A forest, a loaf of bread rising on a counter, and a runner sprinting the final stretch of a race all depend on the same big idea: living things survive by transforming energy. No organism can break the laws of physics. Cells cannot create energy from nothing. Instead, they capture it, store it, transfer it, and release it. Two of the most important processes that make life possible are photosynthesis and cellular respiration. Together, they power most life processes on Earth and connect individual cells to entire ecosystems.
Every living thing needs energy to grow, repair damage, move materials, reproduce, and maintain internal balance. Cells do not usually use sunlight or food directly for most of their work. Instead, they rely on a molecule called ATP, which acts like a short-term energy carrier. When a cell needs to build a protein, transport ions across a membrane, or contract a muscle fiber, ATP provides usable energy.
ATP and usable energy
ATP, or adenosine triphosphate, stores energy in chemical bonds. When one phosphate group is removed, ATP becomes ADP and releases energy that cells can use. Photosynthesis helps build energy-rich molecules, and cellular respiration helps transfer that stored energy into ATP.
In ecosystems, energy enters mostly as sunlight. Producers such as plants, algae, and some bacteria capture part of that energy and store it in sugars. Consumers obtain that stored chemical energy by eating producers or other consumers. Decomposers break down dead matter and wastes, returning materials to the environment. Energy flows through this system, but matter such as carbon, oxygen, and water cycles through it again and again.
Atoms are rearranged in chemical reactions, but they are not created or destroyed. That idea helps explain why the atoms in a glucose molecule made by a leaf may later become part of a carbon dioxide molecule released by an animal or decomposer.
Because life depends on chemical reactions, it is useful to track both matter and energy. Photosynthesis stores energy in glucose, while respiration releases much of that energy in a controlled way. These are not opposite in every detail, but they are deeply linked.
