Your body is carrying out nonstop chemical activity. Even while you sit still, cells in your brain are using energy to send signals, muscle cells are remaining ready to act, and other cells are working to keep your temperature stable and your organs functioning. None of that happens for free. The energy that powers life does not appear out of nowhere; it is transferred through chemical reactions inside cells.
Every organism must obtain matter and energy from its environment. Plants store energy in sugars they make. Animals get food by eating other organisms. But food is not directly useful just because it exists inside the body. Cells must process those molecules in ways that make energy available for movement, transport, growth, repair, and maintaining internal balance.
That is where cellular respiration becomes essential. It is the broad chemical process by which cells transfer energy from food molecules into a more usable form. Although many details exist at the molecular level, the central idea is simpler and more powerful: atoms are rearranged, bonds are broken, new bonds are formed, and energy is transferred.
Recall that in a chemical reaction, atoms are not created or destroyed. Instead, they are rearranged. The total number of each kind of atom stays the same before and after the reaction, even though the substances change.
That principle matters here because cellular respiration is not "making energy from nothing." It is a chemical rearrangement of matter that leads to a change in how energy is stored and released.
In many cells, one important food molecule is glucose, written as \(\textrm{C}_6\textrm{H}_{12}\textrm{O}_6\). Oxygen gas, which organisms such as humans breathe in, is written as \(\textrm{O}_2\). During cellular respiration, glucose and oxygen react to form carbon dioxide and water. The overall chemical equation is:
\[\textrm{C}_6\textrm{H}_{12}\textrm{O}_6 + 6\textrm{O}_2 \rightarrow 6\textrm{CO}_2 + 6\textrm{H}_2\textrm{O} + \textrm{energy}\]
This equation is a model of the overall change. It shows the reactants on the left and the products on the right. It also shows that energy is transferred during the process. Notice that the numbers of atoms match on both sides: there are \(6\) carbon atoms, \(12\) hydrogen atoms, and \(18\) oxygen atoms before and after the reaction.
Reactants are the starting substances in a chemical reaction. Products are the new substances formed. In cellular respiration, glucose and oxygen are reactants, while carbon dioxide and water are products.
This overall equation does not list the detailed stages inside the cell, and that is appropriate here. What matters most is understanding that the process is chemical: old bonds are disrupted, atoms are rearranged, and new substances with new bonds are produced.
A molecular model helps make this idea visible. In the model shown in [Figure 1], each atom is represented as a sphere and each chemical bond as a connection between spheres. The left side contains a glucose molecule with many \(\textrm{C–C}\), \(\textrm{C–H}\), \(\textrm{C–O}\), and \(\textrm{O–H}\) bonds, along with oxygen molecules that contain \(\textrm{O=O}\) bonds.
During the reaction, those original bonds do not simply stay in place. Some are broken, and the atoms are rearranged. Carbon atoms from glucose become part of carbon dioxide molecules, where each carbon is bonded to oxygen. Hydrogen atoms from glucose become part of water molecules, where hydrogen is bonded to oxygen. The oxygen atoms in the products can be tracked to both oxygen gas and the original glucose molecule.
