A disposable hand warmer can heat up in your glove, while an instant cold pack becomes cold as soon as you squeeze it. Both are driven by chemical changes, but they do opposite things with energy. That difference is not random. It depends on what happens to the bonds between atoms during the reaction. At the atomic scale, chemistry is really about rearranging matter and changing how strongly atoms are connected.
When a chemical reaction happens, atoms are not created or destroyed. Instead, atoms in the reactants are rearranged into new substances called products. During this rearrangement, some bonds in the reactants must be broken, and new bonds in the products must be formed. Whether the reaction overall gives off energy or takes in energy depends on which effect is greater.
If the total energy needed to break the original bonds is less than the total energy released when new bonds form, the reaction releases energy to the surroundings. The surroundings may warm up, produce light, or push outward as expanding gases. If more energy is needed to break the original bonds than is released by forming the new ones, the reaction absorbs energy from the surroundings.
This idea helps explain why burning fuel can power cars, why some packs are used for sports injuries, and why living cells depend on carefully controlled reactions. In all of these cases, the big question is the same: are the bonds in the products, taken together, lower in energy or higher in energy than the bonds in the reactants?
From earlier chemistry, recall that atoms contain positively charged nuclei and negatively charged electrons. Chemical bonds form because electrical attractions and repulsions lead atoms to arrange themselves in more stable ways. A reaction changes that arrangement.
To understand bond energy, it helps to think about stability. A more stable arrangement usually has lower potential energy. That means substances with stronger, lower-energy bonds are often more stable than substances with weaker, higher-energy bonds. Reactions tend to move toward arrangements that are more stable, although they may still need an initial input of energy to get started.
A chemical bond is the attraction that holds atoms together in a substance. At the atomic scale, this attraction comes from electric forces involving positively charged nuclei and negatively charged electrons. In covalent bonds, atoms share electrons. In ionic compounds, oppositely charged ions attract one another. In either case, the bonded arrangement has a different energy from the separated particles.
The term bond energy refers to the energy associated with a bond. For this lesson, the essential idea is qualitative: stronger bonds generally require more energy to break, and when those strong bonds form, more energy is released. You do not need to calculate exact total bond energies here. Instead, you need to understand the pattern.
Bond energy is the amount of energy associated with a chemical bond. Breaking a bond requires an input of energy, while forming a bond releases energy.
Reaction system refers to the substances that are reacting. Surroundings are everything outside that reacting system, such as the air, a beaker, or your hand holding a hot pack.
One reason this can seem confusing is that people sometimes say a molecule "contains energy in its bonds" and stop there. That phrase is incomplete. Bonds are not like tiny batteries that always release energy when broken. In fact, breaking any bond requires energy. The energy release in a reaction comes from the formation of new bonds that are more stable than the old ones.
For example, the molecules \(\mathrm{H}_2\) and \(\mathrm{O}_2\) can react to form \(\mathrm{H}_2\mathrm{O}\). In that process, \(\mathrm{H-H}\) and \(\mathrm{O=O}\) bonds are broken, and \(\mathrm{O-H}\) bonds are formed. The overall reaction can be represented as \(2\mathrm{H}_2 + \mathrm{O}_2 \rightarrow 2\mathrm{H}_2\mathrm{O}\). The key idea is not just that bonds change, but that the new \(\mathrm{O-H}\) bonds are associated with a lower-energy, more stable arrangement overall.
A useful model of a chemical reaction begins by separating the process into two parts, as [Figure 1] illustrates: bond breaking and bond forming. This model does not show every detail of electron motion, but it clearly tracks where energy must enter and where energy can leave.
Breaking bonds always requires energy input. You can think of bonded atoms as being in an attracted arrangement. To pull them apart, energy must be supplied to overcome that attraction. This is true whether the bond is \(\mathrm{H-H}\), \(\mathrm{O=O}\), \(\mathrm{C-H}\), or another type.
