A tiny change inside an atom's nucleus can release far more energy than a major chemical reaction. Burning fuel in a car, lighting a match, or digesting food all involve electrons and chemical bonds. Nuclear processes are different: they change the nucleus itself. That difference is why the Sun shines for billions of years, why nuclear power plants can produce enormous amounts of electricity from small amounts of fuel, and why radioactive materials can be used in medicine and industry.
To understand these processes, it helps to build models. A model can be a drawing, a particle diagram, a symbol equation, or a verbal explanation that tracks what enters a process, what leaves, and how the nucleus changes. In nuclear science, the most important questions are: How many protons are in the nucleus? How many neutrons are in it? Is the nucleus stable? And what happens to energy when the nucleus changes?
An atom has a central nucleus made of protons and neutrons, with electrons outside it. Almost all of the atom's mass is packed into that tiny nucleus. When the nucleus changes, the identity or stability of the atom can change too. That is very different from ordinary chemical reactions, where atoms rearrange but their nuclei stay the same.
Because the nucleus is so compact, the forces inside it are intense. A small rearrangement of nuclear particles can release a great deal of energy. This does not mean nuclei are easy to change. In fact, most nuclei are very hard to alter. But when certain nuclei do change, the energy released can be dramatic.
Remember that the atomic number tells how many protons an atom has, and the mass number tells the total number of protons and neutrons. For example, in \(^{14}_{6}\textrm{C}\), the atomic number is \(6\) and the mass number is \(14\), so the nucleus has \(6\) protons and \(8\) neutrons.
Nuclear models often use the notation \(^{A}_{Z}\textrm{X}\), where \(A\) is the mass number, \(Z\) is the atomic number, and \(\textrm{X}\) is the element symbol. This notation is useful because it lets you see nuclear composition at a glance.
[Figure 1] shows that the nucleus contains protons, which are positively charged, and neutrons, which have no electric charge. A nucleus with the same number of protons but a different number of neutrons is called an isotope. The balance of particles and forces inside the nucleus determines whether it remains together.
Protons repel one another because like electric charges push apart. If electric repulsion were the only force present, nuclei would fly apart. But at very short distances another force becomes dominant: the strong nuclear force. This force acts between nucleons, meaning protons and neutrons, and it is strong enough to overcome proton-proton repulsion when the particles are close together.
Neutrons are important because they add attractive strong-force interactions without adding electric repulsion. That is one reason many stable nuclei contain neutrons as well as protons. In heavier nuclei, extra neutrons help offset the growing repulsion among the many protons.
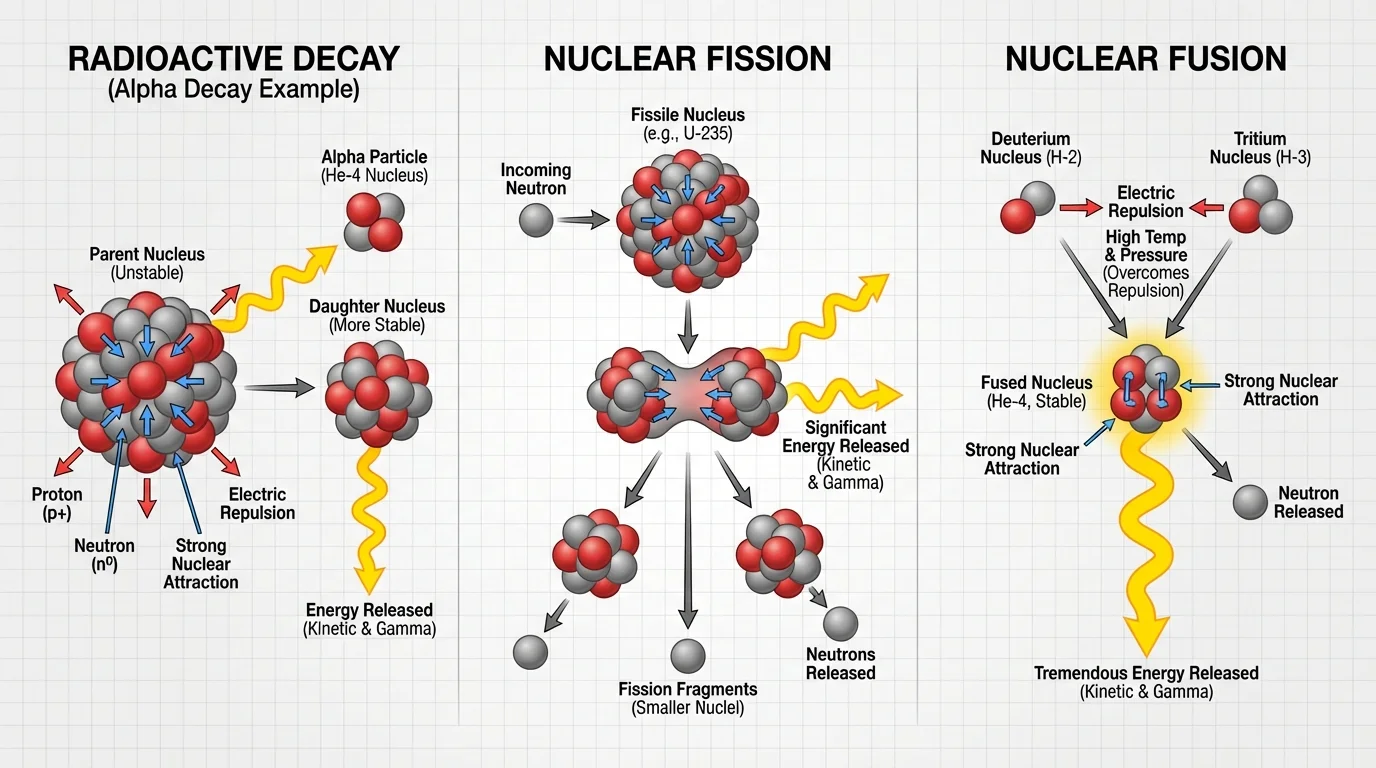
