A tiny difference in the outermost electrons of atoms can determine whether a metal shines, whether a gas burns, or whether two substances react violently the instant they touch. Chemistry often looks complicated because there are many substances, but the logic behind many simple reactions is surprisingly organized. If you know what the outermost electrons are doing, where an element sits on the periodic table, and what patterns similar elements usually follow, you can build a strong explanation for why a reaction produces the products it does.
A chemical reaction is not just substances "mixing." In a reaction, atoms are rearranged into new combinations. The atoms themselves are conserved, but the connections between them change. Bonds in reactants are broken, new bonds form, and the result is a set of products with different properties.
To explain the outcome of a reaction, scientists ask questions such as: Which atoms are likely to lose electrons? Which are likely to gain or share them? Which combinations lead to more stable outer electron arrangements? What periodic trends support that prediction? These questions help turn a reaction from a fact to memorize into a process you can reason through.
Atoms contain a nucleus of protons and neutrons, with electrons outside the nucleus. The electrons farthest from the nucleus are especially important in chemical behavior because they are the ones involved in bonding.
For many main-group elements, the most useful first step is to look at the electrons in the outermost energy level. Those electrons often determine whether the atom reacts at all, what type of bond it tends to form, and what products are likely to appear.
The valence electrons, as shown in [Figure 1], are the electrons in the outermost occupied energy level of an atom. In main-group elements, the number of valence electrons is closely related to the element's group on the periodic table. These electrons help predict whether an atom tends to gain, lose, or share electrons in a reaction.
