A single human hair is about \(7 \times 10^{-5}\) meters thick, while the distance from Earth to the Sun is about \(1.5 \times 10^8\) kilometers. Those numbers are so different in size that writing them in ordinary decimal form can be awkward and easy to misread. Scientific notation gives us a compact, powerful way to work with both extremes.
When numbers become extremely large or extremely small, long strings of zeros get in the way. Scientists, engineers, doctors, and programmers often need to compare, calculate, and communicate such numbers quickly. Scientific notation helps by showing the important digits and the size of the number at the same time.
In scientific notation, a number is written as a product of a number between \(1\) and \(10\) and a power of \(10\):
\[a \times 10^n\]
Here, \(1 \le a < 10\), and \(n\) is an integer. The number \(a\) is called the coefficient, and the exponent tells how many places the decimal point moves.
Scientific notation is a way of writing numbers as \(a \times 10^n\), where \(1 \le a < 10\) and \(n\) is an integer. A positive exponent means a large number, and a negative exponent means a small number.
If the exponent is positive, the number is greater than or equal to \(10\). If the exponent is negative, the number is between \(0\) and \(1\). This pattern connects directly to powers of ten and place value.
To write a number in scientific notation, move the decimal point until the number is at least \(1\) but less than \(10\), as [Figure 1] illustrates. Then count how many places you moved the decimal point. That count becomes the exponent on \(10\).
For a large number, the decimal moves left, so the exponent is positive. For a small decimal, the decimal moves right, so the exponent is negative.

Example conversions:
\(6{,}300{,}000 = 6.3 \times 10^6\) because the decimal moves \(6\) places left.
\(0.000081 = 8.1 \times 10^{-5}\) because the decimal moves \(5\) places right.
A fast check helps: if the original number is very large, the exponent should probably be positive. If the original number is tiny, the exponent should probably be negative.
Worked example 1
Write \(58{,}900\) in scientific notation.
Step 1: Move the decimal point so the leading number is between \(1\) and \(10\).
\(58{,}900 \rightarrow 5.89\)
Step 2: Count the number of places moved.
The decimal moves \(4\) places to the left.
Step 3: Write the result as a product.
\[58{,}900 = 5.89 \times 10^4\]
The coefficient must stay in the range \(1 \le a < 10\). So \(58.9 \times 10^3\) is equal in value, but it is not in proper scientific notation.
Powers of ten follow place-value patterns: \(10^1 = 10\), \(10^2 = 100\), \(10^3 = 1000\). Negative exponents mean reciprocals, such as \(10^{-1} = 0.1\), \(10^{-2} = 0.01\), and \(10^{-3} = 0.001\).
That connection to integer exponents is why scientific notation belongs with exponent rules. It is really a place-value shortcut built from powers of \(10\).
To convert from scientific notation to an ordinary decimal, use the exponent to decide the direction and number of decimal moves. A positive exponent moves the decimal right. A negative exponent moves it left.
For example, \(3.7 \times 10^5\) becomes \(370{,}000\). The decimal moves \(5\) places to the right. But \(4.2 \times 10^{-3}\) becomes \(0.0042\). The decimal moves \(3\) places to the left.
As seen earlier in [Figure 1], the exponent is really telling you how far the decimal point shifts. That makes converting in either direction a reversible process.
Worked example 2
Write \(9.04 \times 10^{-4}\) in decimal form.
Step 1: Notice the exponent is negative.
This means the decimal point moves left.
Step 2: Move the decimal \(4\) places.
\(9.04 \rightarrow 0.000904\)
Step 3: State the decimal form.
\[9.04 \times 10^{-4} = 0.000904\]
One common mistake is forgetting placeholder zeros. If the decimal moves beyond the first digit, zeros are needed so the place value stays correct.
Scientific notation also helps compare sizes. A number written with a factor of \(10^7\) is much larger than one written with a factor of \(10^3\), because \(10^7 = 10{,}000{,}000\) and \(10^3 = 1{,}000\). The exponents tell a story about scale.
[Figure 2] Technology often writes scientific notation in a different but related style. A calculator may display \(3.2\textrm{E}5\) instead of \(3.2 \times 10^5\). The letter E means "times ten to the power of."