However, the strong nuclear force works only over very short distances. In very large nuclei, not every proton is close enough to all the others for the strong force to hold everything equally well. This helps explain why very heavy nuclei are more likely to be unstable or capable of fission.
Nucleus is the dense central region of an atom containing protons and neutrons.
Nuclear stability refers to how likely a nucleus is to remain unchanged rather than spontaneously decay or split.
Binding energy is the energy associated with holding the nucleus together. A more tightly bound nucleus has lower energy than separated particles.
A useful way to think about stability is that nuclei tend to move toward arrangements that are more tightly bound. When a nuclear process produces products that are more stable than the original nucleus, energy can be released to the surroundings.
The idea of binding energy explains why nuclear reactions can release energy. A bound nucleus has less energy than the same protons and neutrons separated from one another. That "missing" energy is connected to the energy required to pull the nucleus apart, or the energy released when the nucleus forms a more stable arrangement.
Albert Einstein expressed the relationship between mass and energy with \(E = mc^2\). In nuclear processes, a tiny difference in mass corresponds to a large amount of energy because \(c^2\), the speed of light squared, is enormous. For example, if a mass change of only \(1.0 \times 10^{-6} \textrm{ kg}\) were converted completely to energy, the result would be \(E = (1.0 \times 10^{-6})(3.0 \times 10^8)^2\), which is about \(9.0 \times 10^{10} \textrm{ J}\). The important idea is not the calculation itself but that a very small mass change can correspond to a very large energy release.
Students often ask whether energy is "stored" inside the nucleus like a battery. A better model is that energy depends on the arrangement of the particles. If a reaction changes the nucleus into a more stable arrangement, the total energy of the system decreases and the difference appears as released energy, often carried by kinetic energy of particles or radiation.
The energy from sunlight begins with nuclear fusion in the Sun's core. Every second, the Sun converts an enormous amount of hydrogen into helium, yet only a tiny fraction of the Sun's total mass is changed each second, which is why it can shine for billions of years.
This same stability idea helps connect fission, fusion, and radioactive decay. Although the processes look different, each one involves a nucleus changing into a configuration that is, in some way, more stable than the starting nucleus.
When you model a nuclear process, two quantities must be tracked carefully: the total number of protons and the total number of nucleons. In symbolic form, that means the total atomic number and the total mass number must balance from the start of the process to the end.
For example, if a nucleus emits an alpha particle, you can model the change by subtracting \(2\) from the atomic number and \(4\) from the mass number, because an alpha particle is a helium nucleus, \(^{4}_{2}\textrm{He}\). If a nucleus emits gamma radiation, the nucleus does not lose protons or neutrons, so the atomic number and mass number remain the same.