Atoms often react in ways that produce a more stable outer electron arrangement. A useful pattern is that many atoms become more stable when their outermost level is filled. Noble gases already have filled outer levels, so they are much less reactive than most other elements. Sodium has one valence electron, chlorine has seven, and oxygen has six. Those numbers strongly affect how each element reacts.
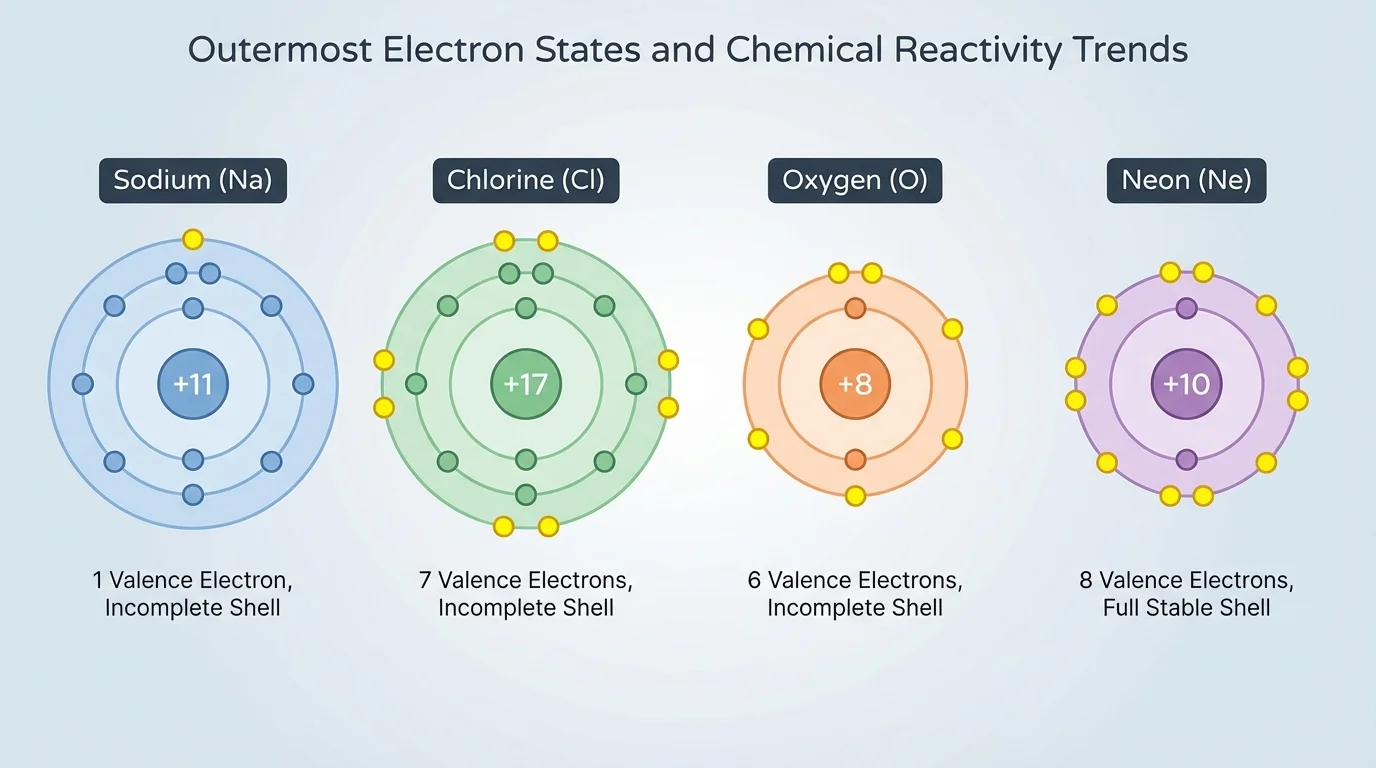
For example, sodium tends to lose one electron. Chlorine tends to gain one electron. Oxygen often gains two electrons or shares two electrons in covalent bonding. These patterns are not random. They reflect how close each atom is to a filled outer shell.
Valence electrons are the electrons in the outermost occupied energy level of an atom and are the main electrons involved in bonding. Stability in this context means an electron arrangement that is lower in energy and less likely to change.
It is important to be careful with language here. Atoms do not "want" anything in a human sense. Instead, reactions occur because certain electron arrangements are energetically more favorable. The language of "tendency" is a shortcut for that idea.
The periodic table is more than a list of elements. It is a map of repeating chemical behavior. Those repeating patterns, shown in [Figure 2], help us predict reaction outcomes. Elements in the same group often have similar valence electron patterns and therefore similar chemical properties.
Group 1 elements such as lithium, sodium, and potassium each have one valence electron. They tend to lose that electron and form ions with a charge of \(+1\). Group 2 elements such as magnesium and calcium tend to lose two electrons and form ions with a charge of \(+2\). Group 17 elements such as fluorine and chlorine tend to gain one electron and form ions with a charge of \(-1\). Group 16 elements such as oxygen and sulfur often gain two electrons or share electrons in covalent compounds.
Across a period from left to right, elements generally change from metallic to nonmetallic behavior. Metals usually lose electrons more easily, while nonmetals are more likely to gain or share electrons. Down a group, reactivity trends also appear. For example, alkali metals generally become more reactive down the group because their outer electron is farther from the nucleus and is easier to remove.