[Figure 1] In plants and algae, chloroplasts capture light energy and transform it into chemical energy. Photosynthesis uses carbon dioxide from the air, water from the environment, and light energy from the Sun to make glucose and oxygen.
The overall chemical equation for photosynthesis is:
\[6\textrm{CO}_2 + 6\textrm{H}_2\textrm{O} + \textrm{light energy} \rightarrow \textrm{C}_6\textrm{H}_{12}\textrm{O}_6 + 6\textrm{O}_2\]
This equation means that six molecules of carbon dioxide and six molecules of water are rearranged using light energy to form one molecule of glucose and six molecules of oxygen. Glucose is an energy-rich organic molecule. Oxygen is released as a product and can be used by many organisms in respiration.
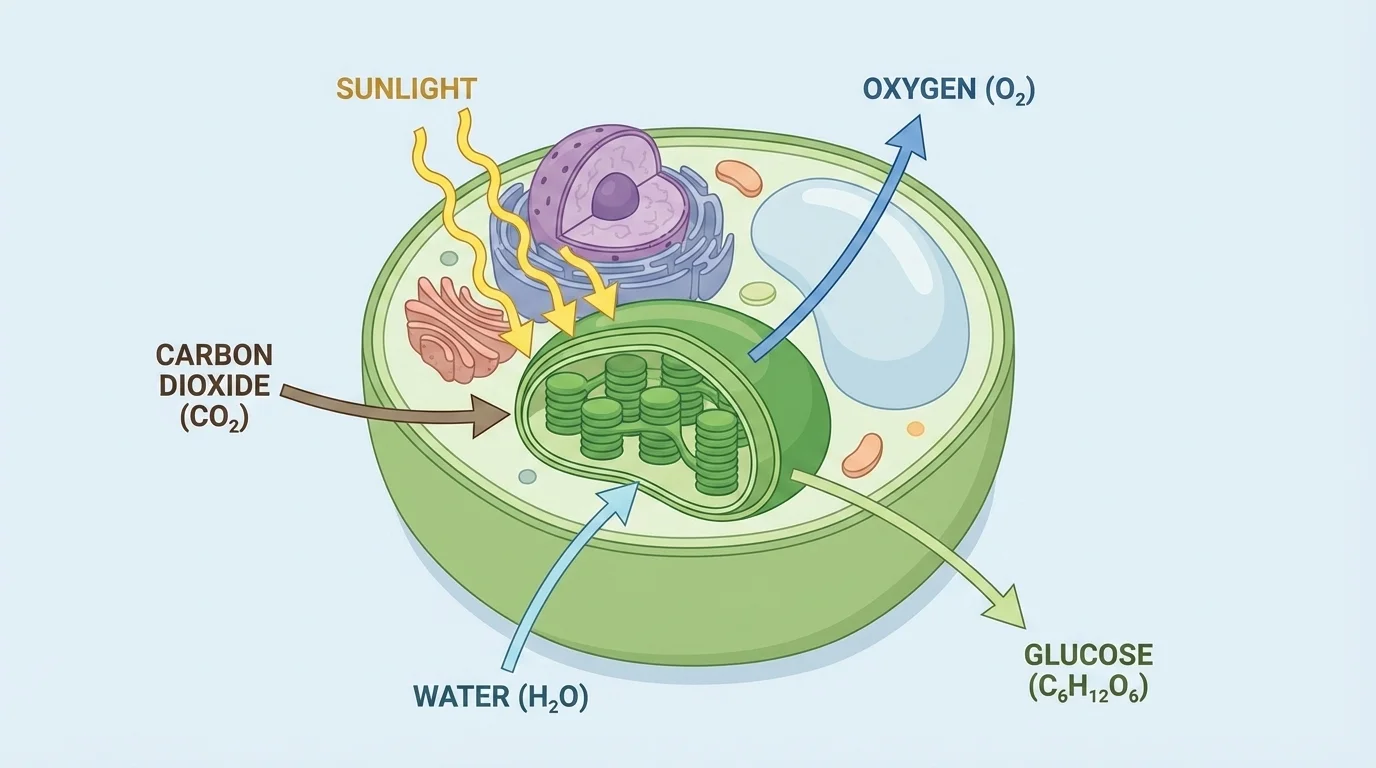
Photosynthesis does not happen equally in every part of a plant. It occurs mainly in green tissues, especially leaves. Inside leaf cells, chloroplasts contain the pigment chlorophyll, which absorbs certain wavelengths of light. Chlorophyll absorbs red and blue light especially well and reflects much of the green light, which is why many plants appear green.
Photosynthesis is the process by which plants, algae, and some bacteria use light energy to convert carbon dioxide and water into glucose and oxygen. Glucose is a simple sugar that stores chemical energy in its bonds.
A quick numerical example helps show the equation's meaning. If a plant uses \(12\) molecules of \(\textrm{CO}_2\), the balanced equation indicates it would need \(12\) molecules of \(\textrm{H}_2\textrm{O}\) and could produce \(2\) molecules of \(\textrm{C}_6\textrm{H}_{12}\textrm{O}_6\) and \(12\) molecules of \(\textrm{O}_2\). In ratio form, the equation is based on \(6:6:1:6\).
[Figure 2] Photosynthesis is not a single event. It is a series of linked reactions that occur in two major stages: the light-dependent reactions and the Calvin cycle. These stages take place in different parts of the chloroplast.
In the light-dependent reactions, chlorophyll and other pigments absorb light. This energy excites electrons and drives reactions that split water molecules. The splitting of water provides electrons and hydrogen ions, and it releases oxygen gas. These reactions also produce ATP and another energy-carrying molecule called NADPH.
