A campfire glows, a bicycle chain rusts, bread rises in the oven, and your body turns food into energy every minute. These events look very different, but they all involve the same big idea: matter can change when substances react chemically. In those reactions, the tiny particles that make up matter are not destroyed. Instead, they are rearranged into new combinations. That is why chemistry is so powerful: the same atoms can be organized into substances with completely different properties.
When scientists study matter, they do more than describe what something looks like. They ask what it is made of, how it behaves, and what happens when it interacts with other substances. A burned marshmallow, a fizzing antacid tablet, and a tarnished coin all show that matter does not always stay the same. Sometimes a change is only physical, such as cutting paper or melting ice. But sometimes a change creates entirely new substances. That kind of change is called a chemical reaction.
Understanding chemical reactions helps explain many real-world processes. Engineers design better batteries by controlling reactions. Doctors rely on chemical changes in medicines and in the human body. Environmental scientists study reactions in air and water to understand pollution and climate. Even cooking depends on chemistry, from browning toast to turning liquid egg into a solid when heated.
Matter is anything that has mass and takes up space. All matter is made of atoms. Atoms can join together to form molecules, and different combinations of atoms give substances different properties.
To understand chemical change, we need to look below what our eyes can see. The visible evidence is important, but the real story happens at the atomic level.
An atom is the smallest unit of an element that still has the properties of that element. Elements such as hydrogen, oxygen, carbon, and iron each have their own kinds of atoms. Atoms can join in fixed ways to form molecules and compounds. For example, water is made of molecules of \(\textrm{H}_2\textrm{O}\), which means each water molecule contains two hydrogen atoms and one oxygen atom.
When atoms join together, they do not lose their identity as atoms. A hydrogen atom remains a hydrogen atom whether it is in water, sugar, or a fuel. What changes is the molecule or structure that the atoms are part of. This idea is essential because in a chemical process, atoms are regrouped into different molecules rather than being created from nothing.