Forming bonds releases energy. When atoms move into a more stable bonded arrangement, energy is transferred away from the system. Often that energy appears in the surroundings as thermal energy, but it can also appear as light or other forms depending on the reaction.
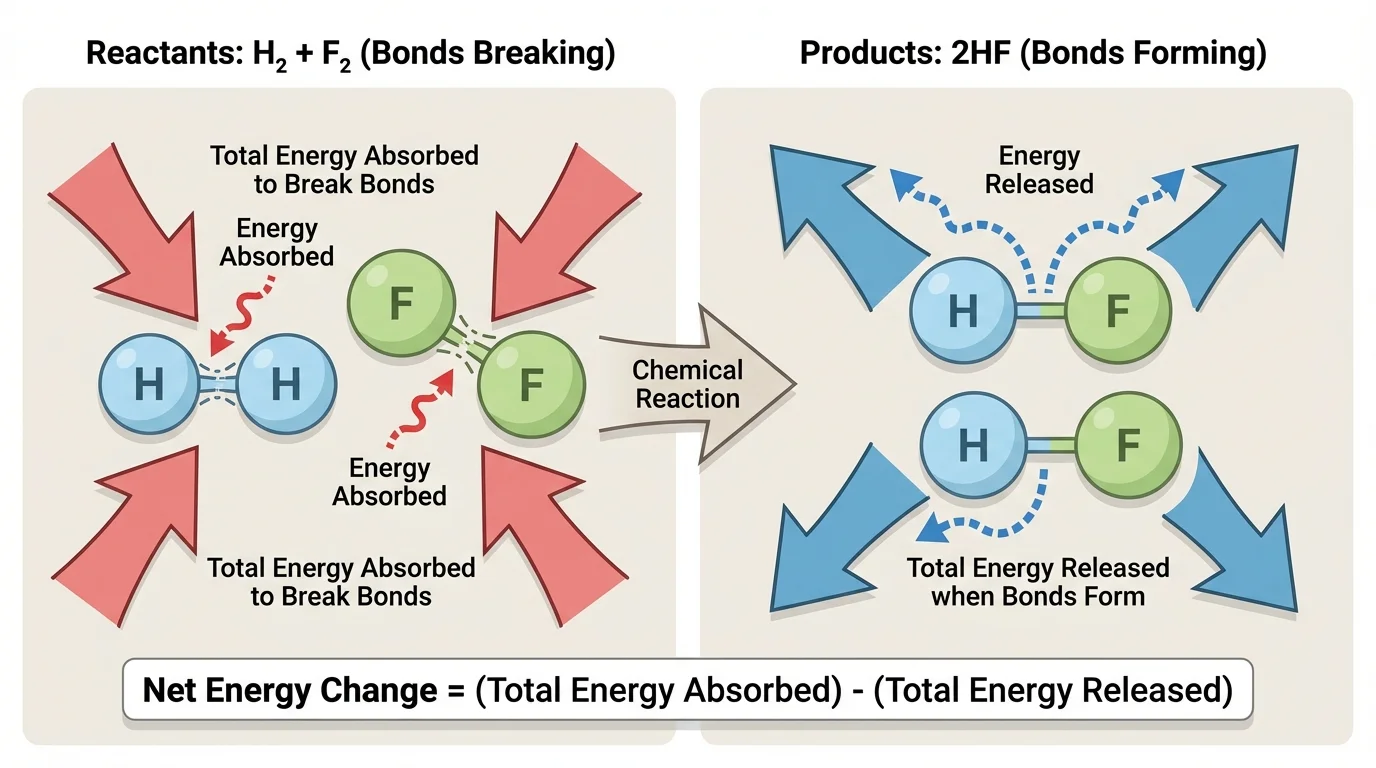
The overall energy change of a reaction depends on the balance between these two processes. If bond formation releases more energy than bond breaking absorbs, the reaction is overall energy-releasing. If bond breaking absorbs more energy than bond formation releases, the reaction is overall energy-absorbing.