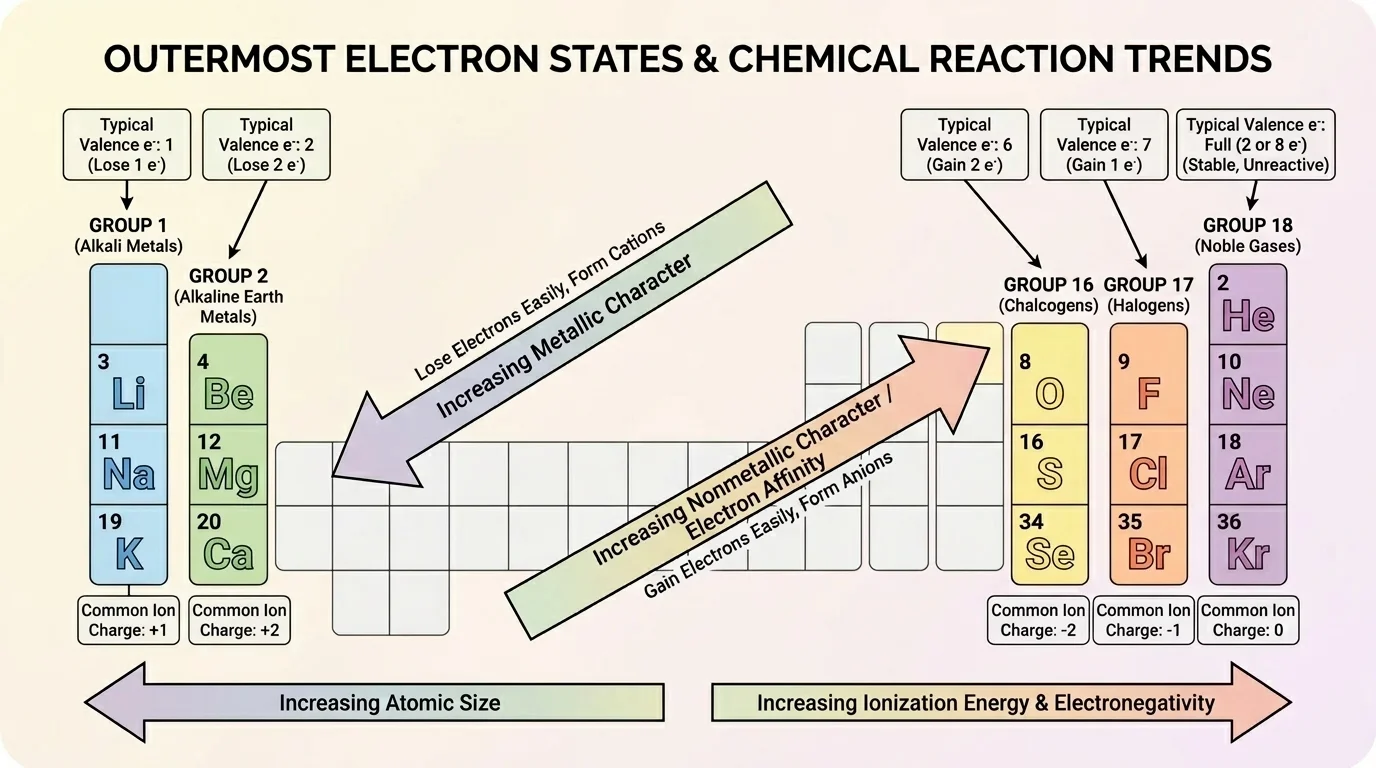
These trends do not replace evidence from actual reactions, but they give a strong starting point for explanation. If a reaction involves sodium and chlorine, the periodic table suggests electron transfer is likely. If a reaction involves oxygen and hydrogen, sharing electrons in molecules is more likely than forming a giant ionic lattice.
| Group | Example element | Valence electrons | Common behavior in simple reactions |
|---|---|---|---|
| Group 1 | Sodium \(\textrm{Na}\) | \(1\) | Loses \(1\) electron |
| Group 2 | Calcium \(\textrm{Ca}\) | \(2\) | Loses \(2\) electrons |
| Group 16 | Oxygen \(\textrm{O}\) | \(6\) | Gains \(2\) or shares \(2\) |
| Group 17 | Chlorine \(\textrm{Cl}\) | \(7\) | Gains \(1\) electron |
| Group 18 | Neon \(\textrm{Ne}\) | \(8\) | Usually unreactive |
A reaction occurs when particles collide in ways that allow old bonds to break and new bonds to form. Not every collision causes a reaction. The particles must collide with enough energy and in a useful orientation. This is why temperature, concentration, and surface area can affect reaction rate, even when the final products stay the same.
When explaining outcomes, it helps to separate two ideas: what forms and how fast it forms. The reaction outcome depends on which rearrangements are possible and energetically favorable. The rate depends on how often and how effectively particles collide.
