Some of the most important chemicals in the world are made in systems that never go fully to completion. Fertilizers, medicines, fuels, and industrial acids are often produced in reactions that can move forward and backward. That creates a challenge for chemists and engineers: if a reaction settles into equilibrium before enough product forms, how can the system be adjusted to make more? The answer is not random trial and error. It comes from understanding how molecules collide, how atoms rearrange, and how the system responds when conditions change.
In many chemical processes, reactants form products, but the products can also react to form the original reactants again. When that happens, the reaction is called reversible. A familiar industrial example is the Haber process:
\[N_2(g) + 3H_2(g) \rightleftharpoons 2NH_3(g)\]
In this reaction, nitrogen and hydrogen form ammonia, but ammonia can also break apart and reform nitrogen and hydrogen. If a factory wants more ammonia, it must refine the system by changing a condition that pushes the equilibrium toward the product side.
This kind of problem is a design problem. The goal is not just to know that equilibrium exists, but to decide which one-variable change will increase the amount of product present when the system settles again.
Chemical reactions happen because particles collide. For a reaction to occur, the collisions must have the right orientation and enough energy to break old bonds and form new ones. Equilibrium does not cancel these collisions; it means both directions continue at matching rates.
To improve product yield, you need to know which side of the reaction is favored when the system is disturbed. This idea is the foundation of chemical process design at equilibrium.
A dynamic equilibrium is reached when the forward reaction and reverse reaction occur at the same rate, as [Figure 1] shows at the particle level. The amounts of reactants and products stay constant, but the particles are still reacting. Equilibrium is not a frozen state. It is active and ongoing.
This happens only in a closed system, where matter does not escape. If product gas leaks out of the container, the system is no longer behaving as a true equilibrium system because the amounts are being changed from outside.
At equilibrium, you may still have a large amount of reactant left, a large amount of product, or a mixture of both. The exact amounts depend on the reaction and the conditions.
It helps to think of equilibrium like two busy escalators moving at the same speed in opposite directions. People are still moving on both escalators, but the number of people on each floor stays the same. In a chemical equilibrium, molecules continue changing in both directions, but the overall amounts no longer change.
A reversible reaction is a reaction that can proceed in both forward and reverse directions.
Equilibrium is the state in which the forward and reverse reaction rates are equal, so the amounts of reactants and products remain constant.
Once equilibrium is established, changing conditions can disturb it. The system then responds by shifting in the direction that reduces that disturbance.
Le Châtelier's principle states that when a system at equilibrium experiences a stress, the system shifts in the direction that opposes the stress. A stress can be a change in concentration, pressure, volume, or temperature.
If you add more reactant, the system tends to use some of that added reactant, so it shifts toward products. If you remove product, the system tends to replace what was removed, so it also shifts toward products. If you increase pressure in some gas systems, the system may shift toward the side with fewer gas particles. If you change temperature, the shift depends on whether heat acts like a reactant or a product.
This principle does not mean the system "thinks." It is simply a pattern that comes from molecular behavior. When conditions change, collision frequencies and energy distributions change too, and the rates in the two directions are no longer equal. The system shifts until a new equilibrium is established.
Equilibrium shift means a change in amounts, not a stop in reaction. When equilibrium shifts to the right, the amount of products increases and the amount of reactants decreases until a new balance is reached. When it shifts to the left, the opposite happens. The reaction continues in both directions before and after the shift.
A key limit in analyzing these systems is that you only need to consider one variable at a time. That means if you are asked how to increase products, you choose one condition to change and justify that single change clearly.