A compound is a substance made of two or more different elements chemically combined. Water, carbon dioxide, and table salt are compounds. Their properties are different from those of the elements that make them. Hydrogen gas can burn, and oxygen gas supports burning, but water puts fires out. This shows that combining atoms in new ways can lead to very different behavior.
chemical reaction is a process in which atoms in substances are rearranged to form new substances.
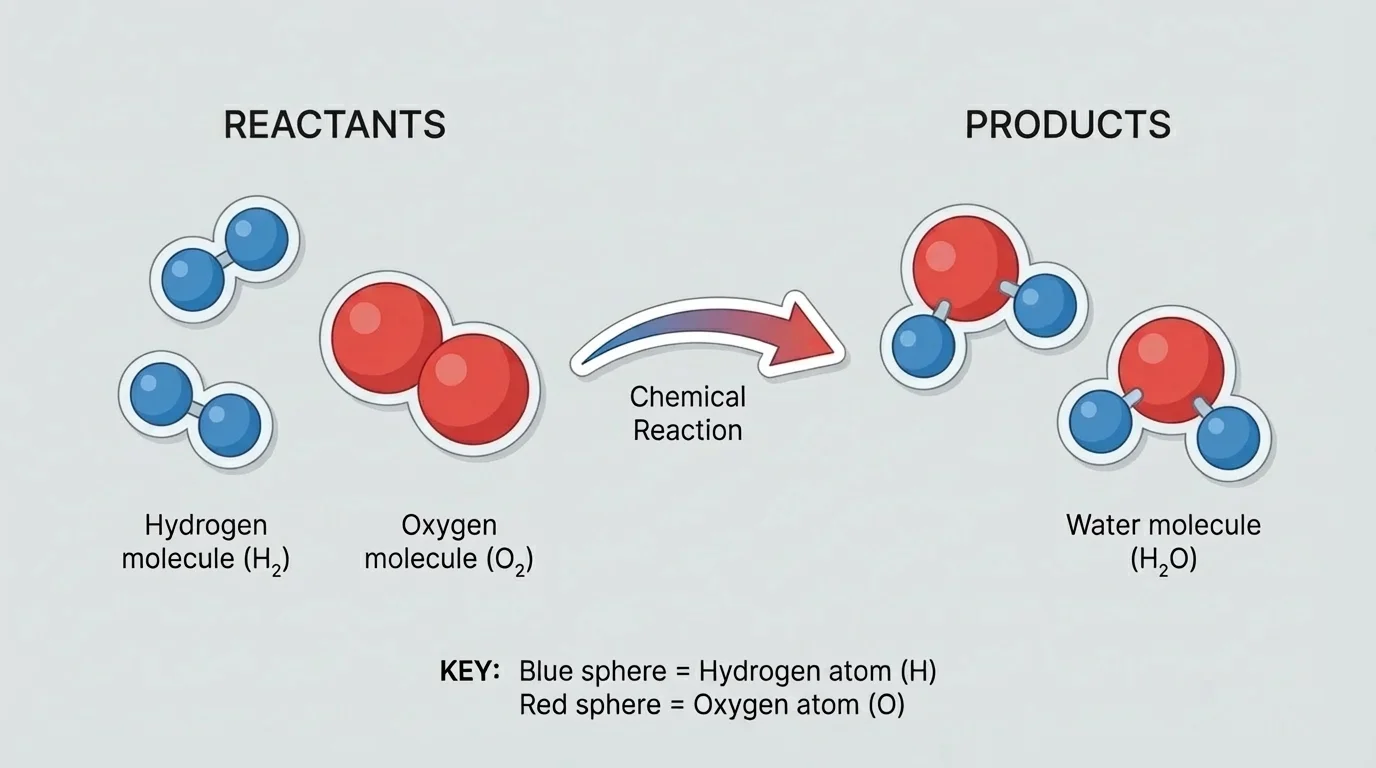
Reactants are the starting substances in a reaction.
Products are the new substances formed in a reaction.
These terms help us describe what goes into a reaction and what comes out of it, as shown in [Figure 1].
In a chemical reaction, the reactants change into products because atoms are regrouped into new arrangements. The atoms themselves stay the same kind of atom, but the bonds between them can break and new bonds can form. This creates new molecules with new properties.
For example, hydrogen gas and oxygen gas can react to make water. Scientists represent that process with a chemical equation:
\[2\textrm{H}_2 + \textrm{O}_2 \rightarrow 2\textrm{H}_2\textrm{O}\]
This equation shows that two molecules of hydrogen react with one molecule of oxygen to form two molecules of water. Notice that there are still four hydrogen atoms and two oxygen atoms after the reaction. The atoms are conserved, but they are connected in a new way.
Chemical reactions often involve energy as bonds break and form. Some reactions release energy to the surroundings as heat or light. Others absorb energy. But whether energy is released or absorbed, the main matter idea stays the same: atoms are rearranged, not lost.
The products of a reaction usually have different properties from the reactants. Water behaves very differently from hydrogen and oxygen gases. Rust looks and acts differently from shiny iron. A cake is not just flour, eggs, and sugar sitting together; baking causes reactions that produce new substances and textures.
Because atoms are too small to see directly without special tools, we often look for clues that a reaction has happened. Several common signs appear in many reactions. A single sign does not always prove a chemical reaction, but multiple pieces of evidence make the case stronger.
[Figure 2] One sign is gas production. If bubbles form when two substances are mixed, a gas may be forming. For example, baking soda and vinegar react to produce carbon dioxide gas:
\[\textrm{NaHCO}_3 + \textrm{CH}_3\textrm{COOH} \rightarrow \textrm{CO}_2 + \textrm{H}_2\textrm{O} + \textrm{CH}_3\textrm{COONa}\]
Another sign is the formation of a precipitate, which is a solid that forms when two liquids react. If clear solutions are mixed and a cloudy solid suddenly appears, that often means a new substance has formed.