Modeling a nucleus before and after decay
Suppose a polonium-210 nucleus undergoes alpha decay:
\(^{210}_{84}\textrm{Po} \rightarrow \; ^{206}_{82}\textrm{Pb} + ^{4}_{2}\textrm{He}\)
Step 1: Check the mass number.
On the left, the mass number is \(210\). On the right, \(206 + 4 = 210\).
Step 2: Check the atomic number.
On the left, the atomic number is \(84\). On the right, \(82 + 2 = 84\).
Step 3: Interpret the model.
The nucleus loses \(2\) protons and \(2\) neutrons, so it becomes a different element and releases energy.
This kind of balancing model is one of the most powerful tools in nuclear science. It lets you see exactly how nuclear composition changes, even when the process itself happens too quickly or on too small a scale to observe directly.
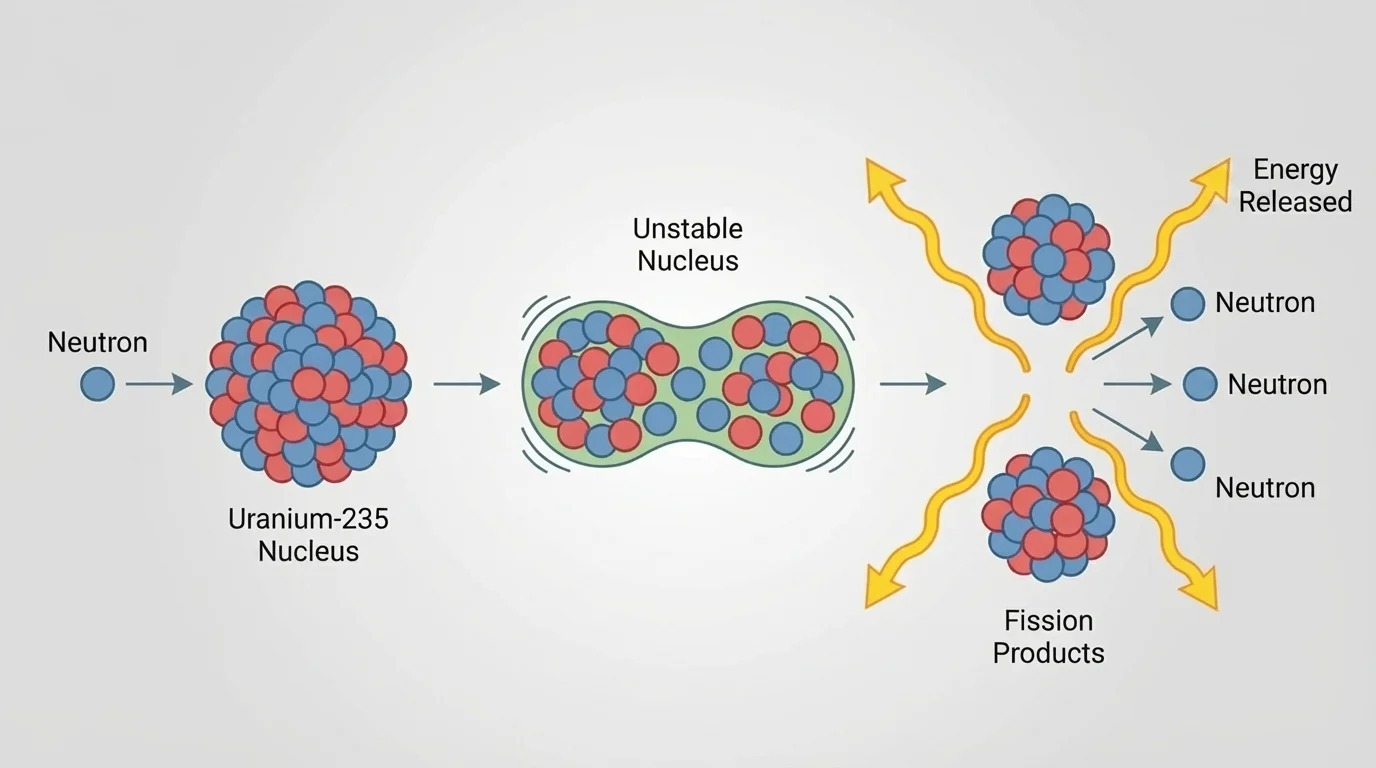
[Figure 2] illustrates that fission is the splitting of a heavy nucleus into smaller nuclei, usually along with free neutrons and released energy. In many models, a heavy nucleus such as \(^{235}_{92}\textrm{U}\) absorbs a neutron and becomes unstable; then it splits.
