Almost every meal you eat is connected to sunlight, even if there was no obvious plant on your plate. Bread, fruit, vegetables, meat, milk, cooking oils, and even many fuels all trace back to one extraordinary process: photosynthesis. Earth receives a vast amount of energy from the Sun, but most of it is not stored for later use. The main way solar energy is captured and stored on Earth is through photosynthesis, a complex chemical process that turns light energy into chemical energy.
Photosynthesis is far more than a biology topic. It is one of the central energy transformations on Earth. Plants, algae, and some bacteria absorb light energy and use it to build energy-rich organic molecules. Those molecules become food for the organisms themselves and, directly or indirectly, for nearly all other living things. Without photosynthesis, most ecosystems would collapse because the steady input of usable chemical energy would disappear.
It also shapes the atmosphere. Much of the oxygen in the air comes from photosynthetic organisms. When these organisms split water molecules during photosynthesis, oxygen is released as a byproduct. The oxygen that fills Earth's atmosphere and supports aerobic respiration in animals, plants, fungi, and many microbes is tied to this process.
The energy stored in fossil fuels originally came from sunlight captured by photosynthesis millions of years ago. Coal, oil, and natural gas are ancient biological matter transformed over long periods of time.
From the viewpoint of energy, photosynthesis is a conversion process. Light energy from the Sun is not destroyed; instead, it is transformed into chemical potential energy stored in the bonds of molecules such as glucose, \(\textrm{C}_6\textrm{H}_{12}\textrm{O}_6\). That stored energy can later be released through cellular respiration, combustion, or decomposition.
To understand photosynthesis clearly, it helps to separate matter from energy. Matter includes atoms and molecules such as carbon dioxide, water, oxygen, and glucose. Energy is not matter; it is transferred and transformed. In photosynthesis, the matter entering the process is mainly carbon dioxide, \(\textrm{CO}_2\), and water, \(\textrm{H}_2\textrm{O}\). The energy entering is light from the Sun.
The overall chemical equation is often written as:
\[6\textrm{CO}_2 + 6\textrm{H}_2\textrm{O} + \textrm{light energy} \rightarrow \textrm{C}_6\textrm{H}_{12}\textrm{O}_6 + 6\textrm{O}_2\]
This equation summarizes the overall change, but it hides many intermediate steps. The carbon atoms in glucose come from carbon dioxide, not from soil. The hydrogen atoms in glucose largely come from water. The oxygen gas released, \(\textrm{O}_2\), comes from water molecules that are split during the light-dependent reactions.
Chemical energy is energy stored in the arrangement of atoms and the bonds between them. Reactants are the starting substances in a chemical process, and products are the substances formed. In photosynthesis, carbon dioxide and water are reactants, while glucose and oxygen are products.
Because energy is stored in chemical bonds, photosynthesis creates molecules that can be used later. A simple way to express this is that low-energy inputs are reorganized into a higher-energy organic molecule with the help of sunlight. The process does not create energy from nothing; it stores incoming solar energy in matter.
A useful numerical comparison is the balanced equation itself. It shows that \(6\) molecules of \(\textrm{CO}_2\) combine with \(6\) molecules of \(\textrm{H}_2\textrm{O}\) to form \(1\) molecule of glucose and \(6\) molecules of \(\textrm{O}_2\). If a plant uses \(12\) molecules of \(\textrm{CO}_2\), then in the same overall ratio it can produce \(2\) molecules of glucose, assuming enough water and light are available.
[Figure 1] Photosynthesis in plants depends on specialized structures. Most photosynthesis occurs in leaves, especially in the mesophyll tissue, where cells are packed with organelles designed to capture light.
Inside those cells are chloroplasts, organelles that carry out photosynthesis. Chloroplasts contain stacks of internal membranes called thylakoids, surrounded by a fluid-filled region called the stroma. The thylakoid membranes hold pigments and proteins that absorb light and begin the energy-conversion process.