The simplified water-splitting relationship can be written as:
\[2\textrm{H}_2\textrm{O} \rightarrow \textrm{O}_2 + 4\textrm{H}^+ + 4e^-\]
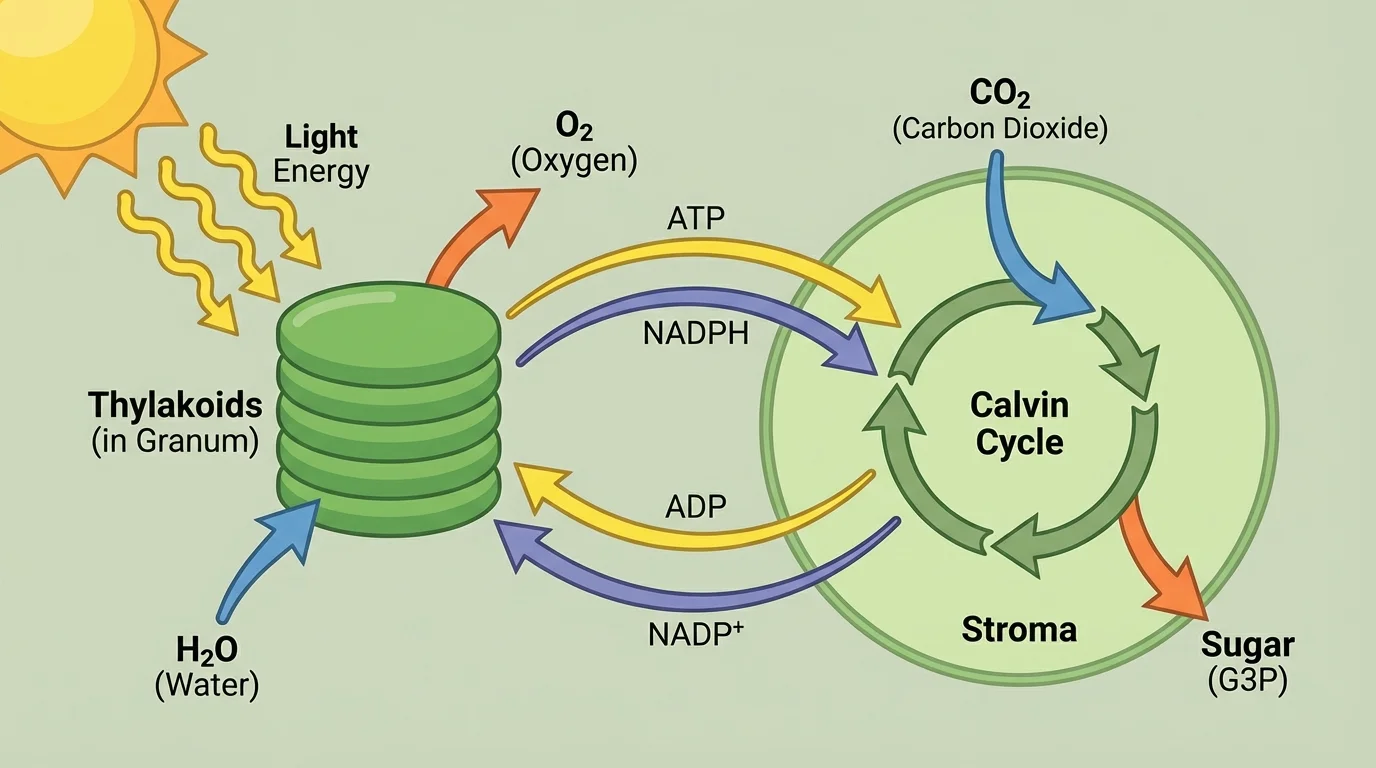
In the Calvin cycle, the cell uses carbon dioxide along with ATP and NADPH to build sugar molecules. This stage does not directly require light, but it depends on the products of the light-dependent reactions. Carbon atoms from \(\textrm{CO}_2\) become part of larger organic molecules. Over many steps, the chloroplast produces molecules that can be assembled into glucose and other carbohydrates.
This two-stage design is important. Light energy is first converted into temporary chemical forms, then those forms drive the construction of energy-rich sugars. The process is elegant because it links physics, chemistry, and biology in one coordinated system.
Earth's atmospheric oxygen is largely a product of photosynthetic organisms. Over geologic time, photosynthesis transformed the planet's atmosphere and made complex aerobic life possible.
Later, when we compare energy release in cells, [Figure 2] remains useful because it shows that glucose is not made from nowhere. Its chemical energy ultimately comes from sunlight captured earlier in the chloroplast.
Cells need a way to release the energy stored in glucose in manageable steps. That process is called cellular respiration. In aerobic respiration, cells use oxygen to break down glucose and transfer much of its stored energy to ATP.