This is why a reaction is not judged by looking at only one bond. You must compare the total bond energy changes in the reactants and products. Even without doing calculations, you can still model this by using phrases such as "more energy in," "less energy out," "stronger product bonds," or "higher-energy reactants."
Later examples in this lesson return to the same idea shown in [Figure 1]: no reaction releases energy simply because bonds break. The release comes from the new bonds formed being associated with a lower-energy arrangement overall.
The core energy rule
Every chemical reaction that involves covalent or ionic interactions can be modeled with the same logic: energy must be absorbed to separate bonded particles, and energy is released when new attractive interactions form. The sign of the overall energy change depends on which side of that comparison is larger.
This model fits with the idea of conservation of energy. Energy is not created by the reaction. Instead, energy is transferred between the reaction system and the surroundings as the particles rearrange into a different set of bonds.
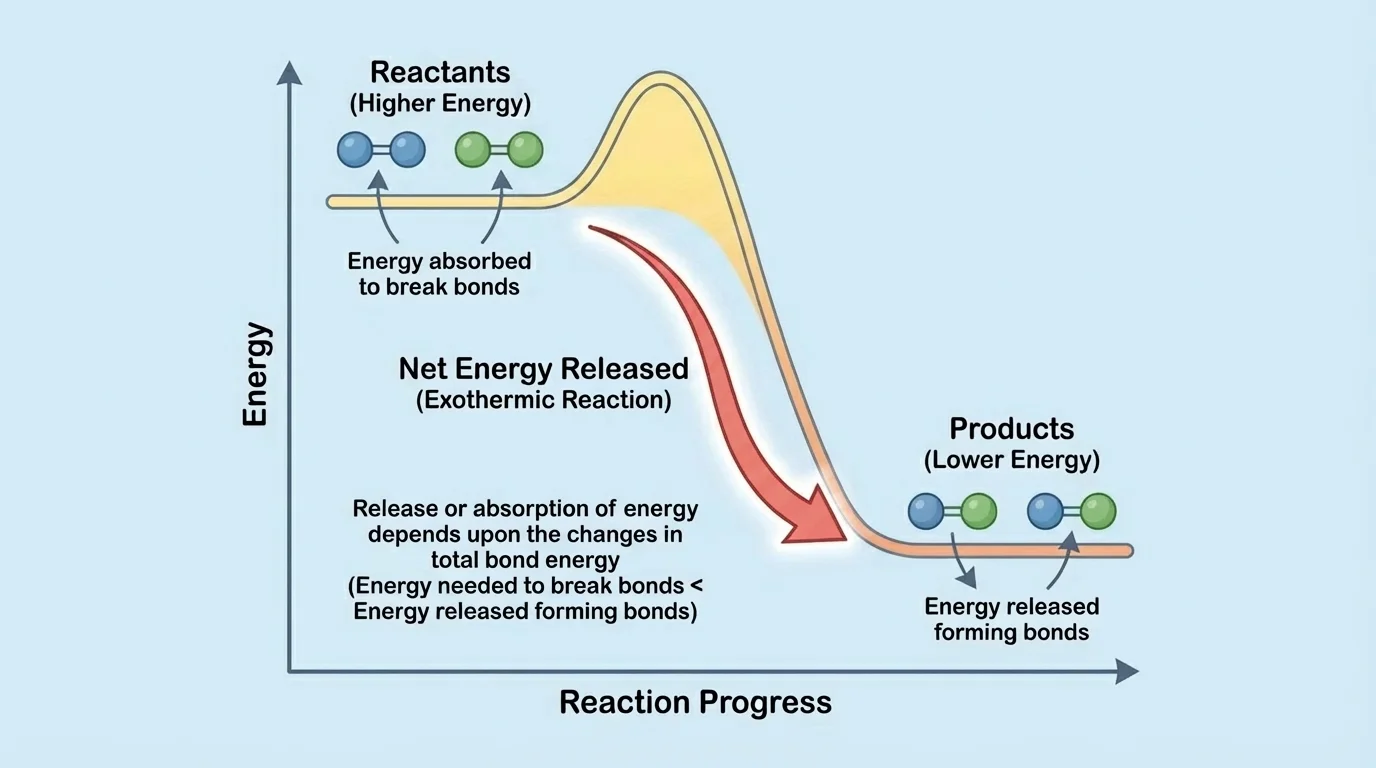
[Figure 2] The energy comparison becomes easier to see in a reaction-energy sketch for an energy-releasing reaction. In this type of model, reactants and products are placed at different relative energy levels. The exact values are not the focus; the important feature is whether the products end up lower or higher in energy than the reactants.