So these are equivalent:
\(6.1\textrm{E}8 = 6.1 \times 10^8\)
\(4.7\textrm{E}{-3} = 4.7 \times 10^{-3}\)
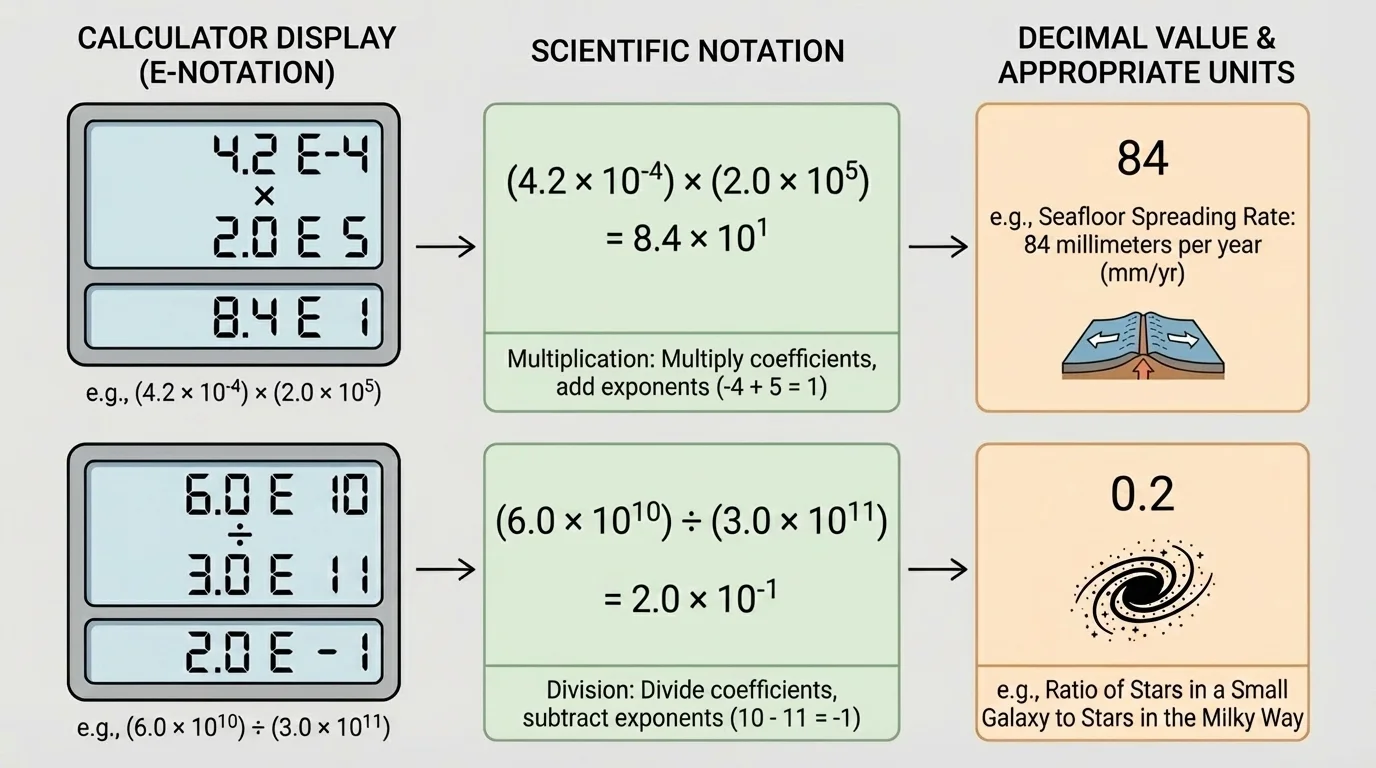
When reading technology output, pay close attention to the sign of the exponent. The difference between \(10^6\) and \(10^{-6}\) is enormous. One is a million; the other is one-millionth.
Computers often use scientific notation because it saves space and keeps calculations manageable. Without it, many scientific values would be too long to display clearly on a screen.
If two numbers have the same exponent, compare the coefficients. For example, \(5.1 \times 10^4 > 3.8 \times 10^4\) because \(5.1 > 3.8\). If the exponents are different, the exponent usually decides first. For example, \(2.0 \times 10^6 > 9.9 \times 10^5\).