The most important pigment is chlorophyll. Chlorophyll absorbs mainly red and blue wavelengths of visible light and reflects more green light, which is why many plants look green. Other pigments, such as carotenoids, can also help capture light energy and protect the photosynthetic machinery.
Leaves are also built for exchange with the environment. Tiny openings called stomata allow carbon dioxide to enter and oxygen to leave. Water usually reaches the leaf through xylem tissue, moving upward from the roots. The structure of the leaf therefore supports both matter intake and light capture.
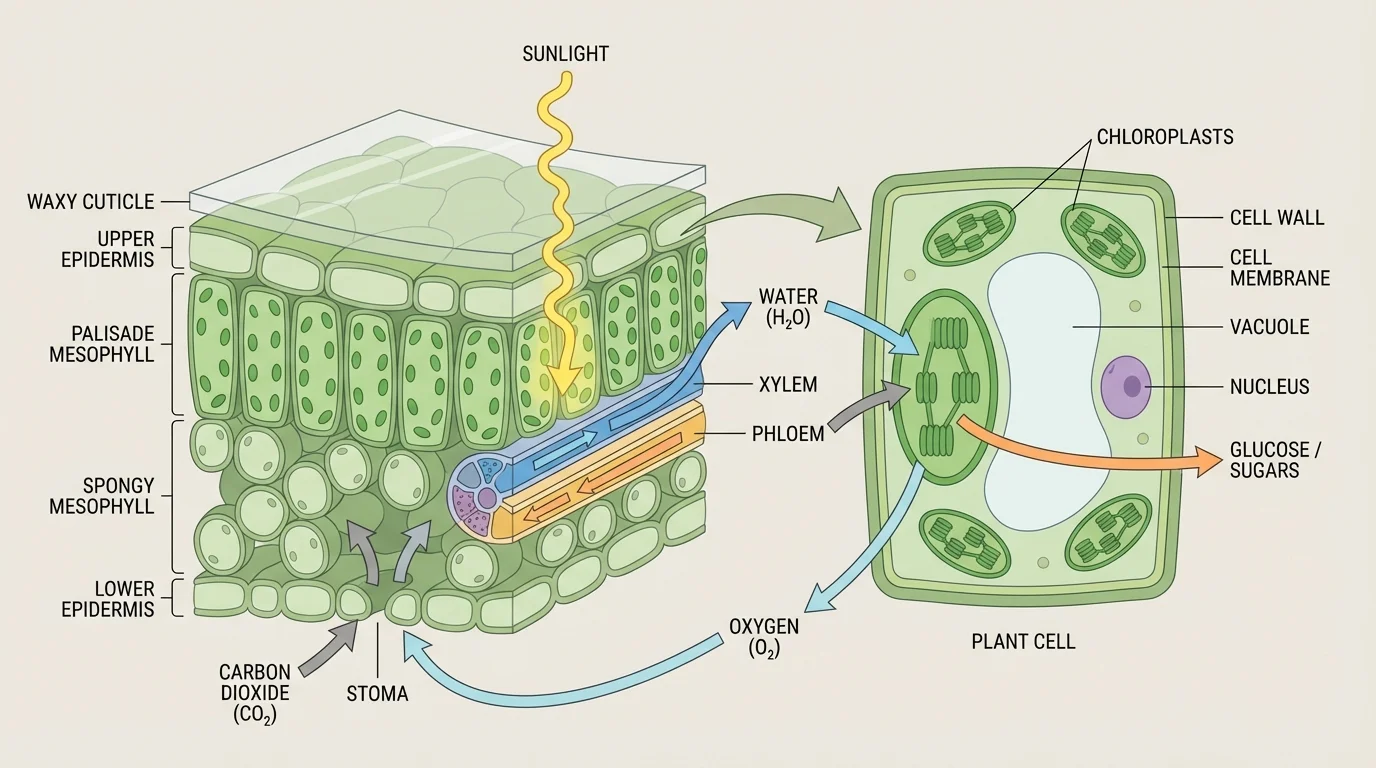
These structures reveal an important idea: photosynthesis is not just a chemical equation floating in isolation. It is a process made possible by anatomy and cell structure. A plant's broad leaf surface increases light absorption, while stomata regulate gas exchange, and chloroplast membranes organize the molecules that carry out the reactions.