If the products have lower total bond energy than the reactants, energy is released to the surroundings. In that case, the products are in a more stable arrangement. This is typical of many combustion reactions, including the burning of methane: \(\mathrm{CH}_4 + 2\mathrm{O}_2 \rightarrow \mathrm{CO}_2 + 2\mathrm{H}_2\mathrm{O}\).
Combustion reactions release energy because the bonds formed in \(\mathrm{CO}_2\) and \(\mathrm{H}_2\mathrm{O}\) are collectively associated with a lower-energy state than the bonds in the fuel and oxygen molecules. Again, the lesson is not to calculate totals, but to compare them qualitatively: stronger, lower-energy product bonds can make the overall reaction release energy.
If the products have higher total bond energy than the reactants, the reaction absorbs energy from the surroundings. The system ends up in a less stable, higher-energy arrangement than where it started. Some reactions need a continuous energy supply for this reason.
A familiar biological example is photosynthesis, represented by \(6\mathrm{CO}_2 + 6\mathrm{H}_2\mathrm{O} \rightarrow \mathrm{C}_6\mathrm{H}_{12}\mathrm{O}_6 + 6\mathrm{O}_2\). This reaction requires energy from sunlight. The products include glucose, which stores energy in a form that living things can later use through other reactions.
Photosynthesis and cellular respiration are often described as opposite processes. One absorbs energy to build higher-energy molecules, and the other releases energy as those molecules are broken down and rearranged into more stable products.
The same visual logic from [Figure 2] works for both cases. If the products are lower on the energy diagram, the reaction releases energy. If the products are higher, the reaction absorbs energy.