Atoms are conserved in every chemical reaction. For example, in the reaction \[2\textrm{H}_2 + \textrm{O}_2 \rightarrow 2\textrm{H}_2\textrm{O}\] there are \(4\) hydrogen atoms and \(2\) oxygen atoms before the reaction and the same numbers after the reaction. The atoms are not created or destroyed; they are simply rearranged into new combinations.
Breaking bonds and forming bonds
Bond breaking requires energy, while bond formation releases energy. A reaction can release energy overall if the energy released when new bonds form is greater than the energy required to break the original bonds. This helps explain why some reactions, especially combustion reactions, give off heat and light.
This idea is essential when revising an explanation. If an equation is balanced but the proposed product does not fit electron patterns or known bonding behavior, the explanation is not strong enough. A good scientific explanation must match conservation of atoms, electron behavior, and observed chemical properties.
[Figure 3] The reaction between sodium and chlorine is a classic example of using electron states and periodic trends together. Sodium is a metal in Group 1, so it tends to lose one valence electron. Chlorine is a nonmetal in Group 17, so it tends to gain one electron. Those tendencies suggest that electron transfer is likely.
When sodium loses one electron, it becomes a positive ion: \(\textrm{Na}^+\). When chlorine gains one electron, it becomes a negative ion: \(\textrm{Cl}^-\). Opposite charges attract, so the ions form an ionic compound, sodium chloride, \(\textrm{NaCl}\).
The balanced reaction is \[2\textrm{Na} + \textrm{Cl}_2 \rightarrow 2\textrm{NaCl}\]. This equation shows that elemental chlorine exists as diatomic molecules, \(\textrm{Cl}_2\), before the reaction. The reaction outcome makes sense because each sodium atom transfers one electron, and each chlorine atom accepts one electron.
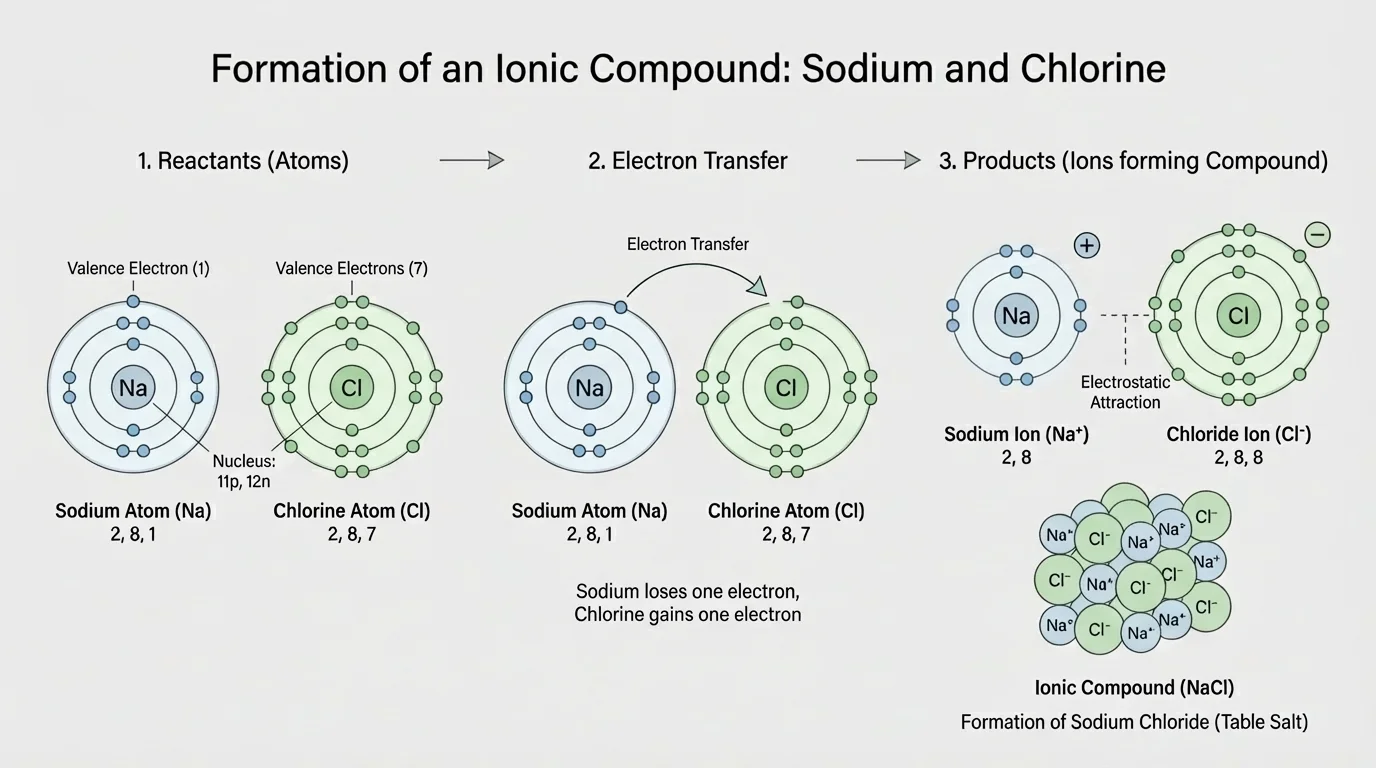