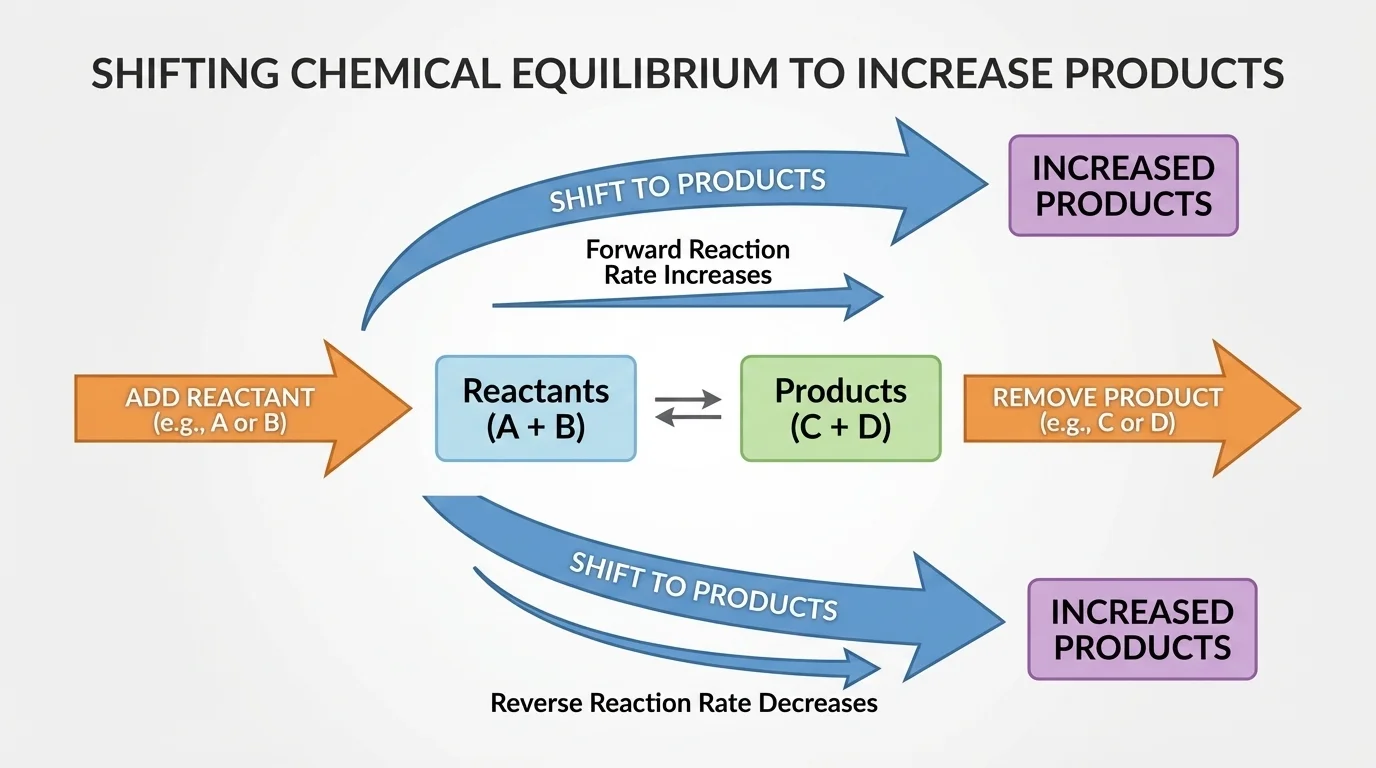
[Figure 2] Changing concentration is often the most direct way to increase products. If you add reactants, the system tends to consume some of them by shifting toward products. If you remove products, the system tends to replace them by shifting toward products as well.
Consider this equilibrium:
\[2SO_2(g) + O_2(g) \rightleftharpoons 2SO_3(g)\]
If more \(\textrm{SO}_2\) is added, the equilibrium shifts right, producing more \(\textrm{SO}_3\). If some \(\textrm{SO}_3\) is removed, the equilibrium also shifts right to replace the removed product.
This idea is used in industrial separation systems. If a product is continuously removed as it forms, more product can be made overall because the equilibrium keeps shifting toward the product side. In real plants, this can be done by cooling, condensing, filtering, or dissolving one substance out of the mixture.
