level, it is often useful to think of electrons as filling outer and inner energy levels. The electrons farthest from the nucleus are especially important because they are the ones most involved in bonding and chemical reactions.
The nucleus is tiny compared with the whole atom. If an atom were enlarged to the size of a stadium, the nucleus would be only a tiny object near the center, while the electrons would occupy the much larger surrounding space.
This picture of the atom helps explain why matter behaves the way it does. The identity of the element depends on protons, the mass depends mostly on protons and neutrons, and the chemical behavior depends strongly on electrons.
The atomic number of an element is the number of protons in its nucleus. This number determines the element's position on the periodic table. Hydrogen has atomic number \(1\), helium has \(2\), lithium has \(3\), and so on. Every step to the right across the periodic table adds one proton to the nucleus.
Atoms of the same element can differ in their number of neutrons. These different forms are called isotopes. For example, carbon-12 and carbon-14 both have \(6\) protons, so both are carbon, but they have different numbers of neutrons. Isotopes usually have similar chemical behavior because chemistry depends mostly on electrons, not neutrons.
Atoms can also gain or lose electrons. When that happens, they become ions. If an atom loses electrons, it becomes positively charged. If it gains electrons, it becomes negatively charged. Sodium often forms \(\textrm{Na}^+\), while chlorine often forms \(\textrm{Cl}^-\). These ions attract each other because opposite charges attract.
Example: figuring out atomic composition
A neutral magnesium atom has atomic number \(12\) and mass number \(24\).
Step 1: Find the number of protons.
The atomic number gives the number of protons, so magnesium has \(12\) protons.
Step 2: Find the number of electrons.
Because the atom is neutral, the number of electrons equals the number of protons, so it has \(12\) electrons.
Step 3: Find the number of neutrons.
Mass number equals protons plus neutrons, so the number of neutrons is \(24 - 12 = 12\).
The atom contains \(12\) protons, \(12\) neutrons, and \(12\) electrons.
This simple calculation shows how nuclear structure and electron count can be connected. It also shows why atomic number is so central: it identifies the element itself.