This explanation is stronger than simply saying "sodium and chlorine make salt." It identifies the role of valence electrons, uses periodic table group patterns, and connects the product to electrostatic attraction between ions. It also explains why sodium chloride has very different properties from the elements it came from. Metallic sodium is soft and highly reactive, chlorine is a toxic gas, and sodium chloride is a stable crystalline solid.
Case study: explaining why \(\textrm{MgO}\) forms
Consider magnesium reacting with oxygen.
Step 1: Use periodic patterns
Magnesium is in Group 2, so it tends to lose \(2\) electrons. Oxygen is in Group 16, so it tends to gain \(2\) electrons.
Step 2: Match electron transfer
One magnesium atom can supply exactly the \(2\) electrons that one oxygen atom tends to gain. That suggests a \(1{:}1\) ratio.
Step 3: Write the product and balanced equation
The likely product is \(\textrm{MgO}\). Because oxygen starts as \(\textrm{O}_2\), the balanced equation is \[2\textrm{Mg} + \textrm{O}_2 \rightarrow 2\textrm{MgO}\]
The explanation is based on electron transfer and periodic trends, not on memorization alone.
Later, when you compare this with water formation, [Figure 1] still matters because oxygen's six valence electrons help explain both ionic and covalent behavior. The kind of atom oxygen reacts with determines whether electrons are transferred or shared.
Molecular rearrangements, shown in [Figure 4], explain many reactions between nonmetals. In these reactions, atoms usually share electrons rather than transfer them completely. The result is a covalent compound or molecule.
Hydrogen and oxygen provide a good example. Hydrogen has one valence electron and needs one more to fill its first energy level. Oxygen has six valence electrons and tends to share two electrons to complete its outer shell. In water, \(\textrm{H}_2\textrm{O}\), oxygen shares electrons with two hydrogen atoms.
The balanced reaction is \[2\textrm{H}_2 + \textrm{O}_2 \rightarrow 2\textrm{H}_2\textrm{O}\]. The products are not predicted by conservation alone. You also need bonding patterns: hydrogen usually forms one bond, while oxygen usually forms two. Those patterns support water as the product.