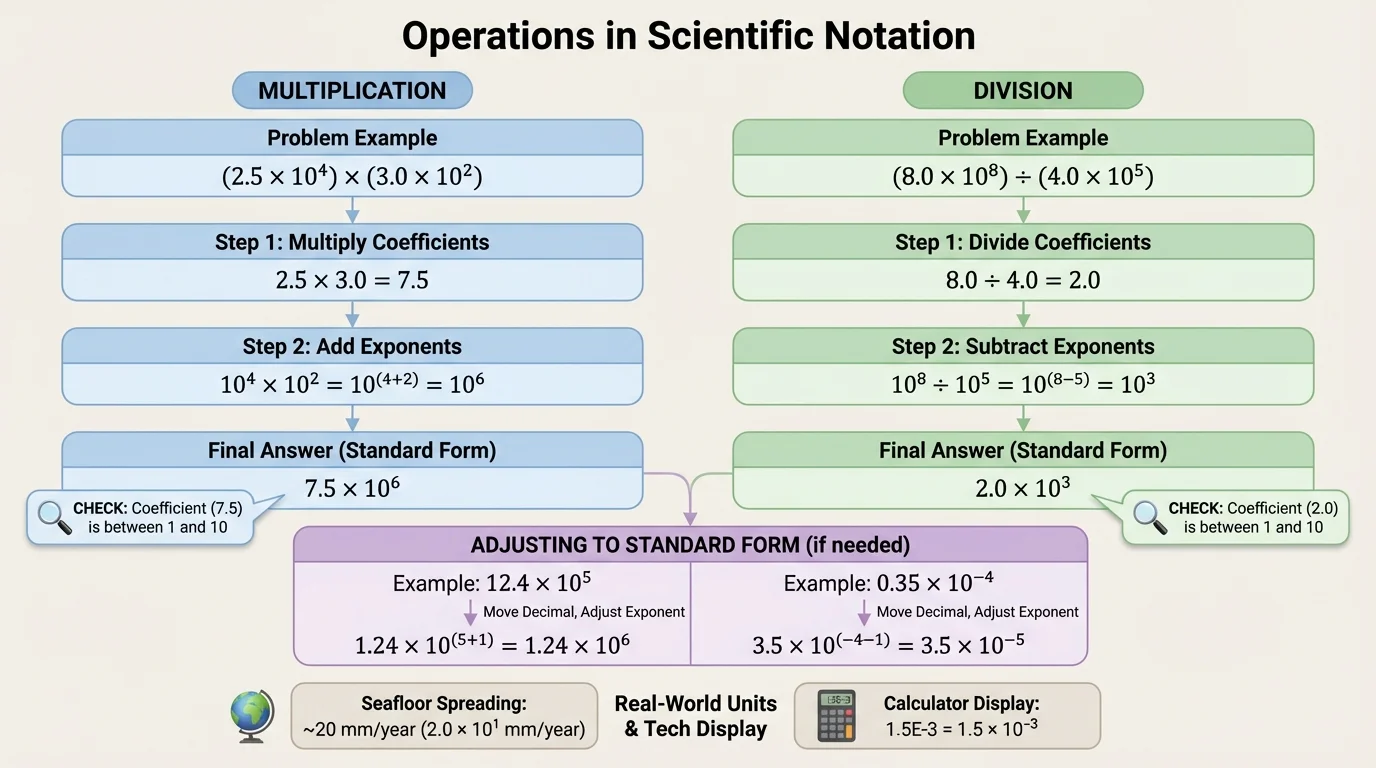
[Figure 3] Operations with scientific notation become easier when you use exponent rules. For multiplication, multiply the coefficients and add the exponents. For division, divide the coefficients and subtract the exponents.
The exponent rules are:
\[10^a \times 10^b = 10^{a+b}\]
\[\frac{10^a}{10^b} = 10^{a-b}\]
After multiplying or dividing, check whether the coefficient is still between \(1\) and \(10\). If not, rewrite the result in proper scientific notation.
Worked example 3
Multiply \((2 \times 10^5)(3 \times 10^4)\).
Step 1: Multiply the coefficients.
\(2 \times 3 = 6\)
Step 2: Add the exponents.
\(10^5 \times 10^4 = 10^9\)
Step 3: Write the product.
\[(2 \times 10^5)(3 \times 10^4) = 6 \times 10^9\]
Now try a case where the coefficient must be adjusted. For \((4 \times 10^3)(5 \times 10^2)\), first multiply: \(4 \times 5 = 20\), and add exponents: \(10^3 \times 10^2 = 10^5\). That gives \(20 \times 10^5\). Since \(20\) is not between \(1\) and \(10\), rewrite it as \(2.0 \times 10^6\).
Worked example 4
Divide \(8.4 \times 10^7\) by \(2 \times 10^3\).
Step 1: Divide the coefficients.
\(8.4 \div 2 = 4.2\)
Step 2: Subtract the exponents.
\(10^7 \div 10^3 = 10^4\)
Step 3: Write the quotient.
\[\frac{8.4 \times 10^7}{2 \times 10^3} = 4.2 \times 10^4\]
Later, when numbers appear in applications such as cell size or population estimates, the same flow from [Figure 3] still works: operate on the coefficients, operate on the powers of ten, and then adjust if needed.