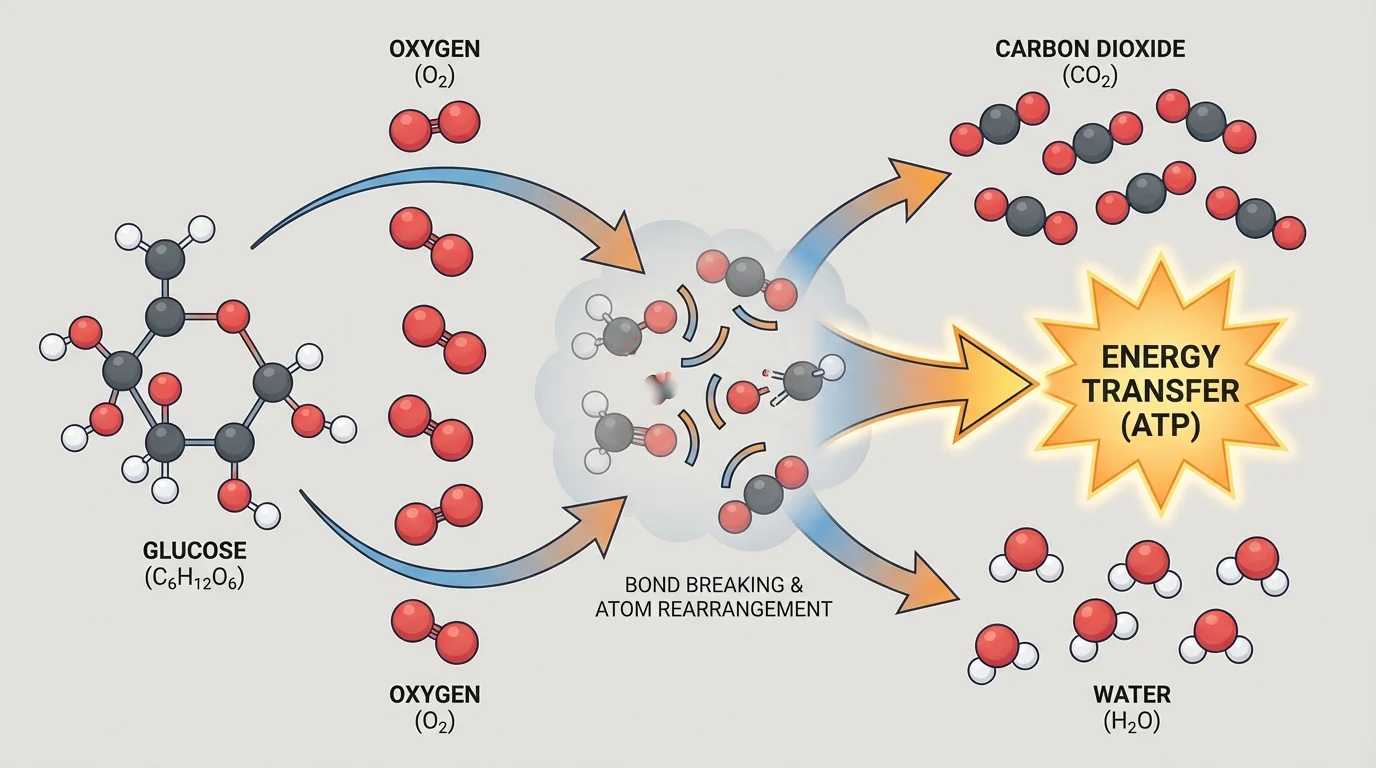
A good model does not need to show every electron or every intermediate change to be useful. It needs to capture the core idea clearly: the atoms present at the start are the same atoms present at the end, but the pattern of bonding is different. That change in bonding is what makes the reaction chemical rather than merely physical.
Using the overall equation to track atoms
Consider the reaction \(\textrm{C}_6\textrm{H}_{12}\textrm{O}_6 + 6\textrm{O}_2 \rightarrow 6\textrm{CO}_2 + 6\textrm{H}_2\textrm{O}\).
Step 1: Count the carbon atoms.
There are \(6\) carbon atoms in one glucose molecule and \(6\) carbon atoms in \(6\textrm{CO}_2\), because each \(\textrm{CO}_2\) contains \(1\) carbon.
Step 2: Count the hydrogen atoms.
There are \(12\) hydrogen atoms in glucose. On the product side, \(6\textrm{H}_2\textrm{O}\) contains \(6 \times 2 = 12\) hydrogen atoms.
Step 3: Count the oxygen atoms.