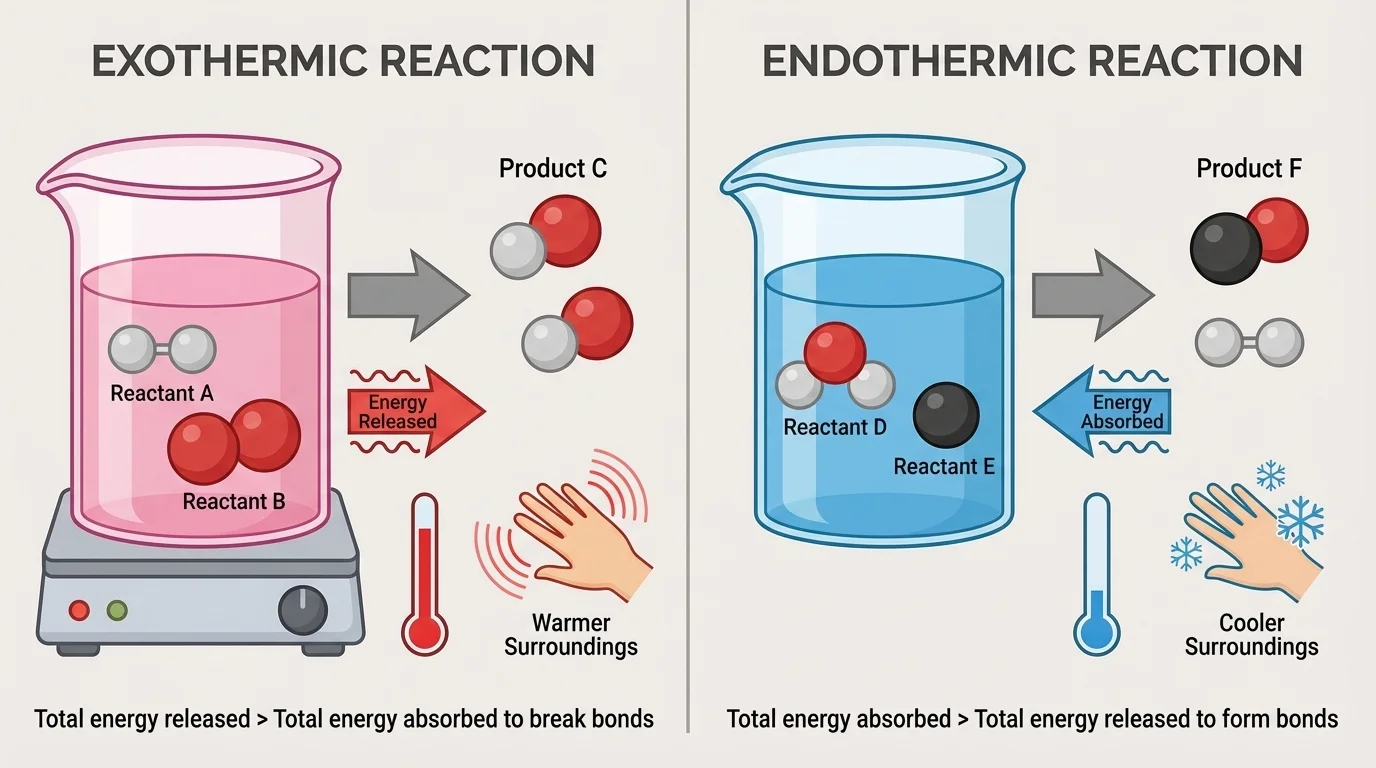
[Figure 3] The contrast between exothermic reactions and endothermic reactions is summarized visually. These two labels describe the direction of energy transfer between the reaction system and its surroundings.
An exothermic reaction releases energy to the surroundings. As a result, the surroundings usually warm up. Burning fuels, many oxidation reactions, and some kinds of neutralization reactions are exothermic. If you hold a hand warmer while its chemicals react, thermal energy is moving from the reaction system into your hand and the air.
An endothermic reaction absorbs energy from the surroundings. As a result, the surroundings often cool down. An instant cold pack works this way: when substances inside mix, the process requires an energy input, so thermal energy is taken from the surroundings.
Students often make two common mistakes here. First, they may think "exothermic" means bonds break and release energy. That is incorrect. Second, they may think a reaction that needs a spark cannot be exothermic. That is also incorrect. Many exothermic reactions need a small starting input called activation energy before they can proceed.
For example, a candle flame starts when heat from a match helps break some initial bonds. After that, the forming of new bonds in the products releases enough energy to keep the combustion going. So a reaction can require an initial trigger but still be overall exothermic.
This helps explain why energy diagrams usually show a small "hill" before products form. Even if a reaction ends at a lower energy state overall, it may need help getting started. That starting barrier does not change the rule shown earlier in [Figure 1]: bond breaking takes in energy, and bond forming releases energy.