Combustion reactions are another major pattern within this topic. In a combustion reaction, a substance reacts with oxygen and often releases a large amount of energy. For hydrocarbons such as methane, \(\textrm{CH}_4\), complete combustion usually produces carbon dioxide and water: \[\textrm{CH}_4 + 2\textrm{O}_2 \rightarrow \textrm{CO}_2 + 2\textrm{H}_2\textrm{O}\]
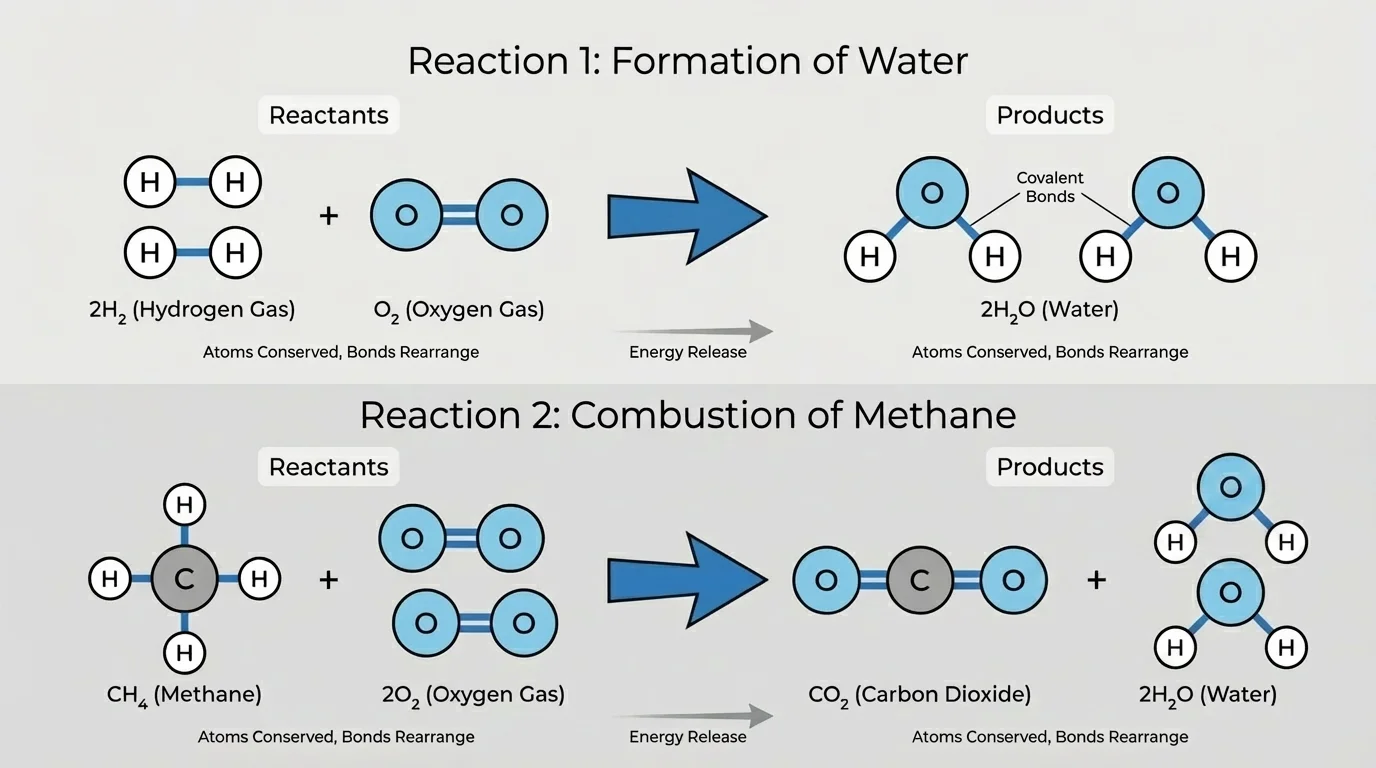
Why these products? Carbon in methane bonds with hydrogen at the start, but in the reaction the atoms rearrange into products that fit common bonding patterns and are more stable in the presence of oxygen. Carbon dioxide has carbon bonded to two oxygen atoms, and water has oxygen bonded to two hydrogen atoms. The atoms are conserved, but the bonds and energy change.
Natural gas used for heating is mostly methane. The same electron and bonding ideas that explain a classroom combustion equation also explain how home heating systems and many power plants release useful energy.
Combustion also helps explain why some reactions release heat and light so strongly. Breaking the original \(\textrm{C-H}\) and \(\textrm{O=O}\) bonds requires energy, but forming the strong bonds in \(\textrm{CO}_2\) and \(\textrm{H}_2\textrm{O}\) releases even more, so the overall reaction is energy-releasing.