The overall equation for aerobic cellular respiration is:
\[\textrm{C}_6\textrm{H}_{12}\textrm{O}_6 + 6\textrm{O}_2 \rightarrow 6\textrm{CO}_2 + 6\textrm{H}_2\textrm{O} + \textrm{energy}\]
The released energy is captured mainly in ATP, while carbon dioxide and water are produced as waste products. Notice how the reactants and products relate to photosynthesis. The products of one process are the reactants of the other, although the pathways and energy changes are very different.
Mitochondria as energy-transforming organelles
In eukaryotic cells, much of aerobic respiration occurs in the mitochondria. These organelles contain folded inner membranes that provide large surface areas for reactions that generate ATP efficiently.
A numerical comparison makes the relationship clearer. One molecule of glucose contains the same atoms before and after respiration, but they are rearranged into \(6\) molecules of \(\textrm{CO}_2\) and \(6\) molecules of \(\textrm{H}_2\textrm{O}\). The key change is not that atoms disappear, but that energy stored in the glucose bonds is transferred into ATP and released as heat.
Aerobic respiration is a multi-step pathway rather than one single reaction. The three major stages usually emphasized are glycolysis, the Krebs cycle, and the electron transport chain.
[Figure 3] Glycolysis occurs in the cytoplasm. It splits one glucose molecule into two smaller molecules called pyruvate. Glycolysis does not require oxygen directly and produces a small amount of ATP. In many textbooks, the net gain is about \(2\) ATP per glucose.
The Krebs cycle takes place in the mitochondrion after pyruvate is further processed. Carbon atoms are released as \(\textrm{CO}_2\), and high-energy electrons are transferred to carrier molecules. These carriers deliver electrons to the electron transport chain.
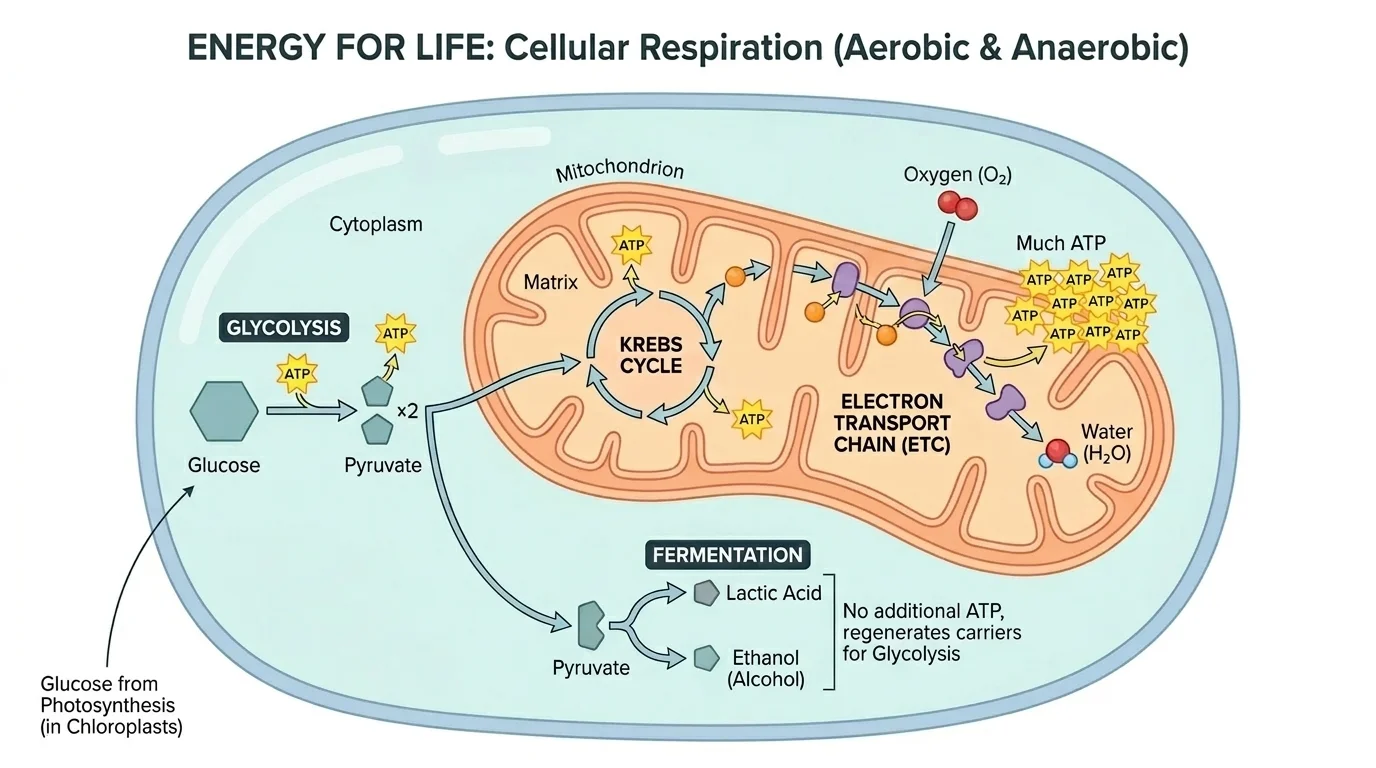
In the electron transport chain, electrons move through membrane proteins. Their energy helps pump hydrogen ions across the inner mitochondrial membrane. As hydrogen ions flow back through ATP synthase, large amounts of ATP are made. Oxygen is the final electron acceptor. Without oxygen, this chain stops operating normally.