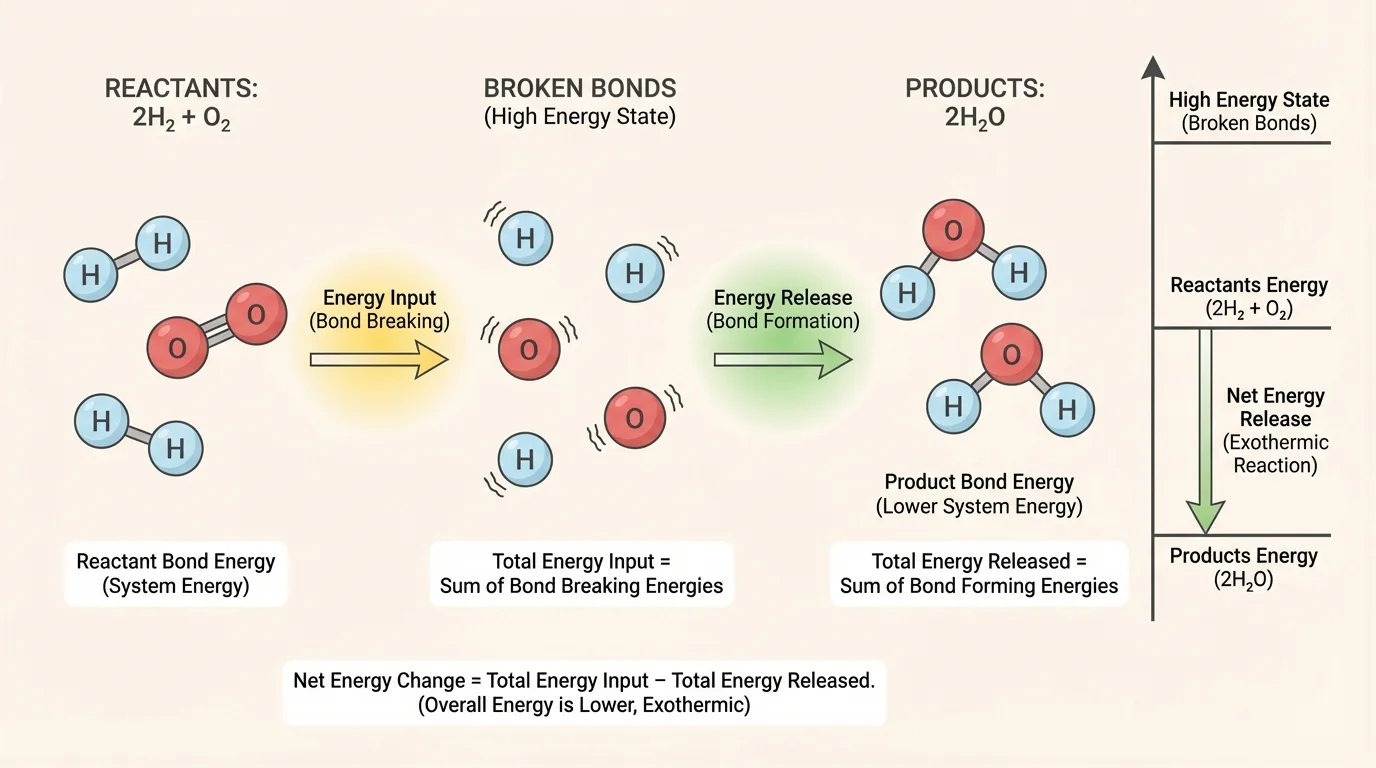
[Figure 4] A structured model helps organize what changes during a reaction. For high school chemistry, a good model does not need exact numbers. It should clearly represent the substances involved, which bonds are broken, which bonds are formed, and whether the total bond energy of the products is lower or higher than that of the reactants.
One simple approach is to use three parts. First, draw the reactant molecules and mark the bonds. Second, indicate that energy must be absorbed to break those bonds. Third, draw the product molecules and indicate that energy is released when new bonds form. Then compare the total energy absorbed with the total energy released.
Suppose you model the formation of water from hydrogen and oxygen. Your reactants are \(\mathrm{H}_2\) and \(\mathrm{O}_2\). Your products are \(\mathrm{H}_2\mathrm{O}\) molecules. In the model, you would show \(\mathrm{H-H}\) and \(\mathrm{O=O}\) bonds breaking, then \(\mathrm{O-H}\) bonds forming. If the energy released in forming the \(\mathrm{O-H}\) bonds is greater than the energy absorbed to break the original bonds, the model predicts an exothermic reaction.