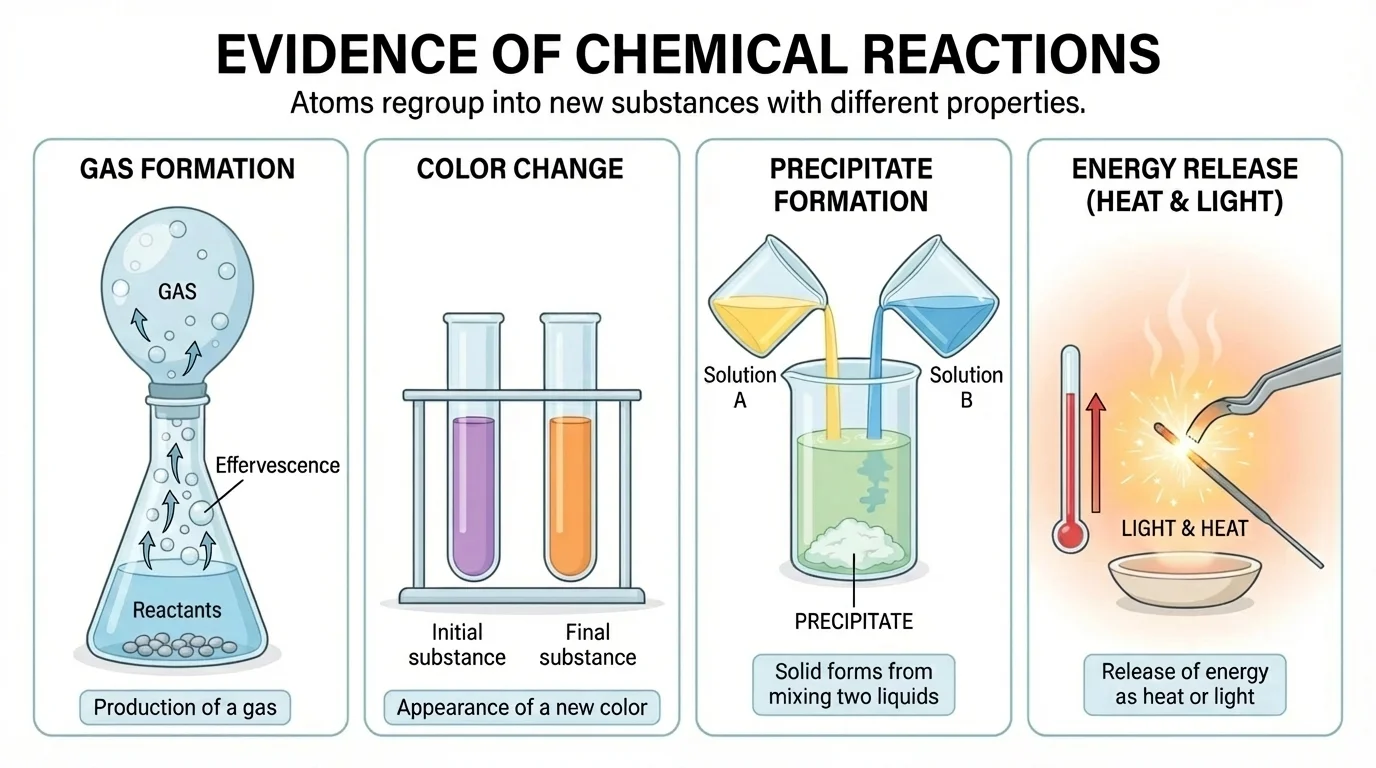
A color change can also be evidence. Iron turning reddish-brown as it rusts is a common example. However, color change alone is not enough to prove a reaction, because physical mixing can also change appearance.
Temperature change is another clue. Some reactions feel warmer because they release heat. Others feel colder because they absorb heat from their surroundings. Light, smell, or sound may also be produced in some chemical changes.
It is important to be careful. Boiling water makes bubbles, but that is a physical change, not a chemical reaction. The bubbles are water vapor, and the substance is still \(\textrm{H}_2\textrm{O}\). Good science depends on looking at all the evidence, not jumping to conclusions from one observation.
Evidence versus proof
Scientists often say that observations provide evidence for a reaction rather than instant proof. A bubbling liquid may mean gas is forming, but it could also be boiling. A color change may come from a reaction, or it may come from mixing colored substances. The strongest conclusion comes from combining several observations with knowledge of the substances involved.
This careful approach helps explain why chemists test substances in systematic ways. Different materials react in characteristic patterns, and those patterns help identify what the substances are.
One of the most important ideas in chemistry is that atoms are conserved in ordinary chemical reactions. The particle model shows that the same number of each kind of atom appears before and after a reaction. Atoms may be separated from old partners and joined to new ones, but they do not vanish.
[Figure 3] This idea is why chemical equations must be balanced. A balanced equation has the same number of each type of atom on both sides of the arrow. Consider methane burning in oxygen:
\[\textrm{CH}_4 + 2\textrm{O}_2 \rightarrow \textrm{CO}_2 + 2\textrm{H}_2\textrm{O}\]
Count the atoms. On the left, there is one carbon atom, four hydrogen atoms, and four oxygen atoms. On the right, there is one carbon atom in \(\textrm{CO}_2\), four hydrogen atoms in \(2\textrm{H}_2\textrm{O}\), and four oxygen atoms total. The numbers match.