As [Figure 2] shows, the periodic table organizes elements horizontally by increasing atomic number, and elements with similar chemical properties appear in vertical columns. These repeating patterns are not accidental. They reflect repeating patterns in the outer electron states of atoms.
A horizontal row is called a period. As you move from left to right across a period, each element has one more proton than the one before it. In a neutral atom, it also has one more electron. A vertical column is called a group or family. Elements in the same group tend to have similar outer electron arrangements and therefore similar chemical behavior.
For example, the alkali metals such as lithium, sodium, and potassium are all very reactive and tend to lose one electron. The halogens such as fluorine and chlorine are also reactive, but they tend to gain one electron. Noble gases such as helium, neon, and argon are much less reactive because their outer electron arrangements are especially stable.
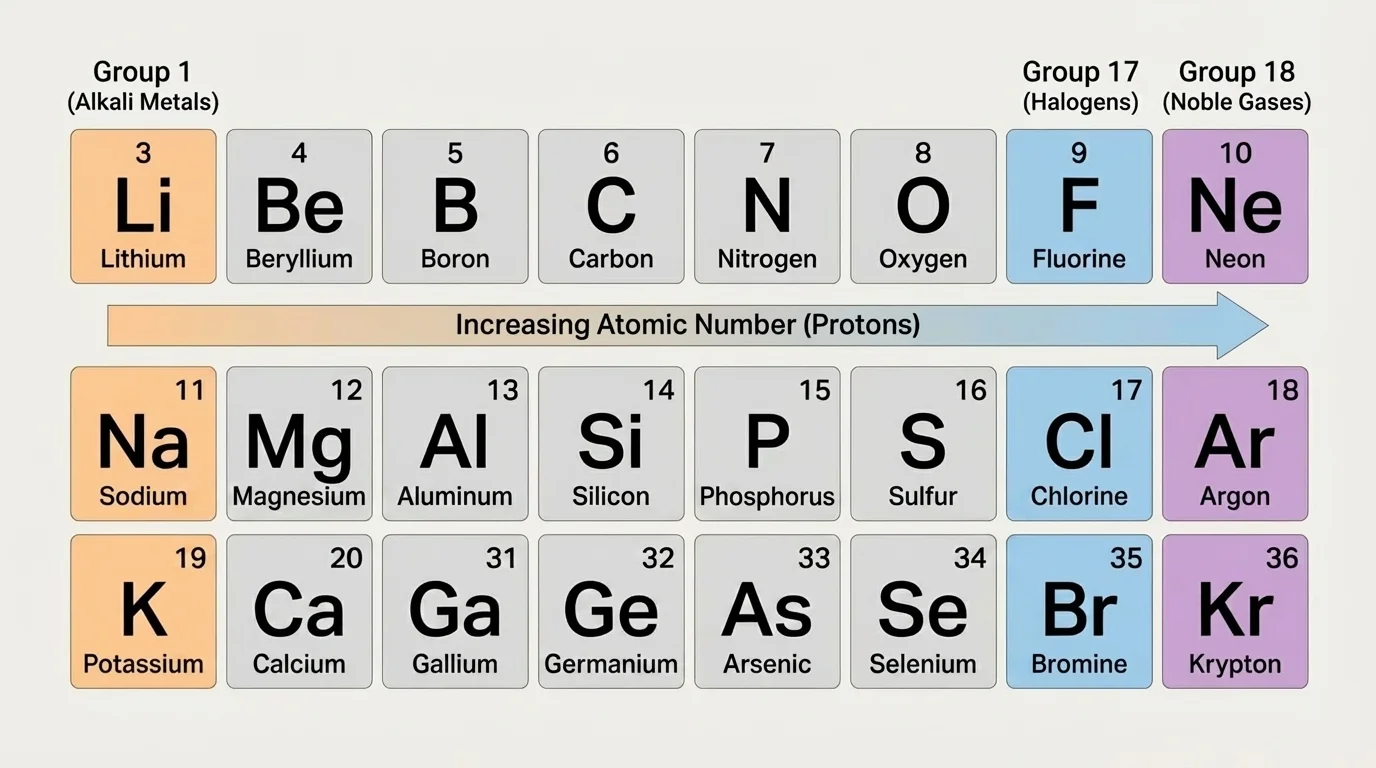
The periodic table is called "periodic" because patterns repeat. After a certain number of elements, similar electron arrangements appear again, and similar properties reappear. This is one of the great organizing ideas in chemistry: the table is not just a list of elements but a map of atomic structure and chemical behavior.
| Group | Typical outer-electron behavior | Common chemical tendency | Example elements |
|---|---|---|---|
| Alkali metals | Usually lose \(1\) electron | Form \(+1\) ions, very reactive | Lithium, sodium, potassium |
| Alkaline earth metals | Usually lose \(2\) electrons | Form \(+2\) ions | Magnesium, calcium |
| Halogens | Usually gain \(1\) electron | Form \(-1\) ions, very reactive | Fluorine, chlorine |
| Noble gases | Outer level already stable | Very low reactivity | Helium, neon, argon |
Table 1. Common periodic table families and their typical electron behavior.
Later, when predicting reactions, chemists often look first at periodic table position. Location on the table gives clues about electron arrangement, likely ion charge, and reactivity.
The electrons most involved in bonding are the valence electrons, the electrons in the outermost occupied energy level. Atoms interact in ways that often make their outer electron arrangements more stable. That is why atoms form bonds, gain electrons, lose electrons, or share electrons.