On the reactant side, glucose has \(6\) oxygen atoms and \(6\textrm{O}_2\) adds \(12\) more, for a total of \(18\). On the product side, \(6\textrm{CO}_2\) has \(12\) oxygen atoms and \(6\textrm{H}_2\textrm{O}\) has \(6\) oxygen atoms, giving \(18\) total.
The model works because matter is conserved while atoms are rearranged into new compounds.
Later, when you think about respiration in exercise or medicine, the same bond-rearrangement idea from [Figure 1] still applies. The products are not hidden versions of the reactants; they are genuinely new substances formed by a new arrangement of atoms.
A physical change might alter state or appearance, such as ice melting into liquid water, while keeping the same molecules. A chemical process changes which molecules are present. Cellular respiration clearly fits that definition because glucose and oxygen are transformed into carbon dioxide and water.
This means cellular respiration involves more than "using up food." The carbon dioxide that leaves your lungs and the water produced inside cells are chemical products. Their bonds differ from those in the original food molecules. If new substances form, then chemical bonds must have changed.
Chemical reactions rearrange atoms and energy storage
When a reaction occurs, the identity of the atoms stays the same, but the way those atoms are bonded can change dramatically. Because chemical bonds are associated with energy, a rearrangement of bonds can also change how energy is stored in matter. That is why cellular respiration is about both matter and energy at the same time.
This is also why the reaction must be considered at the molecular level. On the scale of the whole organism, you may notice breathing, body heat, or muscle movement. On the scale of molecules, those visible effects are connected to countless chemical reactions taking place in cells.
The most important energy idea is not that "breaking bonds releases energy." In fact, breaking chemical bonds requires an input of energy. The larger picture, shown by the energy model in [Figure 2], is that forming new bonds can release energy, and in cellular respiration the total energy released when product bonds form is greater than the energy required to break the original bonds.
As a result, there is a net transfer of energy. The reactants store more chemical energy than the products do, so some energy becomes available to the cell. That transferred energy can be used for cellular work and some of it also contributes to heat.
You can think of it this way: the chemical arrangement of atoms in glucose and oxygen is higher in stored energy than the arrangement in carbon dioxide and water. After the reaction, the products are more stable and lower in chemical energy. The difference in energy is transferred out of the reaction system and captured by the cell in useful ways.