A commonly cited total ATP yield per glucose in school-level biology is roughly \(30\) to \(32\) ATP in eukaryotic cells, though the exact number can vary. For a simple numeric contrast, if glycolysis gives about \(2\) ATP and total aerobic respiration gives about \(32\) ATP, then the subsequent stages provide about \(30\) additional ATP.
Energy yield comparison
A student compares ATP production from one glucose molecule in two situations: aerobic respiration yields about \(32\) ATP, while glycolysis alone yields \(2\) ATP.
Step 1: Identify the values
Aerobic total: \(32\) ATP. Glycolysis only: \(2\) ATP.
Step 2: Find the difference
\(32 - 2 = 30\)
Step 3: Interpret the result
Aerobic respiration provides about \(30\) more ATP per glucose than glycolysis alone.
This helps explain why oxygen is so valuable for organisms with high energy demands.
When athletes breathe heavily after intense exercise, they are trying to deliver oxygen efficiently and remove carbon dioxide. The cell-level events shown earlier in the diagram scale up to whole-body performance.
[Figure 4] When oxygen is limited or absent, cells cannot rely on the full aerobic pathway. Instead, some organisms and some cells use anaerobic processes. These pathways allow glycolysis to continue, but they produce far less ATP than aerobic respiration.
One important anaerobic process is fermentation. Fermentation regenerates the molecules needed for glycolysis so that ATP production can continue without oxygen. However, the ATP yield is low because only glycolysis contributes directly, giving about \(2\) ATP per glucose.
In lactate fermentation, pyruvate is converted into lactate. This occurs in certain bacteria and in human muscle cells during intense activity when oxygen delivery cannot keep up with demand. The process can be summarized as:
\[\textrm{glucose} \rightarrow 2\textrm{lactate} + \textrm{small amount of ATP}\]
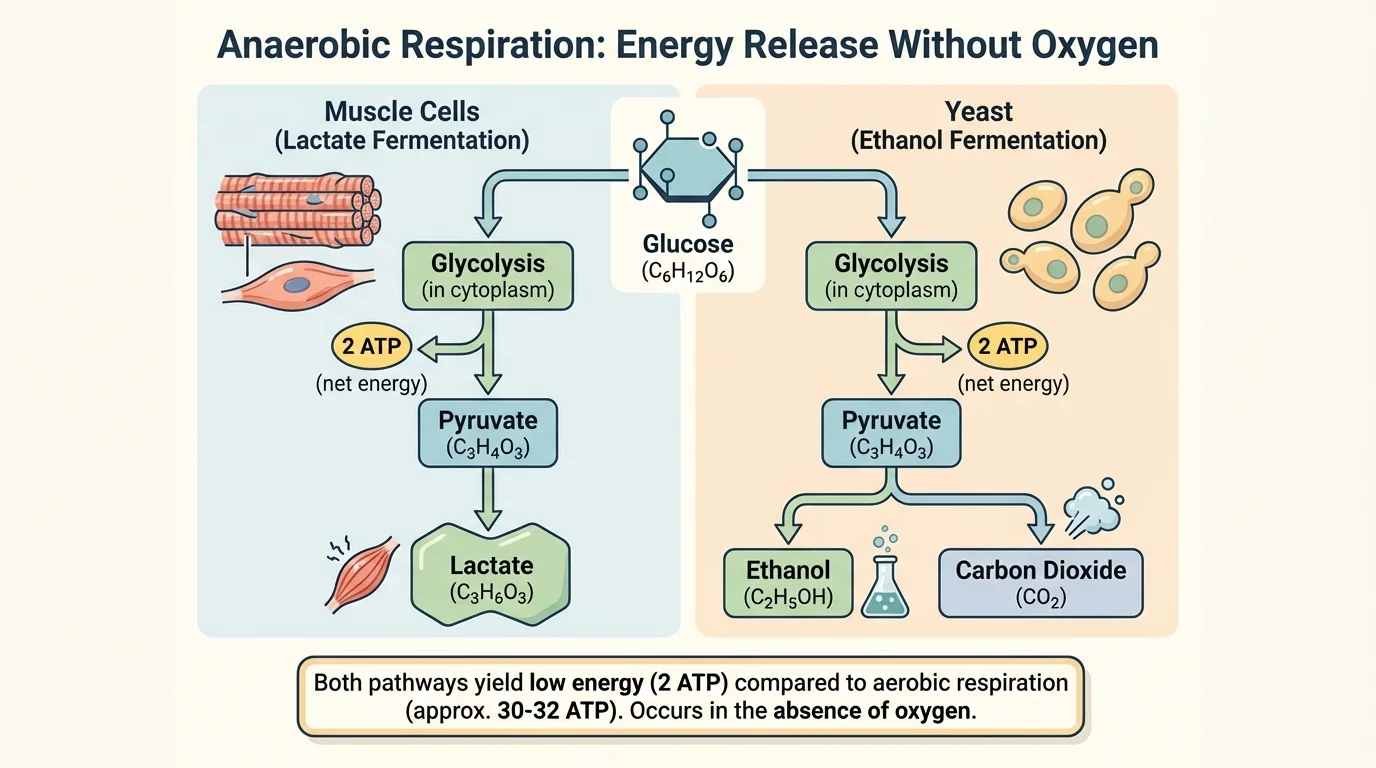