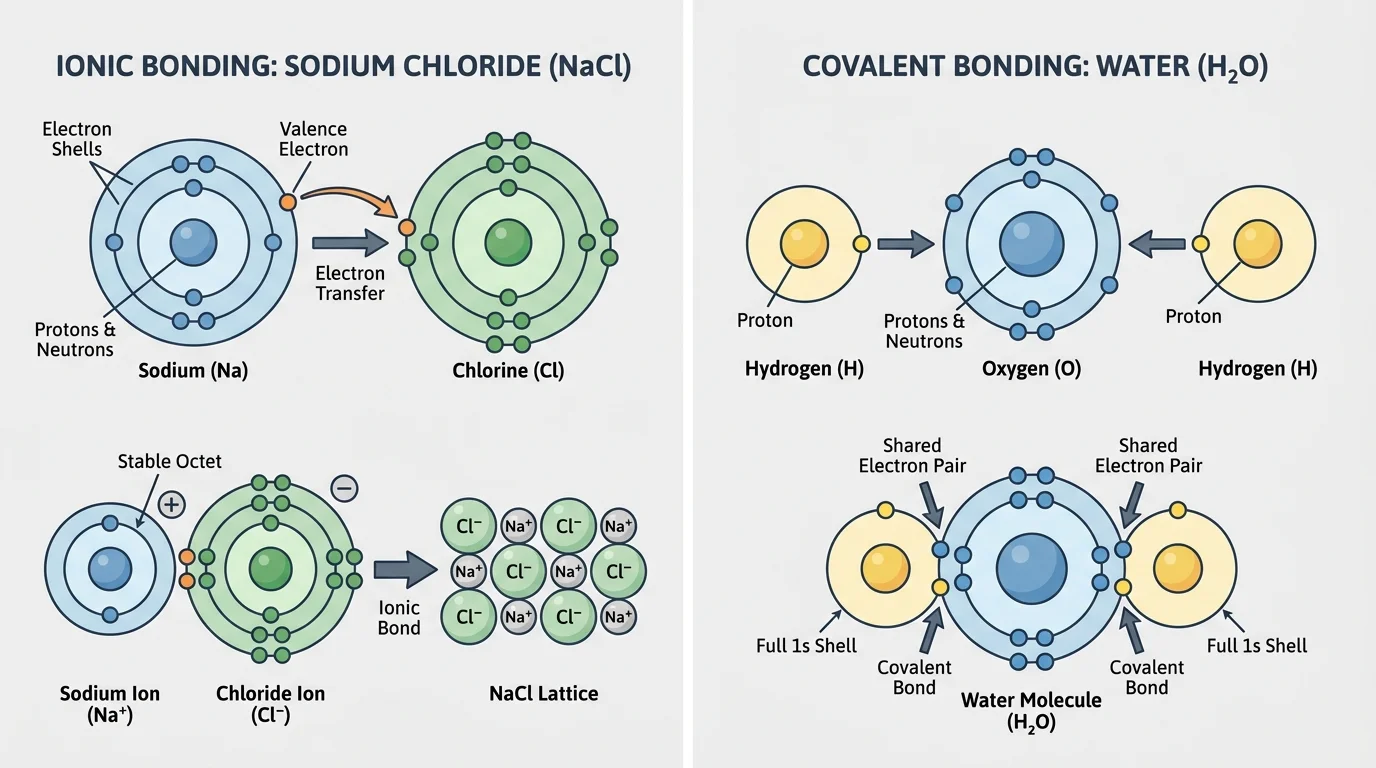
Consider sodium and chlorine. Sodium has one valence electron that it can lose relatively easily. Chlorine has seven valence electrons and tends to gain one more. When sodium transfers one electron to chlorine, they form oppositely charged ions that attract, producing \(\textrm{NaCl}\), common table salt.
Now consider oxygen and hydrogen in water. Instead of fully transferring electrons, the atoms share electrons. The oxygen atom shares electrons with two hydrogen atoms, forming the molecule \(\textrm{H}_2\textrm{O}\). This sharing creates a stable arrangement for the atoms involved.
Why outer electrons matter
Inner electrons are held more tightly by the nucleus and usually do not participate in ordinary chemical reactions. Valence electrons are farther from the nucleus and interact with neighboring atoms, so they largely determine reactivity, bonding, and many material properties.
This is why elements in the same group often behave similarly. They may differ in size or mass, but if they have the same number of valence electrons, their chemistry often follows similar patterns.
As [Figure 3] illustrates for two major bonding types, the structure of matter at the bulk scale is determined by electrical forces within and between atoms. Opposite charges attract, while like charges repel. Inside an atom, negatively charged electrons are attracted to the positively charged nucleus. Between atoms, electrical forces can pull atoms together into compounds or influence how molecules interact.
In ionic bonding, one atom transfers electrons to another, creating positive and negative ions that attract each other strongly. In covalent bonding, atoms share electrons. In metallic bonding, electrons move more freely through a network of positive atomic centers, helping explain why metals conduct electricity and can often be shaped without breaking.
Electrical forces also act between molecules. Water molecules, for example, attract each other because of uneven charge distribution within each molecule. These attractions help explain why water has a relatively high boiling point compared with many small molecules and why it is such an effective solvent.
At the large scale, these tiny electrical interactions produce familiar properties. A crystal of salt is hard and brittle because of the orderly arrangement of ions. Copper wire conducts electricity because charged particles can move through the metal. Plastic is usually a poor conductor because its electrons are not free to move easily.