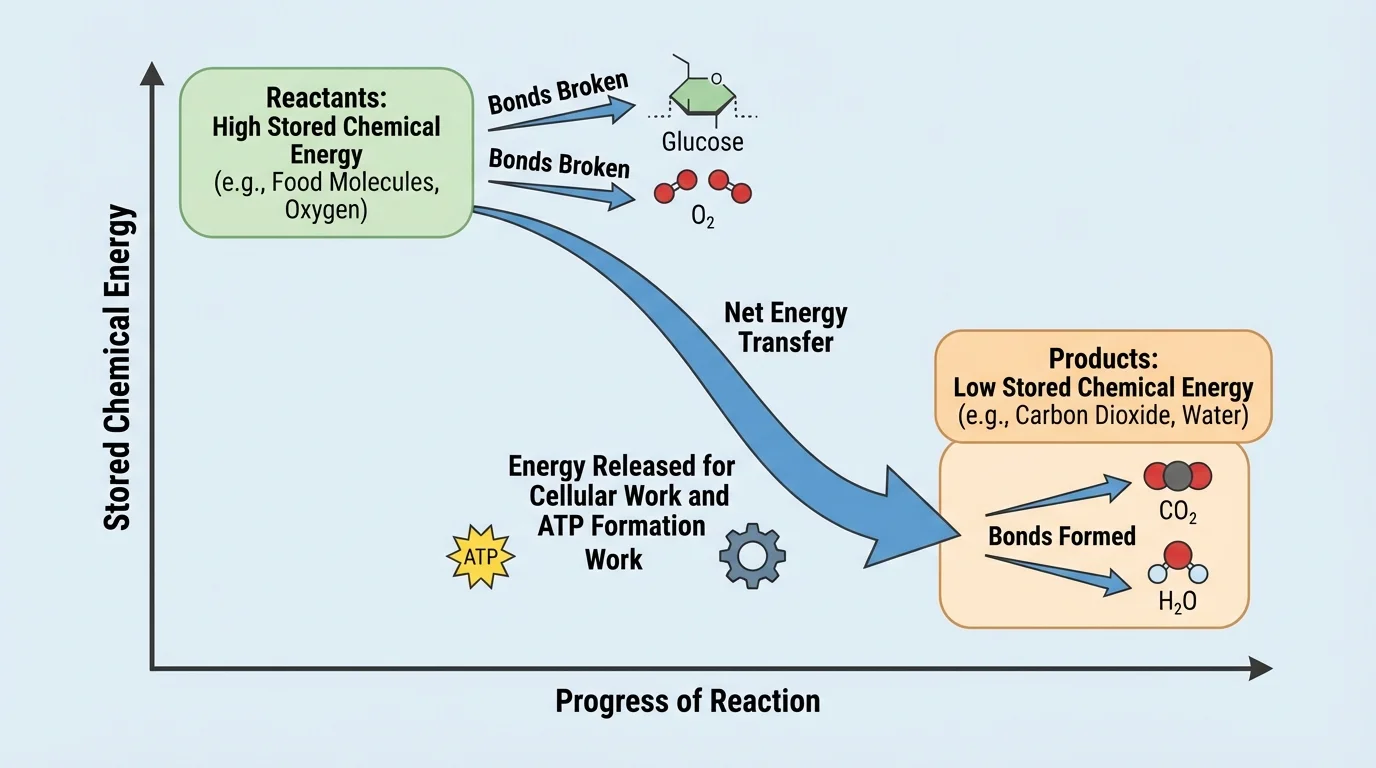
This does not violate conservation of energy. Energy is not created. Instead, energy stored in the reactant molecules is transferred into forms the cell can use. Some is used to support movement of materials, some supports building and repair, some helps create electrical gradients across membranes, and some appears as heat.
Comparing stored chemical energy
If a reaction begins with molecules at an energy level of \(100\) arbitrary units and breaking bonds requires \(30\) units, but forming new bonds releases \(60\) units, the overall change is \(60 - 30 = 30\) units released.
The exact numbers here are simplified, but the idea is accurate: bond breaking requires energy, bond forming releases energy, and the important quantity is the overall difference. In cellular respiration, that overall difference favors a release of usable energy.
When students oversimplify respiration by saying "energy comes from breaking bonds," they miss the full chemistry. The correct idea is that the entire reaction pathway leads to a lower-energy arrangement of atoms, and the difference is transferred out overall. That is why the word net matters.
Cells do not usually use the energy from food in one giant burst. Instead, they transfer much of that energy into molecules of ATP, short for adenosine triphosphate. ATP acts as an immediate energy carrier that cells can use more directly than glucose.
When cells need to power a process such as active transport across a membrane or the contraction of a muscle fiber, ATP can participate in reactions that supply the needed energy in manageable amounts. Food molecules are more like stored fuel, while ATP is more like spendable cellular energy.
Human cells use and remake enormous amounts of ATP each day. Even though your body contains only a limited amount at any one moment, that ATP is constantly recycled because life depends on a continuous flow of energy.
This helps explain why oxygen delivery matters so much in active tissues. If cells cannot continue transferring energy efficiently from food molecules, their supply of ATP becomes limited, and cell functions begin to suffer.
One powerful way to understand respiration is to track matter through the reaction. The atom-flow model in [Figure 3] traces where carbon, hydrogen, and oxygen go during cellular respiration. This reinforces that organisms do not "destroy" food; they chemically transform it.
The carbon atoms in glucose end up in carbon dioxide. That is why the carbon dioxide you exhale is connected to the food you have eaten. The hydrogen atoms from glucose are incorporated into water. Oxygen atoms from inhaled oxygen also become part of the products, especially water and carbon dioxide.