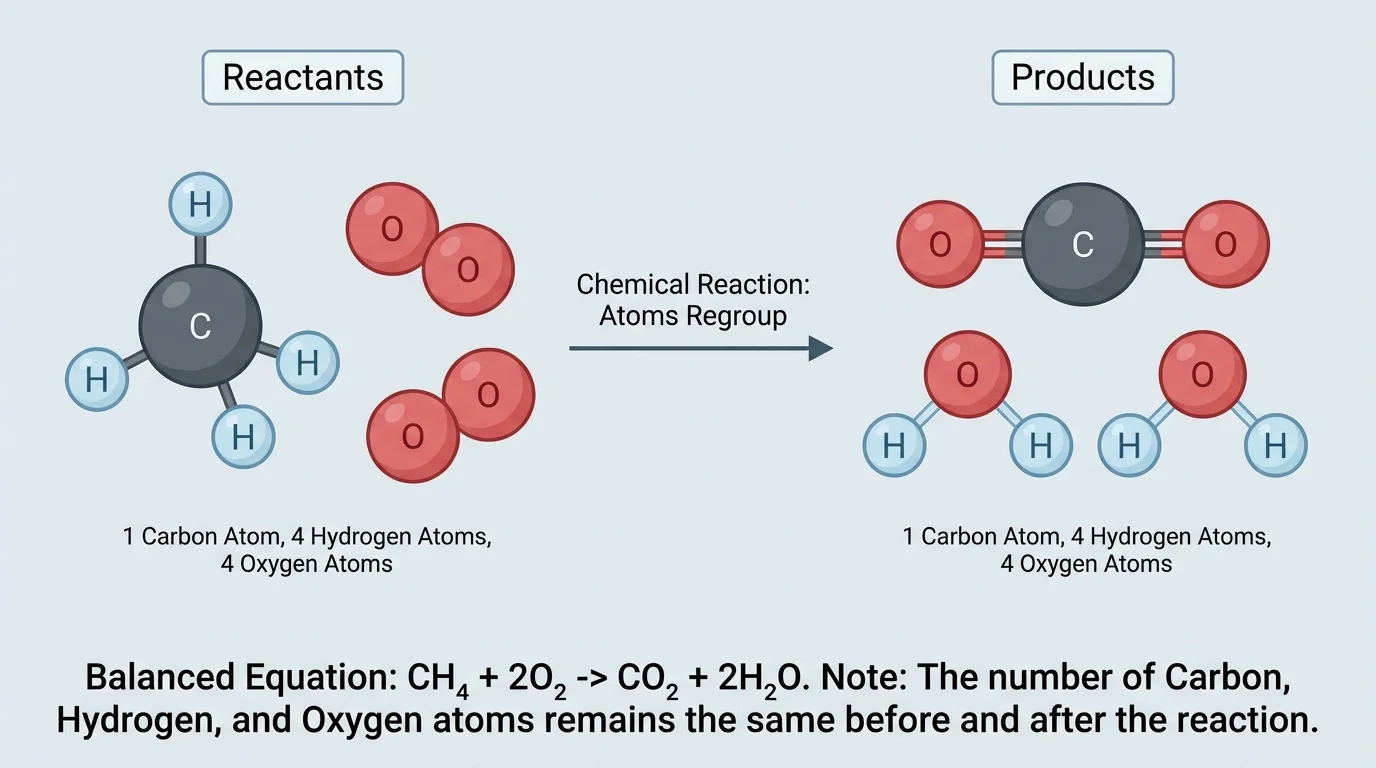
This balancing does not mean the amounts have to look the same. The reactants and products may be gases, liquids, or solids. They may have very different colors, smells, or temperatures. Conservation means that the atom counts match, not that the substances look alike.
| Equation | Reactants | Products | What stays the same |
|---|---|---|---|
| \(2\textrm{H}_2 + \textrm{O}_2 \rightarrow 2\textrm{H}_2\textrm{O}\) | Hydrogen and oxygen | Water | \(4\) H atoms, \(2\) O atoms |
| \(\textrm{CH}_4 + 2\textrm{O}_2 \rightarrow \textrm{CO}_2 + 2\textrm{H}_2\textrm{O}\) | Methane and oxygen | Carbon dioxide and water | \(1\) C, \(4\) H, \(4\) O atoms |
| \(4\textrm{Fe} + 3\textrm{O}_2 \rightarrow 2\textrm{Fe}_2\textrm{O}_3\) | Iron and oxygen | Iron oxide | \(4\) Fe atoms, \(6\) O atoms |
Table 1. Examples of balanced chemical equations showing that atom counts are conserved.