Science is not just about making an explanation once. It is also about improving that explanation when evidence shows a weakness. To revise an explanation means to change it so that it better fits observations, data, and scientific principles.
Suppose a student says that sodium and chlorine react because "metals always combine with gases." That statement is too vague. A revised explanation should include the evidence from group trends and valence electrons: sodium loses one electron, chlorine gains one electron, and the resulting ions attract to form \(\textrm{NaCl}\), as we saw in [Figure 3].
Or suppose someone predicts that hydrogen and oxygen form \(\textrm{HO}\) because there is one hydrogen and one oxygen in the reactants. That explanation fails because it ignores known bonding patterns and the fact that hydrogen and oxygen begin as \(\textrm{H}_2\) and \(\textrm{O}_2\). A better explanation uses valence electrons and molecular bonding to justify \(\textrm{H}_2\textrm{O}\), as illustrated earlier in [Figure 4].
How a weak explanation becomes stronger
A weak claim: "Methane burns because fire makes it disappear."
Step 1: Keep conservation of atoms
Carbon, hydrogen, and oxygen atoms must still be present after the reaction. They do not disappear.
Step 2: Use known product patterns
For complete combustion of a hydrocarbon, carbon usually ends up in \(\textrm{CO}_2\) and hydrogen usually ends up in \(\textrm{H}_2\textrm{O}\).
Step 3: Include energy and bond changes
The reaction releases energy because new bonds in the products are more stable overall than the bonds in the reactants.
A stronger explanation is that methane reacts with oxygen through collisions that allow atoms to rearrange into \(\textrm{CO}_2\) and \(\textrm{H}_2\textrm{O}\), releasing energy in the process.
Good revision often means adding one of three missing pieces: electron behavior, periodic patterns, or evidence from observed products and properties. The best explanations combine all three.
These ideas are not limited to textbook equations. They help explain why table salt forms, why fuels burn, why metals corrode, and why some substances are stored carefully to prevent dangerous reactions.
Corrosion often involves reactions with oxygen. Magnesium, for example, reacts readily because metals on the left side of the periodic table tend to lose electrons more easily, a pattern connected to [Figure 2]. Some metals form protective oxide layers, while others continue reacting more extensively.
Combustion chemistry is central to engines, heating systems, and power generation. The same bond-rearrangement logic from [Figure 4] explains why fuels with many \(\textrm{C-H}\) bonds can release large amounts of energy when they react with oxygen.
Why product properties can be so different
One of the most powerful clues in chemistry is that products often behave nothing like the reactants. This happens because properties depend on structure and bonding. Ionic solids, small covalent molecules, and elemental metals have very different particle arrangements and therefore different melting points, conductivity, and reactivity.
Even everyday antacids connect to these patterns. Compounds containing metals from Groups 1 or 2 often form ionic substances, and those ionic compounds can react with acids in predictable ways because the identities and charges of the ions matter.
A common mistake is treating the periodic table like a chart to memorize instead of a system that explains behavior. If you only memorize that \(\textrm{Na} + \textrm{Cl}\) leads to \(\textrm{NaCl}\), you may struggle when asked about magnesium and oxygen or calcium and chlorine. But if you understand valence electrons and group trends, you can reason through unfamiliar examples.
Another mistake is confusing the balanced equation with the explanation itself. Balancing tells you the atom counts work. It does not by itself explain why those products are likely. The explanation comes from electron arrangements, periodic trends, collision ideas, and known chemical property patterns.
A stronger explanation usually includes these parts: identify the valence electrons, use the periodic table to predict whether electrons are transferred or shared, name the likely product based on common chemical patterns, and connect the product to changes in bonding and energy. When those parts agree with evidence, the explanation is scientifically strong.
"Chemical reactions are stories of atoms being conserved while electrons and bonds decide the ending."
That idea captures the heart of this topic. The atoms remain, but the outermost electrons and periodic patterns guide how those atoms are rearranged into new substances.