In alcoholic fermentation, used by yeast and some microorganisms, pyruvate is converted into ethanol and carbon dioxide. A simplified form is:
\[\textrm{glucose} \rightarrow 2\textrm{ethanol} + 2\textrm{CO}_2 + \textrm{small amount of ATP}\]
This is why bread dough rises: yeast release \(\textrm{CO}_2\) gas, which forms bubbles in the dough. It is also why fermentation is important in food production and some industrial processes.
Real-world case: sprinting muscles
During a short, intense sprint, a runner's muscles may require ATP faster than oxygen can be supplied.
Step 1: Demand rises sharply
Muscle cells use ATP rapidly for contraction.
Step 2: Oxygen becomes limiting
The electron transport chain cannot maintain maximum ATP production.
Step 3: Fermentation helps temporarily
Cells continue glycolysis and regenerate needed molecules through lactate fermentation.
This keeps ATP production going briefly, but it is not efficient enough for long-term, high-output activity.
The contrast in [Figure 4] matters because both fermentation types solve the same basic problem: how to keep energy production going when oxygen is not available for full aerobic respiration.
[Figure 5] At the ecosystem level, photosynthesis and respiration are tightly connected. Producers capture energy and build biomass. Consumers and decomposers obtain that biomass and release energy from it through respiration.
This relationship helps explain why ecosystems depend on a constant input of energy. Sunlight enters. Photosynthetic organisms store part of that energy in organic molecules. As organisms eat, grow, move, and reproduce, some of that energy is transferred, and much is lost as heat at each level.
The carbon cycle is also linked to these processes. Photosynthesis removes \(\textrm{CO}_2\) from the atmosphere or water and incorporates carbon into sugars and other biomolecules. Respiration returns much of that carbon to the environment as \(\textrm{CO}_2\). Decomposition does the same with dead organisms and wastes.