Example: choosing a concentration change
For the reaction \(\textrm{H}_2(g) + \textrm{I}_2(g) \rightleftharpoons 2\textrm{HI}(g)\), specify one change that would increase the amount of \(\textrm{HI}\) at equilibrium.
Step 1: Identify the product side.
The product is \(\textrm{HI}\), which is on the right side of the equation.
Step 2: Choose a stress that makes the system shift right.
Adding more \(\textrm{H}_2\) or more \(\textrm{I}_2\) would increase reactant concentration. Removing \(\textrm{HI}\) would also work.
Step 3: State one acceptable design change.
Add more \(\textrm{H}_2\). The system shifts right to use some of the added reactant, so more \(\textrm{HI}\) forms.
Notice that this explanation does not require any calculation of equilibrium constants or concentrations. The reasoning is entirely qualitative: identify the stress, then predict the shift.
[Figure 3] Pressure and volume matter most when gases are involved, because changing the container size changes how often particles collide. In gas equilibria, increasing pressure by decreasing volume tends to shift the system toward the side with fewer total moles of gas. That side produces less crowding.
Look again at the Haber process:
\[N_2(g) + 3H_2(g) \rightleftharpoons 2NH_3(g)\]
The left side has \((1 + 3) = 4\) moles of gas. The right side has \(2\) moles of gas. If pressure increases, the equilibrium shifts to the right, toward ammonia, because that side has fewer gas particles.
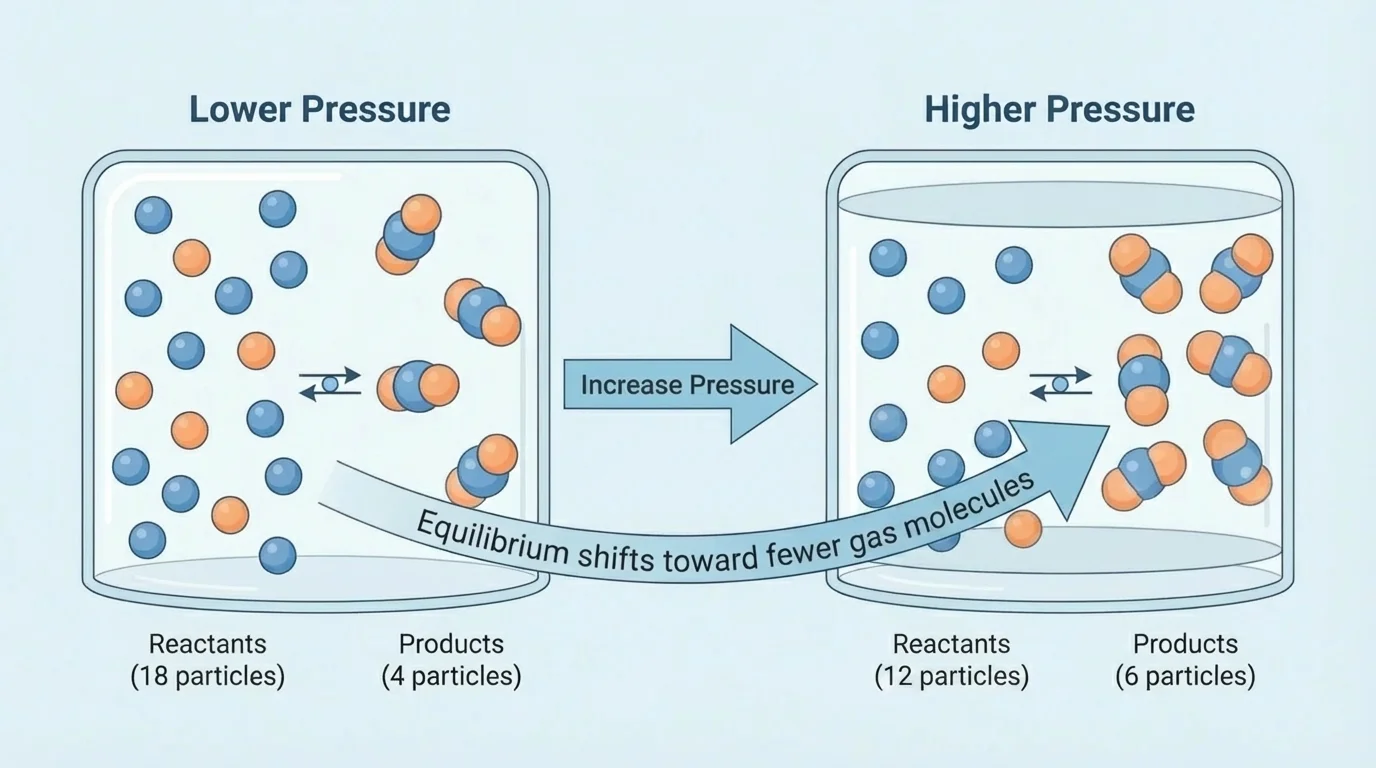
If pressure decreases by increasing volume, the equilibrium shifts toward the side with more gas particles. That is the opposite effect. So if your goal is to increase product, you must compare the total gas moles on each side before deciding.