A fission event does not always produce the same pair of smaller nuclei, but the overall pattern is the same: one large nucleus becomes two medium-sized nuclei, plus neutrons and energy. Those emitted neutrons matter because they can strike other heavy nuclei and trigger more fission events.
That repeated triggering is called a chain reaction. If each fission event causes more than one additional fission, the reaction can grow rapidly. If the reaction is controlled so that, on average, one event leads to about one more event, energy can be released steadily.
In a nuclear reactor, control rods, coolant, and careful design help regulate the chain reaction. The goal is not an explosion, but a steady release of energy that can be used to heat water, produce steam, spin turbines, and generate electricity. In this way, the structure of the nucleus is connected directly to large-scale engineering systems.
Fission releases energy because the daughter nuclei are more tightly bound, on average, than the original very heavy nucleus. The details are complex, but the core idea is simple: the products have lower total nuclear energy than the starting arrangement, and the difference is released.
Interpreting a fission model
One possible fission reaction is:
\(^{235}_{92}\textrm{U} + ^{1}_{0}\textrm{n} \rightarrow \; ^{141}_{56}\textrm{Ba} + ^{92}_{36}\textrm{Kr} + 3\,^{1}_{0}\textrm{n}\)
Step 1: Check mass numbers.
Left side: \(235 + 1 = 236\). Right side: \(141 + 92 + 3 = 236\).
Step 2: Check atomic numbers.
Left side: \(92 + 0 = 92\). Right side: \(56 + 36 = 92\).
Step 3: Describe the composition change.
A heavy uranium nucleus becomes barium and krypton nuclei, and neutrons are released that may continue the chain reaction.
The reactor model also helps explain why shielding and safety systems are essential. Fission products can be radioactive, and the released energy must be managed carefully. The same process that makes fission useful for electricity also demands responsible design and long-term waste handling.
Later, when comparing nuclear processes, it is helpful to remember the pattern in [Figure 2]: one large nucleus becomes smaller pieces, with neutrons often acting as both trigger and product.
[Figure 3] shows that fusion is the joining of light nuclei to form a heavier nucleus. In stars, very light nuclei, mainly forms of hydrogen, combine under extreme temperature and pressure. Light nuclei must move fast enough to get close together despite electric repulsion.