When comparing materials, it is often useful to return to the bonding picture from [Figure 3]. The way charges are arranged and how tightly electrons are held helps explain conductivity, hardness, flexibility, and melting point.
As [Figure 4] shows, a chemical bond forms when a set of atoms reaches a lower-energy arrangement than the same atoms would have if they were separated. This idea is essential: a stable molecule has less energy than the same set of atoms apart from one another.
Because the bonded arrangement is lower in energy, energy must be added to pull the atoms apart. This added energy is called bond energy. Bond energy is the minimum energy needed to break a particular bond. If a molecule contains strong bonds, more energy is required to break it.
For example, breaking the bonds in \(\textrm{O}_2\) requires an energy input. During many chemical reactions, old bonds break and new bonds form. Breaking bonds requires energy, while forming bonds releases energy. Whether the total reaction gives off energy or absorbs it depends on the balance between these two processes.
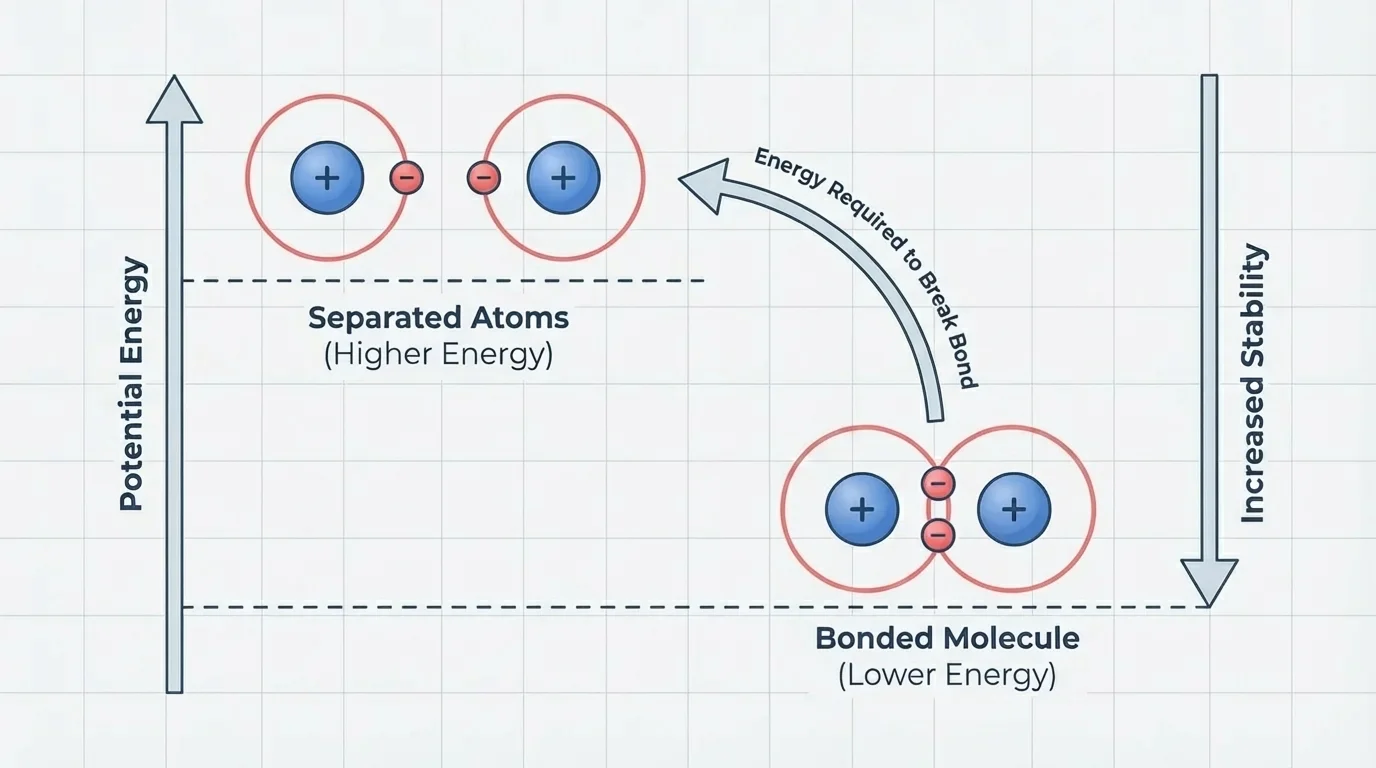
This idea helps explain why combustion reactions involving fuels can release energy. In combustion, the reactant molecules and oxygen rearrange into products such as \(\textrm{CO}_2\) and \(\textrm{H}_2\textrm{O}\). The energy released when strong new bonds form in the products is greater than the energy needed to break the original bonds, so the overall process releases energy.