Later, when chemists measure mass very carefully, they find that total mass is also conserved in a closed system because the same atoms are still present. If matter seems to disappear, it often has changed form, such as turning into a gas that moves away.
Different substances tend to react in predictable ways. Oxygen often reacts with fuels in combustion and with metals in oxidation. Acids react with certain bases and carbonates in recognizable patterns. These characteristic reactions help scientists identify substances and predict what may happen when materials are mixed.
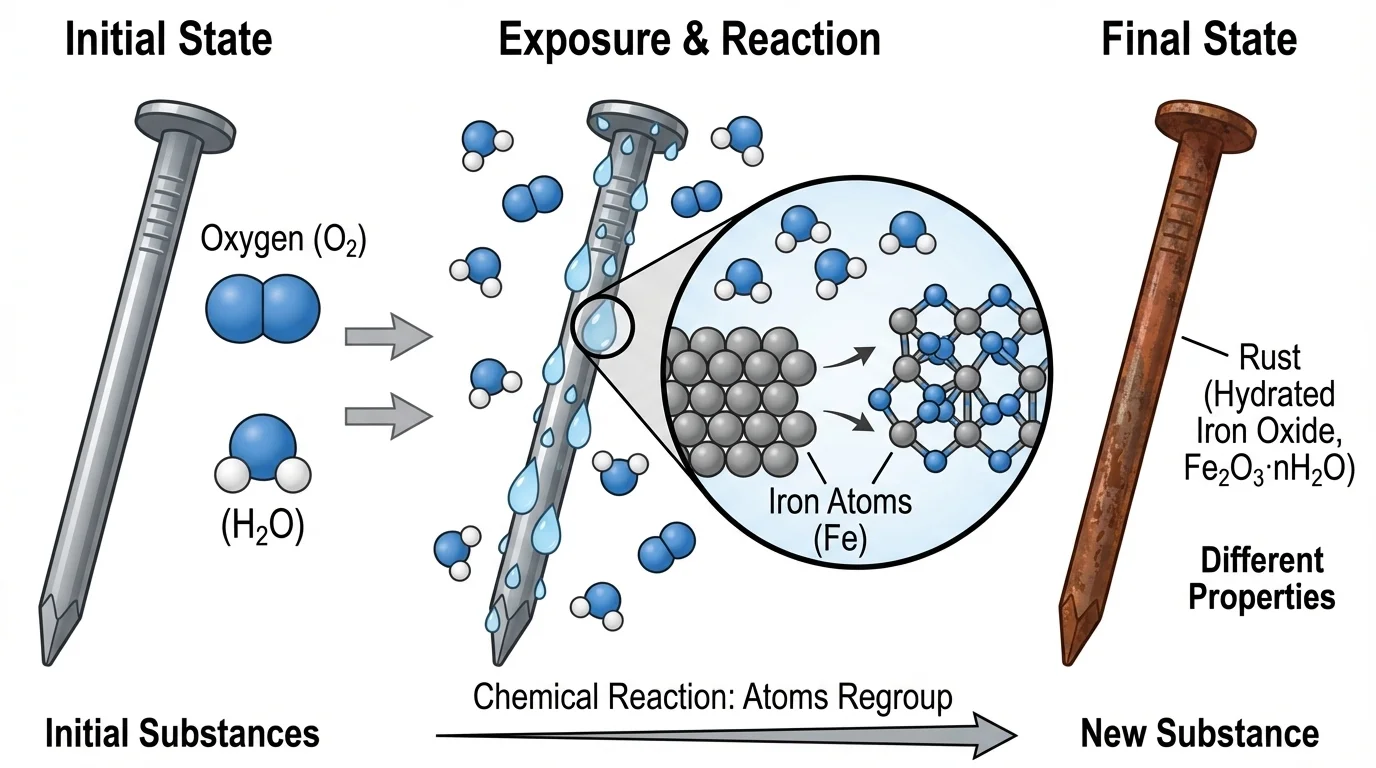
[Figure 4] A familiar example is rusting. Iron reacts with oxygen, often with water helping the process happen more easily. The oxidation pattern leads to a reddish-brown substance commonly called rust, which is mainly iron oxide. A simplified equation is:
\[4\textrm{Fe} + 3\textrm{O}_2 \rightarrow 2\textrm{Fe}_2\textrm{O}_3\]
Rust has very different properties from the iron it came from. Iron is strong and metallic. Rust is brittle and flaky. This is a powerful example of how regrouping atoms creates a new substance with new properties.