This rule only works when the number of gas particles differs between the two sides. If both sides have the same total moles of gas, changing pressure or volume does not shift equilibrium.
| Reaction | Gas moles on left | Gas moles on right | Effect of increasing pressure |
|---|---|---|---|
| \(\textrm{N}_2 + 3\textrm{H}_2 \rightleftharpoons 2\textrm{NH}_3\) | \(4\) | \(2\) | Shifts right |
| \(\textrm{H}_2 + \textrm{I}_2 \rightleftharpoons 2\textrm{HI}\) | \(2\) | \(2\) | No shift |
| \(2\textrm{NO}_2 \rightleftharpoons \textrm{N}_2\textrm{O}_4\) | \(2\) | \(1\) | Shifts right |
Table 1. Comparison of how increasing pressure affects gas equilibria based on the number of gas moles on each side.
As seen earlier in [Figure 1], equilibrium is dynamic, so a pressure change does not stop the reaction. It changes which direction is temporarily favored until a new equilibrium is reached.
Example: choosing a pressure change
For the reaction \(\textrm{N}_2\textrm{O}_4(g) \rightleftharpoons 2\textrm{NO}_2(g)\), specify one change that would increase the amount of \(\textrm{NO}_2\) at equilibrium.
Step 1: Count gas moles on each side.
Left side: \(1\) mole of gas. Right side: \(2\) moles of gas.
Step 2: Decide which pressure change favors the product side.
The product side has more gas particles, so lowering pressure favors the right side.
Step 3: State the design change.
Increase the volume of the container, which lowers pressure. The equilibrium shifts right, producing more \(\textrm{NO}_2\).
A strong explanation connects pressure changes to particle crowding and the number of gas moles, not just to memorized arrows.