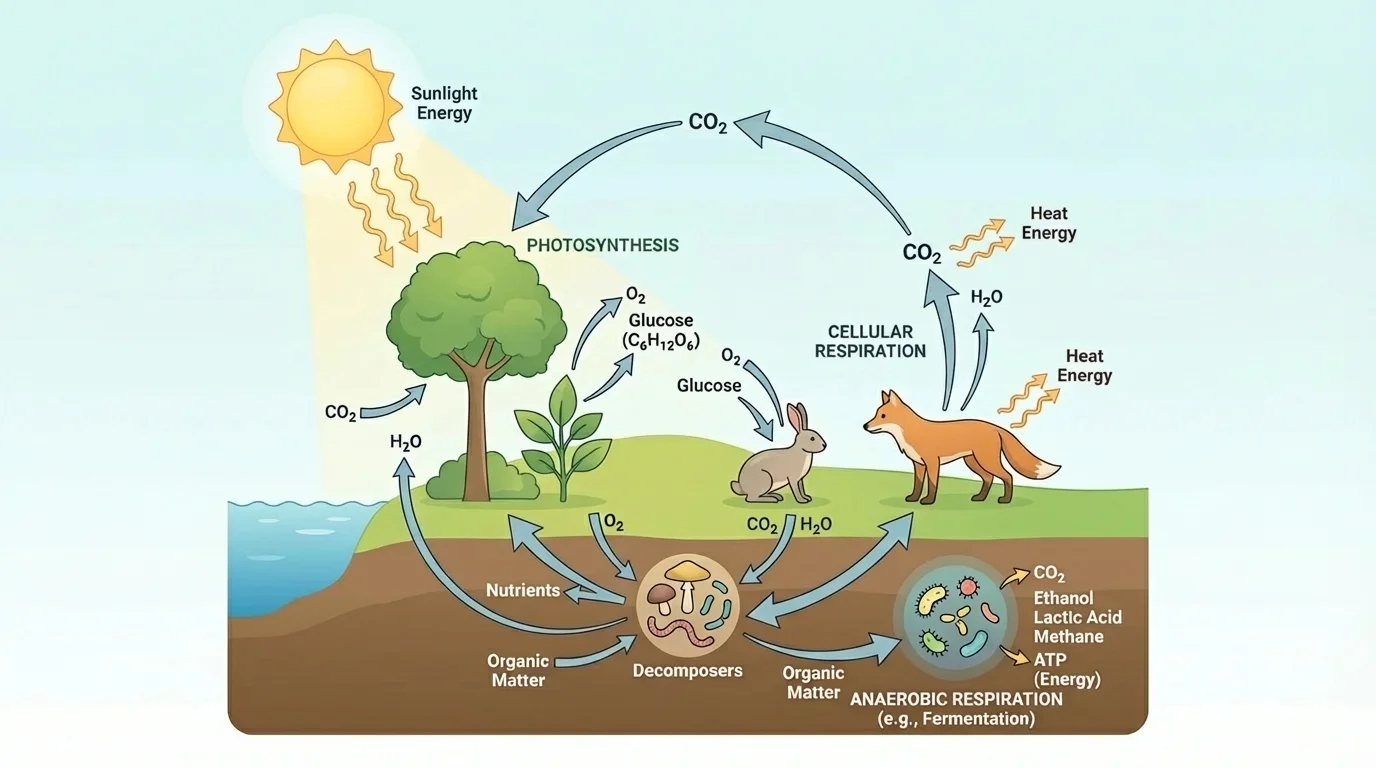
Plants are especially important because they form the base of most food webs. They do not just feed themselves; they support herbivores, predators, scavengers, and decomposers. In aquatic ecosystems, algae and photosynthetic plankton play a similar role.