Combustion is another common pattern. When a fuel such as wood, wax, or methane reacts with oxygen, it can release heat and light. In complete combustion, carbon-containing fuels often produce carbon dioxide and water. That is part of why flames are both useful and dangerous: they are visible signs of fast chemical reactions releasing energy.
Acid-base reactions are important in digestion, cleaning, and manufacturing. Antacid tablets work because basic compounds react with acid in the stomach. Vinegar reacting with baking soda is another acid-base example that produces carbon dioxide gas.
Cooking provides many chemical changes. When an egg is heated, proteins change structure and new interactions form, turning the runny liquid into a solid. When bread bakes, gases expand, browning reactions occur, and the dough becomes bread with new texture, smell, and taste. These are not just physical rearrangements; they involve chemistry.
Glow sticks work because chemicals react to produce light directly. Unlike a hot light bulb, the reaction releases much of its energy as visible light rather than as large amounts of heat.
These examples show that substances do not react randomly. Their structures and properties affect how they combine and what products they form.
Chemical reactions are closely linked to energy. To start a reaction, some bonds in the reactants must break, which requires energy. When new bonds form in the products, energy is released. Whether the whole reaction feels hot or cold depends on the balance between these two changes.
An exothermic reaction releases more energy than it absorbs. Burning fuel and many hand-warmer reactions are exothermic. An endothermic reaction absorbs more energy than it releases. Some instant cold packs work this way, becoming colder as the reaction takes in heat from the surroundings.
Real-world example: a cold pack
Step 1: A chemical pack is squeezed, breaking a small inner pouch.
Step 2: Two substances mix and react.
Step 3: The reaction absorbs thermal energy from nearby surroundings, including your skin.
Step 4: The pack feels cold because energy is transferred away from your hand into the reaction.
The products are different substances from the reactants, and the temperature change gives evidence that a chemical process is happening.
Energy changes matter in real life because they affect safety, usefulness, and speed. Engineers designing fuels, batteries, or heating materials need to know not only what products form but also how much energy the reactions involve.
Not all chemical reactions happen at the same speed. Conditions can make a reaction faster or slower. These conditions do not change the rule that atoms are conserved, but they can change how quickly reactants turn into products.
Higher temperature usually speeds reactions up because particles move faster and collide more often with enough energy to react. Greater concentration can also increase reaction rate because more reacting particles are crowded into the same space. Smaller pieces of a solid react faster than one large chunk because more surface is exposed. That is why powdered materials often react more quickly than solid blocks of the same substance.