Photosynthesis takes place in two linked stages, as [Figure 2] illustrates: the light-dependent reactions and the Calvin cycle. These stages are connected because the first stage captures light energy and temporarily stores it in chemical forms that power the second stage.
In the light-dependent reactions, which occur in the thylakoid membranes, chlorophyll absorbs photons. This excites electrons to higher energy states. The energized electrons move through a chain of proteins in the membrane, and their energy helps produce two important energy-carrying molecules: ATP and NADPH. Water is split during this stage, providing electrons and hydrogen ions, and oxygen gas is released.
The simplified splitting of water can be represented as part of a larger reaction sequence, but one key idea is that water is the source of the electrons replacing those lost by chlorophyll. This is why oxygen is released. When water molecules are broken apart, oxygen atoms can combine to form \(\textrm{O}_2\).
In the Calvin cycle, which occurs in the stroma, carbon dioxide is incorporated into organic molecules. Using ATP and NADPH from the light-dependent reactions, the plant builds higher-energy carbon compounds that can eventually be assembled into sugars such as glucose. The Calvin cycle does not directly need light at the moment it runs, but it depends on products made using light.
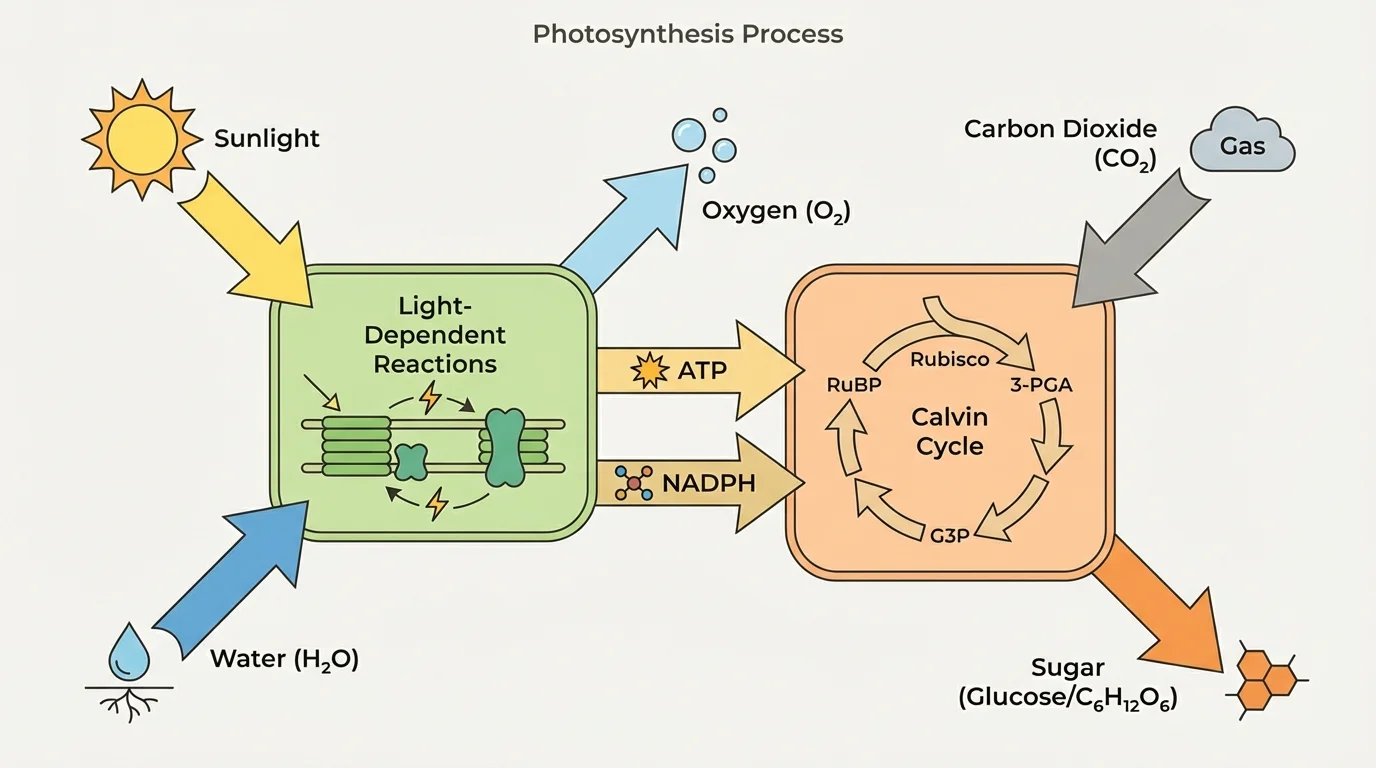
A common misunderstanding is that sunlight directly "turns into glucose" in one instant. In reality, the transformation happens through a series of carefully organized reactions. First, light energy is captured and converted into temporary chemical energy. Then that temporary chemical energy is used to build stable, energy-rich carbon compounds.
Why two stages are necessary
Light energy is powerful but hard for cells to store directly for long periods. Photosynthetic organisms first convert it into short-term chemical carriers, mainly ATP and NADPH. Those carriers then help drive the formation of sugars, which store energy more stably in chemical bonds and can be transported, stored, or used later by the organism.