Once the nuclei are close enough, the strong nuclear force can pull them together. The result is a more tightly bound nucleus and released energy. Fusion is the main energy source for stars, including the Sun.
A simple model involves hydrogen isotopes combining to make helium. One well-known fusion reaction uses deuterium and tritium:
\(^{2}_{1}\textrm{H} + ^{3}_{1}\textrm{H} \rightarrow \; ^{4}_{2}\textrm{He} + ^{1}_{0}\textrm{n}\)
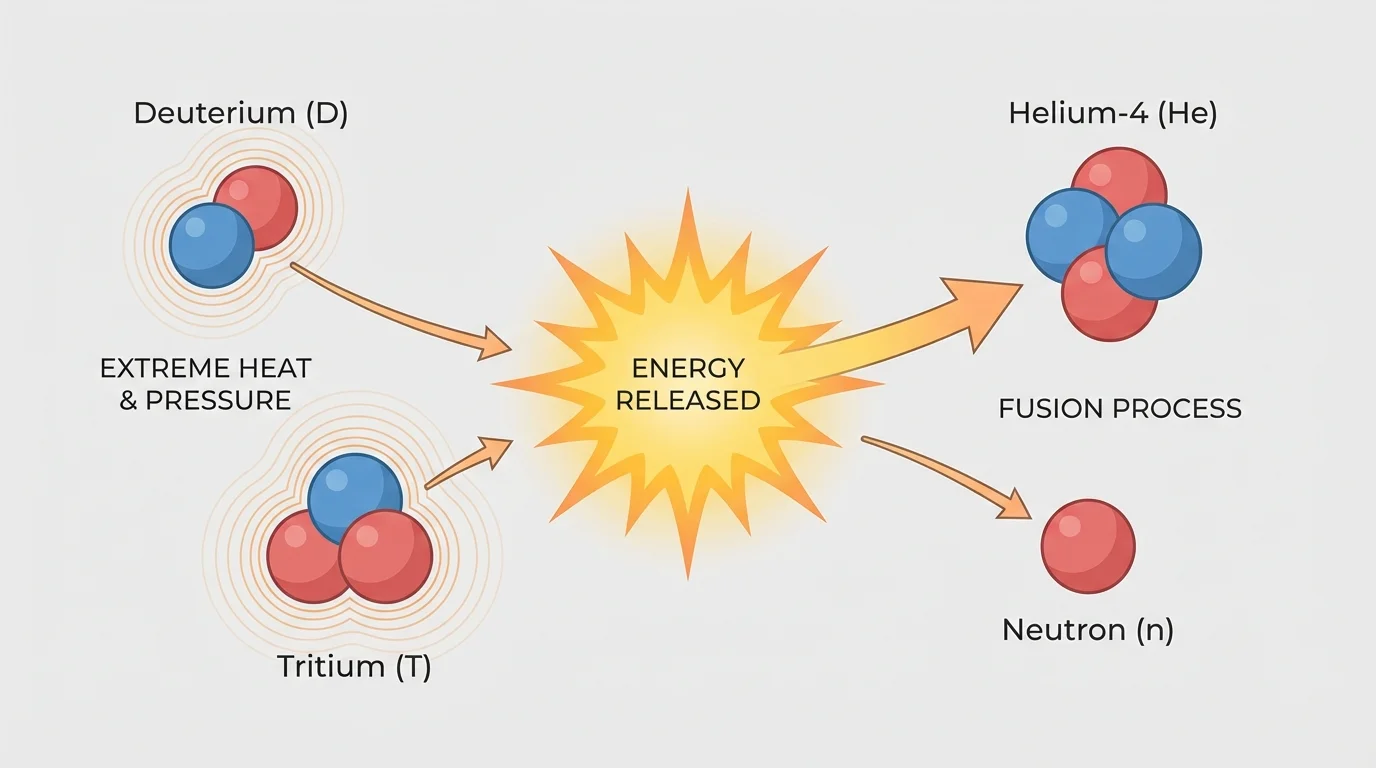
Again, the model balances. The mass numbers give \(2 + 3 = 5\) on the left and \(4 + 1 = 5\) on the right. The atomic numbers give \(1 + 1 = 2\) on the left and \(2 + 0 = 2\) on the right. The products are more tightly bound than the original nuclei, so energy is released.
Fusion is attractive as an energy source because its fuel can be derived from abundant materials and because it does not produce the same long-lived fission products as nuclear reactors. However, controlled fusion on Earth is extremely difficult. It requires temperatures so high that matter exists as plasma, a state in which electrons are not bound to nuclei.
Why both fission and fusion release energy
This may seem like a paradox: how can both splitting nuclei and joining nuclei release energy? The answer is nuclear binding. Very heavy nuclei can release energy by splitting into more stable medium-sized nuclei, while very light nuclei can release energy by joining into more stable larger nuclei. In both cases, the products are more tightly bound than the starting nuclei.
Engineers are developing fusion reactors that use magnetic fields or powerful lasers to confine plasma. These designs try to reproduce, in a controlled way, the conditions that stars create naturally through gravity. The star-like process in [Figure 3] is therefore not just astronomy; it is also a major engineering challenge.
[Figure 4] shows that some nuclei are unstable and change on their own. This process is called radioactive decay. Unlike fission and fusion, decay does not need an outside trigger such as a neutron collision or extreme pressure. Different types of decay change the nucleus in different ways.
For this topic, the key radioactive decays are alpha decay, beta decay, and gamma decay. Each one changes the composition or energy of the nucleus in a characteristic way, and each one releases energy.
An unstable nucleus tends to decay toward a more stable arrangement. The released energy may appear as moving particles, electromagnetic radiation, or both. By modeling what leaves the nucleus, you can determine how the remaining nucleus changes.