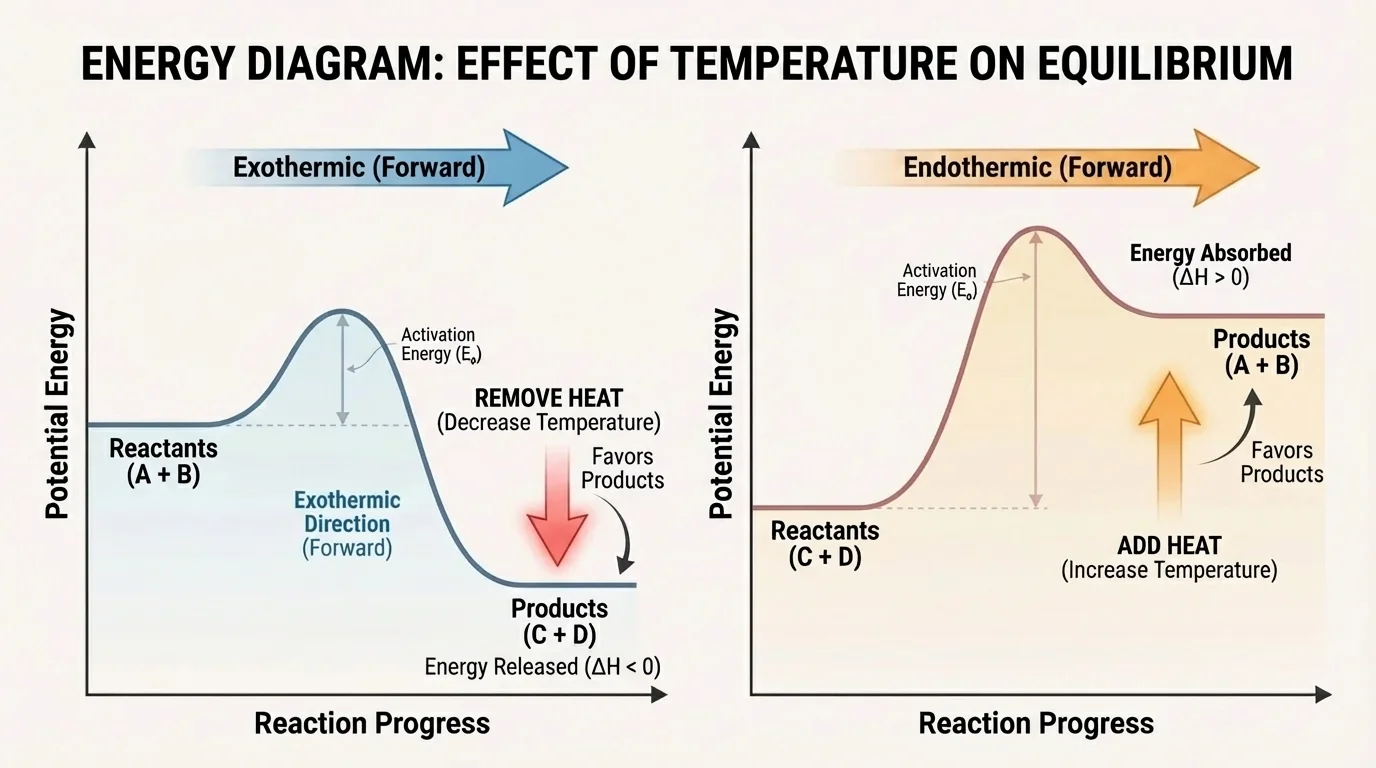
[Figure 4] Endothermic and exothermic behavior determine how temperature affects equilibrium. Temperature changes the energy balance of the reaction. You can think of heat as part of the reaction itself.