Qualitative modeling example: methane combustion
Consider the reaction \(\mathrm{CH}_4 + 2\mathrm{O}_2 \rightarrow \mathrm{CO}_2 + 2\mathrm{H}_2\mathrm{O}\).
Step 1: Identify bonds in the reactants
Methane contains \(\mathrm{C-H}\) bonds, and oxygen gas contains \(\mathrm{O=O}\) bonds.
Step 2: Recognize that breaking these bonds requires energy
The system must absorb energy to separate the atoms from their original bonded arrangements.
Step 3: Identify bonds in the products
Carbon dioxide contains \(\mathrm{C=O}\) bonds, and water contains \(\mathrm{O-H}\) bonds.
Step 4: Compare the overall bond energies qualitatively
The product bonds are associated with a lower-energy, more stable arrangement overall, so more energy is released when they form than is absorbed when the reactant bonds break.
The model predicts that methane combustion is exothermic.
You can also use arrows, bars, or relative energy labels such as "higher" and "lower." What matters is not artistic detail but scientific meaning. A good model makes your reasoning visible.
When using a model, be sure to state what it explains: the release or absorption of energy depends on changes in total bond energy as atoms rearrange from reactants to products. That is the central idea the model must communicate.
Later, if you compare your own drawing to [Figure 4], check whether you included all three essential features: the original bonds, the new bonds, and the direction of overall energy transfer.
Chemistry becomes more meaningful when you connect these models to situations outside the classroom. Fuels, food, batteries, industrial materials, and living cells all depend on reactions in which bond energies change.
In cellular respiration, organisms break down glucose in reactions that ultimately produce \(\mathrm{CO}_2\) and \(\mathrm{H}_2\mathrm{O}\). Overall, these reactions release energy that cells capture and use. This is one reason food is an energy source: its atoms can be rearranged into more stable products with lower total bond energy.
In sports medicine, cold packs help reduce swelling because the process inside the pack is endothermic. The reaction absorbs thermal energy from the injured area. Heat packs do the opposite through exothermic processes, often involving oxidation of iron or crystallization in reusable packs.
In engineering and transportation, fuels work because combustion reactions form very stable product molecules such as \(\mathrm{CO}_2\) and \(\mathrm{H}_2\mathrm{O}\). Car engines, jet turbines, and rockets all depend on rapid exothermic reactions that transfer energy to moving gases.
"Energy cannot be created or destroyed, only transformed and transferred."
— Principle of energy conservation
In materials science, manufacturers control reaction conditions to manage energy transfer safely. A reaction that releases energy too quickly can overheat equipment or cause pressure buildup. A reaction that absorbs energy may require continuous heating to continue.
Even in environmental science, this idea matters. The combustion of fossil fuels releases energy that societies use, but it also produces \(\mathrm{CO}_2\), which affects Earth's climate system. Understanding bond-energy changes helps explain both the usefulness and the consequences of those reactions.
Like all scientific models, this one is useful but simplified. It focuses on bond energy changes and the overall direction of energy transfer. It does not show every electron movement, every collision between particles, or all factors that affect reaction rate.
One misconception is that a single bond determines the entire reaction. In reality, the outcome depends on the total changes across all bonds broken and all bonds formed. Another misconception is that "energy stored in bonds" means bond breaking releases that energy. A more accurate statement is that reactions can release energy when the products have lower total bond energy than the reactants.
A third misconception is that all spontaneous reactions are fast. Some are extremely slow unless conditions change. Reaction speed depends on factors such as temperature, concentration, surface area, and catalysts, not only on whether the reaction is exothermic or endothermic.
What the model should and should not claim
The model should show relative energy changes and bond rearrangements. It should not claim exact numerical totals unless data are provided and calculation is required. For this topic, the key goal is conceptual understanding, not computing total bond energy from tables.
That final point is important. You should be able to explain, for example, why forming stronger product bonds can make a reaction release energy, or why an endothermic reaction leaves products at a higher energy state. You are not expected here to calculate total bond energy changes from bond-energy values.