So when you breathe out, you are not just releasing waste gas in a general sense. You are releasing one of the actual chemical products of cellular respiration. The same model also shows how matter from food and matter from the air interact inside cells.
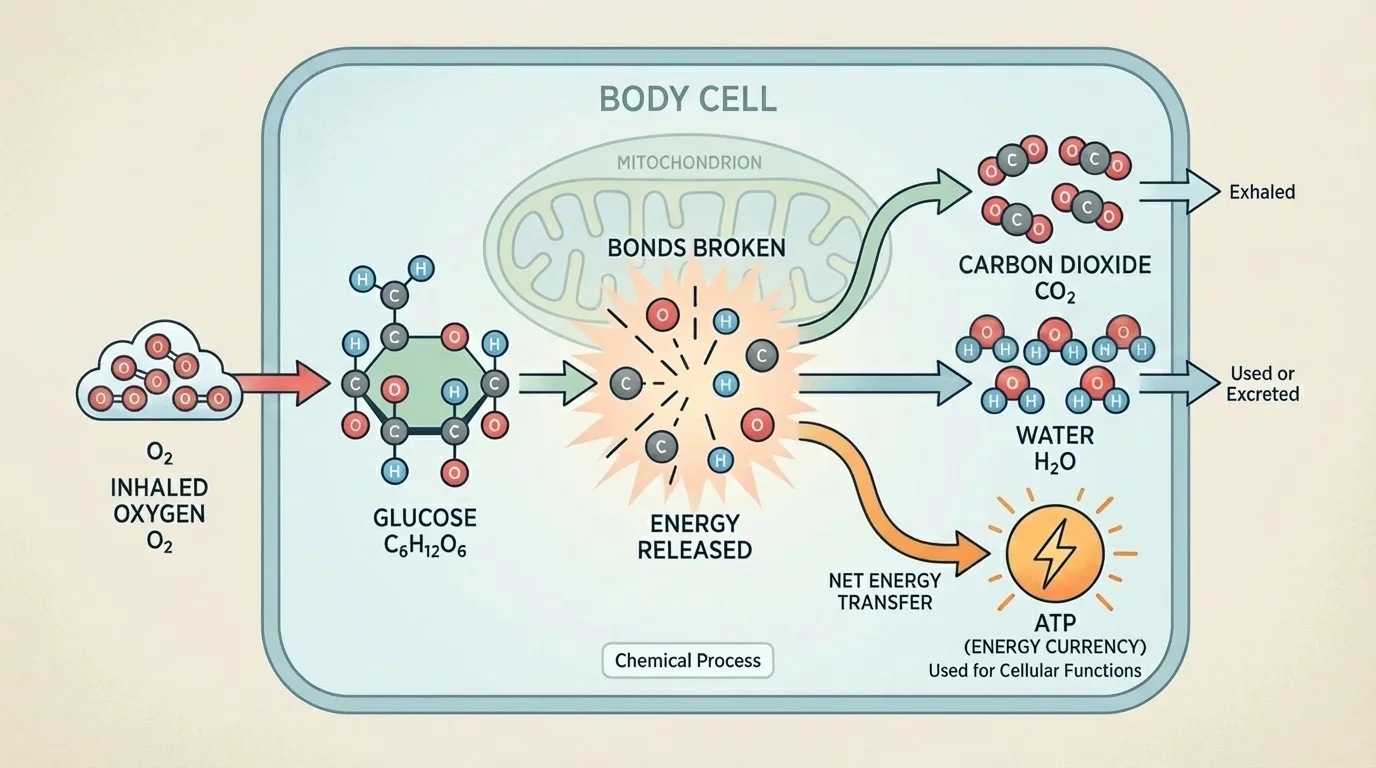
This matters for the larger theme of life science: organisms use matter and energy to live and grow. Matter from food contributes to the body in multiple ways. Some becomes part of body structures. Some is chemically processed in cellular respiration. Energy transfer from those reactions then supports the functions needed for survival.