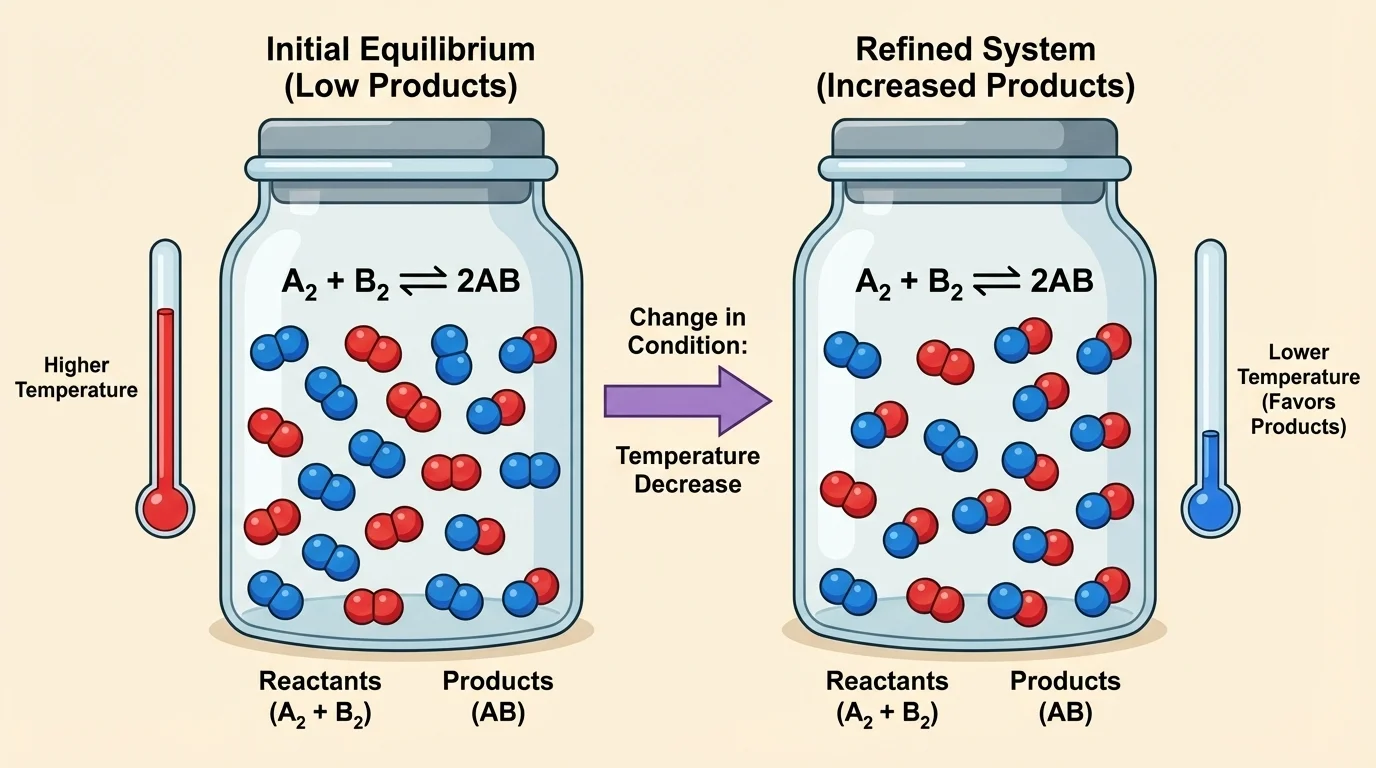
If the forward reaction is exothermic, heat behaves like a product. Adding heat by increasing temperature shifts equilibrium toward the reactants. Removing heat by decreasing temperature shifts equilibrium toward the products.
If the forward reaction is endothermic, heat behaves like a reactant. Increasing temperature shifts equilibrium toward the products. Decreasing temperature shifts equilibrium toward the reactants.
Consider this exothermic reaction:
\[N_2(g) + 3H_2(g) \rightleftharpoons 2NH_3(g) + heat\]
Because heat is on the product side, lowering temperature shifts equilibrium right and increases ammonia yield. Raising temperature shifts equilibrium left.
Now consider an endothermic example:
\[CaCO_3(s) + heat \rightleftharpoons CaO(s) + CO_2(g)\]
Here, heat is on the reactant side. Increasing temperature shifts equilibrium right, producing more \(\textrm{CaO}\) and \(\textrm{CO}_2\).
The same chemical system can respond very differently to temperature depending on whether the forward direction absorbs or releases energy. That is why chemists must know the energy change of the reaction before deciding whether heating will help product yield.
Temperature is especially important because it is the one common variable that can change the equilibrium amounts in a way directly tied to energy. This is why industrial systems often balance a temperature choice against other concerns such as speed, safety, and cost.
Example: choosing a temperature change
For the exothermic equilibrium \(\textrm{SO}_2(g) + \dfrac{1}{2}\textrm{O}_2(g) \rightleftharpoons \textrm{SO}_3(g) + \textrm{heat}\), specify one change that would increase the amount of \(\textrm{SO}_3\) at equilibrium.