Addition and subtraction are different from multiplication and division. You cannot simply add or subtract exponents unless the powers of ten already match. First, rewrite the numbers so they have the same exponent.
For example:
\(3.2 \times 10^5 + 4.7 \times 10^5 = 7.9 \times 10^5\)
That works because both terms use \(10^5\).
But if the exponents are different, rewrite one number. For instance:
\(6.1 \times 10^4 + 2.3 \times 10^3\)
Rewrite \(2.3 \times 10^3\) as \(0.23 \times 10^4\). Then add:
\[6.1 \times 10^4 + 0.23 \times 10^4 = 6.33 \times 10^4\]
Why equal exponents matter
Adding scientific notation works like combining like terms in algebra. Just as \(3x + 2x = 5x\) because both terms have the same variable, \(3 \times 10^6 + 2 \times 10^6 = 5 \times 10^6\) because both terms have the same power of ten.
Subtraction follows the same idea. Rewrite first if needed, then subtract the coefficients.
Worked example 5
Find \(5.6 \times 10^{-3} - 2.1 \times 10^{-4}\).
Step 1: Rewrite with matching exponents.
\(2.1 \times 10^{-4} = 0.21 \times 10^{-3}\)
Step 2: Subtract the coefficients.
\(5.6 - 0.21 = 5.39\)
Step 3: Write the result.
\[5.6 \times 10^{-3} - 2.1 \times 10^{-4} = 5.39 \times 10^{-3}\]
This is one of the most important differences between operations: multiplication and division use exponent rules directly, but addition and subtraction require matching powers of ten first.
Sometimes a problem gives one number in decimal form and another in scientific notation. In that case, convert one form so both numbers are easier to combine.
Suppose a microscope image shows a bacterium with length \(0.0000032\) meters, and another measure is \(1.8 \times 10^{-6}\) meters. To add them, convert \(0.0000032\) to scientific notation:
\(0.0000032 = 3.2 \times 10^{-6}\)
Now add:
\[3.2 \times 10^{-6} + 1.8 \times 10^{-6} = 5.0 \times 10^{-6}\]
The key decision is choosing the form that makes the operation more organized. For very large or very small numbers, scientific notation is usually the better choice.
Worked example 6
Find \(420{,}000 \div (2.1 \times 10^2)\).
Step 1: Rewrite the decimal in scientific notation.
\(420{,}000 = 4.2 \times 10^5\)
Step 2: Divide coefficients and subtract exponents.
\(4.2 \div 2.1 = 2\) and \(10^5 \div 10^2 = 10^3\)
Step 3: Write the quotient.
\[\frac{420{,}000}{2.1 \times 10^2} = 2 \times 10^3\]
You can also convert the final answer back to decimal form if the context makes that easier to understand: \(2 \times 10^3 = 2{,}000\).
[Figure 4] Sometimes the smartest move is not just changing the notation, but changing the units. Scientists choose units that make the number easier to read across different measurement scales.
For example, seafloor spreading may be only a few centimeters per year, or it may be easier to report it in millimeters per year. If a tectonic plate moves \(0.025\) meters in one year, that is \(2.5\) centimeters per year, which is also \(25\) millimeters per year. The unit \(25 \textrm{ mm/year}\) is often more natural than \(0.025 \textrm{ m/year}\).