| Process | Main Purpose | Typical Location | Uses | Produces |
|---|---|---|---|---|
| Photosynthesis | Store energy in glucose | Chloroplasts | \(\textrm{CO}_2\), \(\textrm{H}_2\textrm{O}\), light | \(\textrm{C}_6\textrm{H}_{12}\textrm{O}_6\), \(\textrm{O}_2\) |
| Aerobic respiration | Release energy into ATP | Cytoplasm and mitochondria | \(\textrm{C}_6\textrm{H}_{12}\textrm{O}_6\), \(\textrm{O}_2\) | \(\textrm{CO}_2\), \(\textrm{H}_2\textrm{O}\), ATP |
| Fermentation | Allow glycolysis without oxygen | Cytoplasm | Glucose-derived molecules | Lactate or ethanol, \(\textrm{CO}_2\) in some cases, small ATP yield |
Later, climate discussions often return to the ecosystem diagram, because changing rates of photosynthesis and respiration can affect atmospheric carbon dioxide levels and therefore influence global systems.
Photosynthesis depends on environmental conditions. Light intensity matters because light provides the energy that drives the first stage. Carbon dioxide concentration matters because carbon is needed to build sugars. Temperature matters because the enzymes involved have optimal ranges. Water availability also matters, especially in land plants.
For example, if light intensity increases from a low level, the rate of photosynthesis often rises. But it does not increase forever. Eventually another factor becomes limiting, such as \(\textrm{CO}_2\) concentration or temperature. This is an important biological example of interacting variables.
Numeric example: limiting factors
A plant is measured under two conditions. Under low light it produces \(4\) units of oxygen per hour. After light intensity is increased, it produces \(9\) units per hour. Later, more light is added again, but oxygen production stays at \(9\) units per hour.
Step 1: Compare the first change
\(9 - 4 = 5\)
Step 2: Interpret the increase
The first increase in light raised photosynthesis by \(5\) units per hour.
Step 3: Explain the plateau
When output remains at \(9\) units per hour despite more light, another factor has become limiting.
This could be low carbon dioxide, unsuitable temperature, or limited enzyme activity.
Respiration is also affected by conditions. Temperature influences enzyme activity in respiration too. Oxygen availability is especially important for aerobic respiration. In active tissues, the rate of respiration can increase because ATP demand rises. That is why heart rate and breathing rate often increase during exercise.
Cells do not switch between pathways because one is philosophically better than another. They respond to conditions. If oxygen is available and the cell has mitochondria, aerobic respiration is usually favored because it yields much more ATP. If oxygen is scarce, fermentation may help maintain short-term energy production.
Photosynthesis and respiration are not just classroom topics. Farmers depend on photosynthesis when managing crop growth. Greenhouses may adjust light, temperature, and carbon dioxide to increase productivity. Plant scientists study how drought or heat affects photosynthesis because food security depends on it.
Much of the oxygen produced by photosynthesis each year comes from ocean organisms rather than forests alone. Tiny photosynthetic plankton have a global impact far greater than their size suggests.
In medicine and sports science, respiration and anaerobic metabolism help explain fatigue, endurance, and recovery. Doctors monitor oxygen use, carbon dioxide production, and blood lactate levels to understand how well tissues are functioning. Trainers design workouts that challenge both aerobic and anaerobic systems.
Biotechnology also uses these ideas. Yeast fermentation is used in baking and beverage production. Scientists engineer microorganisms to improve industrial fermentation, biofuel production, and waste treatment. In environmental science, measurements of photosynthesis and respiration help estimate carbon storage in forests, grasslands, and oceans.
One common misunderstanding is that plants only do photosynthesis. In reality, plants also perform respiration all the time because their cells need ATP just like animal cells do. During daylight, a plant may do both photosynthesis and respiration at once. If photosynthesis exceeds respiration, the plant has a net gain of stored chemical energy.
Another misunderstanding is that respiration means breathing. Breathing is the physical movement of air into and out of an organism. Cellular respiration is the chemical process by which cells transfer energy from food molecules to ATP. Breathing supports respiration in many animals, but the two are not identical.
"Energy cannot be created or destroyed, only transformed."
— First Law of Thermodynamics
A final point of precision matters for ecosystems: energy flows, but matter cycles. Sunlight enters an ecosystem and is gradually lost as heat after being transformed through food webs. Carbon atoms, oxygen atoms, and water molecules, however, are reused in cycles that link atmosphere, organisms, soils, and water.