| Atom type | Main source in reactants | Where it appears in products |
|---|---|---|
| Carbon | Glucose, \(\textrm{C}_6\textrm{H}_{12}\textrm{O}_6\) | Carbon dioxide, \(\textrm{CO}_2\) |
| Hydrogen | Glucose, \(\textrm{C}_6\textrm{H}_{12}\textrm{O}_6\) | Water, \(\textrm{H}_2\textrm{O}\) |
| Oxygen | Glucose and oxygen gas, \(\textrm{O}_2\) | Carbon dioxide and water |
Table 1. Tracking major atom types from reactants in cellular respiration to products.
When you revisit the molecular model from [Figure 1], you can see that respiration is really a story of atom bookkeeping. Nothing disappears. The atoms are simply redistributed into molecules with different properties and lower stored chemical energy.
During exercise, muscle cells need more ATP, so the demand for cellular respiration rises. Breathing rate and heart rate increase partly because cells need more oxygen delivered and more carbon dioxide removed. The chemistry inside the cells connects directly to the physical experience of running, lifting, or cycling.
In medicine, cellular respiration is relevant when doctors monitor oxygen levels, carbon dioxide levels, and metabolic function. If tissues are not receiving enough oxygen, cells may not transfer energy from food as effectively. This can affect organ function quickly, especially in the brain and heart.
Exercise and exhaled carbon dioxide
Suppose a student starts jogging and their muscles require a faster rate of ATP production. As muscle cells increase cellular respiration, they use oxygen more rapidly and produce carbon dioxide more rapidly.
Step 1: More cellular activity raises ATP demand.
Muscle contraction, ion pumping, and recovery all require ATP.
Step 2: Faster respiration increases reactant use and product formation.
Cells consume more glucose and \(\textrm{O}_2\), while producing more \(\textrm{CO}_2\) and \(\textrm{H}_2\textrm{O}\).
Step 3: The body responds.
Breathing becomes deeper or faster to take in oxygen and remove carbon dioxide.
The observable change in breathing reflects an underlying chemical process in cells.
Cellular respiration is also central to food science and agriculture. Stored sugars in crops represent chemical energy. When living plant or animal cells respire, they are continually transferring energy needed to stay alive. Even harvested produce continues respiring for a time, which can affect freshness and storage.
One common misunderstanding is that oxygen itself is the energy source. Oxygen is not the fuel. It is a reactant that participates in the chemical process. The food molecule, such as glucose, contains much of the stored chemical energy that can be transferred during respiration.
Another misunderstanding is that respiration means only breathing. Breathing is a body-level process of moving air in and out of the lungs. Cellular respiration happens inside cells. The two are related, but they are not the same thing.
"Living systems survive by managing flows of matter and transfers of energy."
A third misunderstanding is that cellular respiration must be understood by memorizing every stage name and enzyme. For this topic, the important idea is broader: respiration is a chemical process with bond changes, new compounds, and a net transfer of energy. That core model is enough to explain why it supports life.
Scientific models are simplified representations of reality. A chemical equation is a model. A ball-and-stick drawing is a model. An energy diagram is a model. Each highlights some features while leaving out others.
For example, the overall equation is excellent for showing conservation of atoms, but it does not show the three-dimensional structure of molecules. The bond model helps you see rearrangement, while the energy model in [Figure 2] makes the net transfer of energy clearer. The matter-flow model in [Figure 3] is especially useful when connecting cellular respiration to whole-organism biology, such as exhaling carbon dioxide or needing oxygen during exercise.
A strong scientific explanation often uses more than one model because no single representation captures everything. In this case, combining a reaction equation, a bond-rearrangement model, and an energy comparison gives the clearest understanding of how organisms use matter and energy to live and grow.