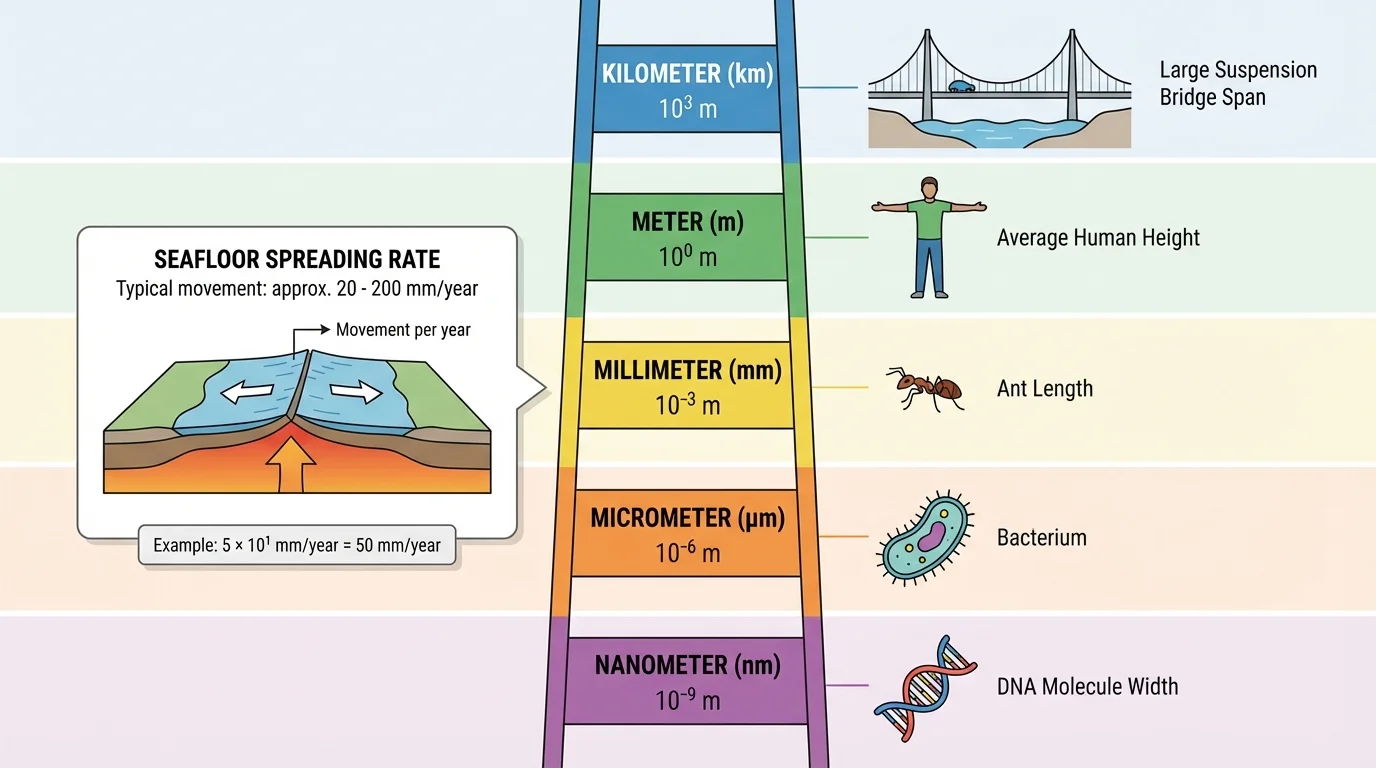
Very large distances may be better expressed in kilometers instead of meters. Very small lengths may be better expressed in millimeters, micrometers, or nanometers.
Examples:
A road length of \(12{,}000\) meters is often clearer as \(12\) kilometers.
A cell width of \(0.000012\) meters is clearer as \(12 \times 10^{-6} \textrm{ m}\), or \(12\) micrometers.
A virus diameter might be written in nanometers instead of meters because the meter value would contain many zeros.
Later, if you compare sizes from planets to bacteria, the unit ladder in [Figure 4] keeps the scale sensible. Good unit choice makes the number easier to communicate before any calculation even begins.
Scientific notation is not just a classroom tool. It is used in astronomy for enormous distances, in chemistry for tiny particles, in medicine for microscopic measurements, and in computer science for calculator and data output.
In astronomy, the distance to a star might be written with very large powers of ten. In biology, the size of a cell might be written with negative powers of ten. In geology, plate motion can be measured in millimeters per year. In each case, the notation reveals both the value and its scale.
| Field | Example quantity | Useful form |
|---|---|---|
| Astronomy | \(150{,}000{,}000\) kilometers | \(1.5 \times 10^8\) kilometers |
| Biology | \(0.000007\) meters | \(7 \times 10^{-6}\) meters |
| Geology | \(0.025\) meters per year | \(25 \textrm{ mm/year}\) |
| Technology | \(3.6\textrm{E}4\) | \(3.6 \times 10^4\) |
Table 1. Examples of large and small quantities written in scientific notation or more appropriate units.
Notice that scientific notation and unit choice often work together. A number can be easier to understand because of the notation, the unit, or both.
One mistake is using a coefficient outside the allowed range. For example, \(0.45 \times 10^6\) and \(45 \times 10^4\) are equal to the same number, but neither is proper scientific notation. The correct form is \(4.5 \times 10^5\).
Another mistake is moving the decimal in the wrong direction when converting. Remember: large number to scientific notation means move left and use a positive exponent; small decimal to scientific notation means move right and use a negative exponent.
A third mistake is adding exponents during addition problems. That rule belongs to multiplication, not addition. If you are adding or subtracting, first make the exponents match.
"The power of scientific notation is that it shows size and value at the same time."
With practice, these patterns become reliable: check the coefficient, check the sign of the exponent, and match exponents when adding or subtracting.