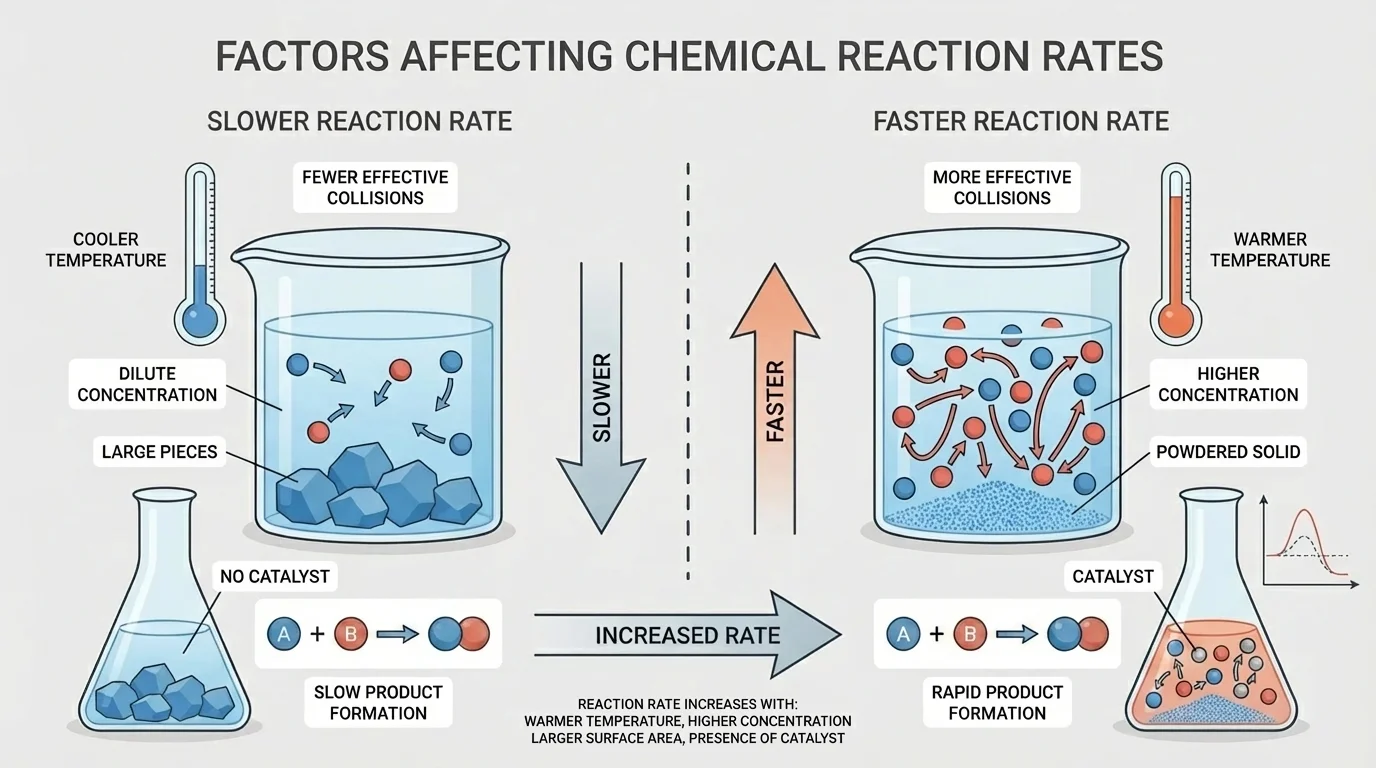
[Figure 5] A catalyst is a substance that speeds up a reaction without being used up in the process. Catalysts are extremely important in industry and biology. Enzymes in your body are biological catalysts that help digestion and other life processes happen fast enough to keep you alive.
Reaction conditions matter in cooking, manufacturing, and environmental science. Refrigeration slows many reactions, helping food last longer. Catalytic converters in cars use catalysts to reduce harmful emissions. Crushing a tablet can increase the speed at which it dissolves and reacts.
Even when the speed changes, the products still depend on the kinds of atoms present and how those atoms can rearrange. Faster or slower does not mean atoms are created or destroyed.
Chemical reactions are not just lab topics. Your body uses reactions to break down food and release usable energy. Plants use reactions in photosynthesis to build sugars from carbon dioxide and water using light energy. Batteries work because chemical reactions push electrons through a circuit, powering phones, flashlights, and electric cars.
Controlling reactions is also a big challenge. Rust damages bridges, tools, and cars, so people paint metal, coat it, or use different materials to slow oxidation. In medicine, drug makers design compounds that react in very specific ways in the body. In environmental science, researchers track reactions that create smog, acid rain, or greenhouse gases.
The idea applies far beyond rusty nails. Whenever a material changes because atoms combine differently, its properties can change in ways that matter for safety, health, and technology.
One common misunderstanding is that any visible change must be a chemical reaction. That is not true. Melting ice, tearing paper, and dissolving sugar in water are physical changes. The substances may look different, but no new substances form.
Another misunderstanding is that atoms disappear during a reaction. The particle picture from [Figure 3] helps correct this idea. If a candle seems to lose matter while burning, much of the matter has turned into gases such as \(\textrm{CO}_2\) and water vapor and moved into the air.
Students also sometimes think that mixing substances automatically means a reaction happened. Sometimes substances simply form a mixture. Sand and salt stirred together stay as sand and salt. But if substances react chemically, new products form with different properties.
Learning to tell physical change from chemical change takes practice in careful observation and particle-level thinking. The key question is not just "What do I see?" but "Have the atoms been rearranged into new substances?"
"In a chemical change, matter is not lost; it is rearranged."
That single idea connects campfires, batteries, rust, digestion, and baking. Chemistry becomes easier to understand when you picture atoms as pieces being rearranged into new structures rather than being destroyed or created.