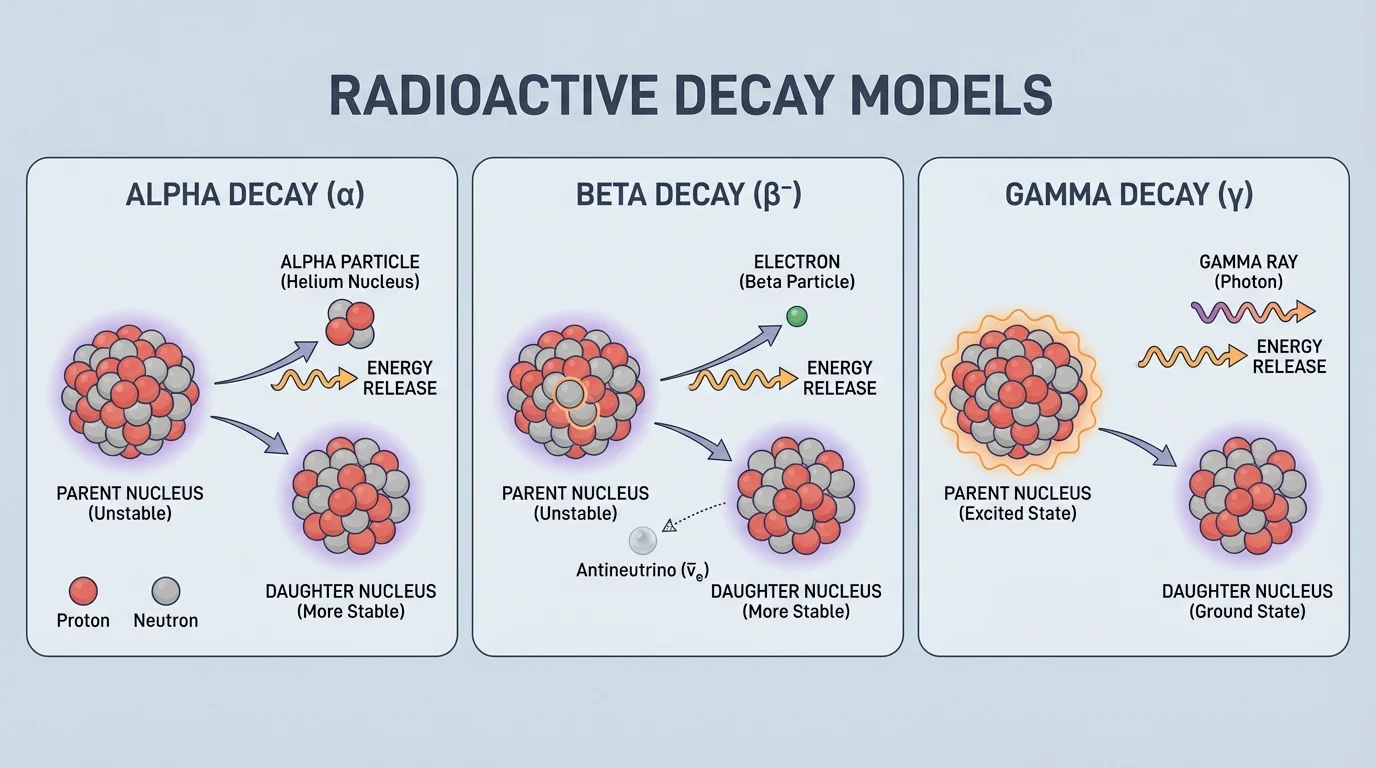
In alpha decay, the nucleus emits an alpha particle, which is \(^{4}_{2}\textrm{He}\). Because the nucleus loses \(2\) protons and \(2\) neutrons, the atomic number decreases by \(2\) and the mass number decreases by \(4\).
For example:
\(^{226}_{88}\textrm{Ra} \rightarrow \; ^{222}_{86}\textrm{Rn} + ^{4}_{2}\textrm{He}\)
This changes the element because the number of protons changes from \(88\) to \(86\). Alpha particles are relatively massive and carry positive charge, so they interact strongly with matter and are stopped easily by paper or skin.
In beta decay, a neutron in the nucleus changes into a proton, and a fast electron is emitted from the nucleus. The mass number stays the same because the total number of nucleons does not change, but the atomic number increases by \(1\) because there is now one more proton.