Example: using bond energy as an energy threshold
Suppose a bond in a molecule requires \(250\) kilojoules per mole to break.
Step 1: Identify the meaning of the value.
This means at least \(250\) kilojoules of energy are needed to break one mole of those bonds.
Step 2: Compare an energy input.
If only \(180\) kilojoules per mole are supplied, that is less than the required amount because \(180 < 250\).
Step 3: Decide what happens.
The bond will not be fully broken by that amount alone. If \(300\) kilojoules per mole are supplied, then \(300 > 250\), so enough energy is available.
This illustrates the statement that one must provide at least the bond energy to take a molecule apart.
The energy picture in [Figure 4] is also why chemistry is not just about what substances are present, but about how stable their arrangements are.
Microscopic structure leads directly to macroscopic properties. The type of atoms present, the way their electrons are arranged, and the electrical forces between particles help determine whether a substance is solid, liquid, or gas at room temperature, whether it dissolves in water, whether it conducts electricity, and how hard or flexible it is.
For instance, diamond and graphite are both made entirely of carbon atoms, but they have very different structures. In diamond, each carbon atom is strongly bonded in a rigid three-dimensional network, making diamond extremely hard. In graphite, carbon atoms are arranged in layers that can slide past one another, making graphite soft enough to leave marks on paper.
Graphite and diamond are both pure carbon, but their different atomic arrangements give them strikingly different properties. One is among the hardest known materials, while the other is soft and can conduct electricity.
Melting point is another example. Substances with strong attractions between particles generally require more energy to melt. Ionic compounds often have high melting points because strong electrical attractions hold the ions in place. Many molecular substances have lower melting points because the forces between separate molecules are weaker.
Even gases reflect atomic-scale interactions. In a gas, particles move freely and are far apart, so attractions are weak compared with those in liquids or solids. Heating matter generally increases particle motion, and phase changes occur when energy changes the balance between particle motion and attractive forces.
These atomic ideas power modern technology. Batteries work because chemical reactions move electrons through a circuit. Semiconductors in phones and computers depend on precise control of electron behavior in solid materials. Drug molecules work because their shapes and charge distributions determine how they interact electrically with biological molecules in the body.
Materials science also relies on atomic structure. Engineers choose alloys, ceramics, polymers, and composites based on how bonding and electron arrangement influence strength, weight, heat resistance, and conductivity. A jet engine component, a surgical implant, and a solar panel each depend on matter behaving in very specific ways at the atomic level.
Example: conductivity and structure
Why does copper wire conduct electricity well while solid sodium chloride does not?
Step 1: Consider the particles in copper.
In metallic copper, some electrons are able to move through the material.
Step 2: Consider the particles in solid sodium chloride.
In solid \(\textrm{NaCl}\), ions are locked into a rigid crystal structure.
Step 3: Connect motion of charge to conductivity.
Electric current requires moving charged particles. Copper has mobile charge carriers, but solid \(\textrm{NaCl}\) does not.
This is why copper is widely used for wiring, while salt crystals are not.
Chemical energy is another major application. Fuels, foods, and batteries all involve energy stored in chemical arrangements. Understanding that stable products can be lower in energy than reactants helps explain why some reactions power cars, living cells, and electric devices.
You can observe electrical effects in matter with a simple static electricity investigation. Rub a balloon on dry hair or wool, then bring it near tiny bits of paper. The balloon attracts the paper because rubbing transfers electrons, creating an imbalance of charge. This demonstrates that electrical forces operate at small scales but produce visible effects at larger scales.
You can also compare substances qualitatively by asking which are conductors and which are insulators in a classroom demonstration with proper equipment and supervision. Metals usually conduct because charged particles can move, while rubber and plastic usually resist charge motion. These observations connect directly to atomic structure and electron behavior.
Charges follow a simple rule: opposite charges attract and like charges repel. That one rule helps explain atomic structure, bonding, molecular interactions, and many bulk properties of matter.
From the smallest nucleus to the largest engineered material, the same central idea keeps reappearing: structure determines properties. When we understand the particles, charges, and energies involved, many features of matter become not just observable, but explainable.