Step 1: Identify the role of heat.
Heat is on the product side, so the forward reaction is exothermic.
Step 2: Decide how the system responds to temperature.
Lowering temperature removes heat, so the system shifts to replace it by making more products.
Step 3: State the design change.
Decrease the temperature. The equilibrium shifts right, increasing \(\textrm{SO}_3\).
As with concentration and pressure, only one variable is changed in this analysis. That keeps the cause-and-effect reasoning clear.
A catalyst speeds up a reaction by lowering the activation energy needed for successful collisions. Catalysts are extremely useful in industry, but they do not increase the amount of product present at equilibrium.
Why not? A catalyst speeds up both the forward and reverse reactions. That means equilibrium is reached faster, but the final equilibrium position does not change.
This is one of the most common mistakes in equilibrium questions. If the question asks how to increase the amount of product at equilibrium, adding a catalyst is not the correct answer.
Rate versus equilibrium position is a crucial distinction. A system can reach equilibrium quickly or slowly, but the equilibrium amounts depend on conditions such as concentration, pressure, volume, and temperature, not on the presence of a catalyst alone.
In a factory, a catalyst may still be essential because waiting too long for equilibrium would be inefficient. But if the design goal is specifically "more product at equilibrium," choose a variable that shifts equilibrium rather than one that only changes speed.
When you are asked to refine a chemical system to increase products, use a simple decision process. First, identify the product side. Next, identify which single variable you are allowed to change. Then predict whether that change shifts equilibrium right or left.
If the variable is concentration, add reactant or remove product. If the variable is pressure or volume, compare total gas moles on each side. If the variable is temperature, decide whether heat is a reactant or a product.
In many cases, more than one change could work, but you only need to specify one. The best answer is clear, specific, and justified with equilibrium reasoning.
For example, for \(\textrm{N}_2(g) + 3\textrm{H}_2(g) \rightleftharpoons 2\textrm{NH}_3(g) + \textrm{heat}\), several valid choices can increase ammonia at equilibrium: add \(\textrm{N}_2\), add \(\textrm{H}_2\), remove \(\textrm{NH}_3\), increase pressure, or decrease temperature. Each works for a different reason, but each shifts the system toward the product side.
"A chemical system at equilibrium responds to change by shifting toward a new balance."
— Central idea of equilibrium design
That statement captures the heart of the topic: design means selecting a condition that pushes the balance in the desired direction.
The Haber process is one of the most important examples because ammonia is used to make fertilizers that support food production around the world. Engineers use high pressure because, as discussed with gas moles and shown earlier in [Figure 3], higher pressure favors the side with fewer gas particles, which is the ammonia side.
Another example is the contact process for making sulfur trioxide, a step in sulfuric acid production:
\[2SO_2(g) + O_2(g) \rightleftharpoons 2SO_3(g) + heat\]
To increase \(\textrm{SO}_3\), chemists can use lower temperature because the forward reaction is exothermic, and they can remove \(\textrm{SO}_3\) as it forms to shift the equilibrium right.
Even in environmental chemistry, equilibrium matters. Dissolved gases in water, acid formation in the atmosphere, and mineral formation in caves all involve systems that shift when conditions such as concentration or temperature change.
One mistake is assuming equilibrium means equal amounts of reactants and products. It does not. It means equal rates.
Another mistake is forgetting to count only gases when predicting pressure effects. Solids and liquids are not counted in the simple gas-mole comparison used here.
A third mistake is treating catalysts as if they shift equilibrium. They do not increase product amount at equilibrium; they only help the system reach equilibrium faster.
A fourth mistake is changing more than one variable at once in your explanation. For this topic, focus on a single clear change and justify that one change only.
Finally, avoid jumping into calculations. You do not need equilibrium constants or concentration calculations here. The goal is to reason qualitatively from the reaction equation and the condition being changed.