One example is:
\(^{14}_{6}\textrm{C} \rightarrow \; ^{14}_{7}\textrm{N} + ^{0}_{-1}\textrm{e}\)
Before decay, carbon-14 has \(6\) protons and \(8\) neutrons. After decay, nitrogen-14 has \(7\) protons and \(7\) neutrons. The nucleus has changed composition even though the mass number remains \(14\). As the beta panel in [Figure 4] shows, the emitted electron is one sign that the internal balance of the nucleus has changed.
In gamma decay, the nucleus emits high-energy electromagnetic radiation called a gamma ray. This usually happens when a nucleus is already in an excited state and drops to a lower energy state. Unlike alpha and beta decay, gamma decay does not change the number of protons or neutrons.
A model can be written as:
\(^{*}\textrm{X} \rightarrow \textrm{X} + \gamma\)
Here the star indicates an excited nucleus. After gamma emission, the nucleus is the same element and isotope, but it has less energy. Gamma rays are highly penetrating and usually require dense shielding such as lead or thick concrete.
Comparing the three decay types
Step 1: Alpha decay removes \(2\) protons and \(2\) neutrons.
Atomic number changes by \(-2\); mass number changes by \(-4\).
Step 2: Beta decay changes a neutron into a proton.
Atomic number changes by \(+1\); mass number changes by \(0\).
Step 3: Gamma decay lowers nuclear energy without changing composition.
Atomic number changes by \(0\); mass number changes by \(0\).
These differences matter in real applications. Alpha emitters can be dangerous if inhaled or swallowed, beta radiation can penetrate more than alpha but less than gamma, and gamma rays are valuable in imaging and treatment because of their penetrating power.
Although all three are nuclear processes, they are not the same. Fission starts with a heavy nucleus and produces smaller nuclei. Fusion starts with light nuclei and produces a heavier nucleus. Radioactive decay starts with an unstable nucleus and changes it spontaneously into a more stable form.
| Process | Starting nucleus or nuclei | Main change in composition | Typical products | Energy release idea |
|---|---|---|---|---|
| Fission | Heavy nucleus | One large nucleus splits | Two smaller nuclei, neutrons | Products are more tightly bound than the original heavy nucleus |
| Fusion | Light nuclei | Two small nuclei join | Heavier nucleus, sometimes neutron | Product nucleus is more tightly bound than the starting light nuclei |
| Radioactive decay | Unstable nucleus | Nucleus emits particle or radiation | New nucleus plus alpha, beta, or gamma emission | Nucleus moves to a more stable, lower-energy state |
Table 1. Comparison of nuclear processes by starting conditions, composition changes, products, and qualitative energy release.
The models in [Figure 2], [Figure 3], and [Figure 4] reveal a unifying theme: nuclear energy comes from changes in binding and stability, not from ordinary chemical bonds.
Another important comparison is predictability. A fission chain reaction can be engineered and controlled in a reactor. Fusion can be sustained naturally in stars but is difficult to control on Earth. Radioactive decay is spontaneous and probabilistic for individual nuclei, though large samples show predictable patterns over time.
Nuclear science affects daily life more than many students realize. Nuclear power plants use controlled fission to generate electricity for cities, schools, hospitals, and transportation systems. Smoke detectors often use radioactive materials to help detect particles from smoke. In medicine, radioactive isotopes can be used for imaging organs or targeting certain cancers with radiation.
Radiation therapy uses carefully measured doses of radiation to damage cancer cells. Medical imaging can use gamma-emitting tracers to reveal how organs function. These technologies depend on understanding exactly how nuclei change and what type of radiation is emitted.
Safety is essential because nuclear radiation can also damage healthy tissue. Shielding depends on the type of radiation: alpha particles are stopped easily, beta particles require more protection, and gamma rays are the most penetrating of the three covered here. The visual comparison in [Figure 4] helps explain why different types of radiation require different handling strategies.
"The release of atomic energy has changed everything except our way of thinking."
— Albert Einstein
Understanding nuclear processes also matters in Earth science and space science. Radioactive decay helps scientists date rocks and fossils indirectly through isotopes. Fusion explains how stars produce light and how heavier elements are eventually formed in the universe. Fission and decay both play roles in discussions about energy policy, waste management, and environmental impact.
The most powerful takeaway from nuclear models is that tiny particles inside the nucleus determine large-scale effects in the world. Electricity grids, medical treatments, radiation detectors, and the light from the Sun all depend on changes in nuclear composition and nuclear energy.