This two-stage arrangement makes photosynthesis both efficient and controllable. It also helps explain why photosynthesis links energy transfer to matter rearrangement. The Sun provides the energy, but atoms from carbon dioxide and water are what actually become sugar.
The key energy idea is that photosynthesis stores energy in the bonds of organic molecules. Glucose is not simply "food" in the everyday sense; it is a compact package of carbon, hydrogen, and oxygen atoms arranged so that its bonds contain usable chemical potential energy. When cells later break down glucose during respiration, some of that stored energy becomes available for life processes.
This is why growing plant mass represents stored solar energy. A tree trunk, a grain of rice, a potato, and a leaf are all made of matter that has been assembled partly using energy captured from sunlight. If a plant produces starch from glucose, that starch also represents stored chemical energy. If an animal eats the plant, some of that stored energy is transferred into the animal's body.
Consider a simple ratio example using the overall equation. If a plant produces \(3\) molecules of glucose, then the balanced equation implies it must have used \(18\) molecules of \(\textrm{CO}_2\) and \(18\) molecules of \(\textrm{H}_2\textrm{O}\), while producing \(18\) molecules of \(\textrm{O}_2\). The numbers matter because they show that chemical energy storage is tied to specific rearrangements of matter.
Example: Using the balanced equation to track matter
Suppose a lab setup measures that an aquatic plant released \(24\) molecules of oxygen gas in an idealized model. How many molecules of carbon dioxide were used in the same overall photosynthesis ratio?
Step 1: Read the ratio from the equation
From \(6\textrm{CO}_2 + 6\textrm{H}_2\textrm{O} \rightarrow \textrm{C}_6\textrm{H}_{12}\textrm{O}_6 + 6\textrm{O}_2\), the ratio of \(\textrm{CO}_2\) used to \(\textrm{O}_2\) produced is \(6:6\), or \(1:1\).
Step 2: Apply the ratio
If \(24\) molecules of \(\textrm{O}_2\) are produced, then \(24\) molecules of \(\textrm{CO}_2\) are used.
The plant used 24 molecules of carbon dioxide in this simplified case.
The same logic helps scientists measure photosynthesis in ecosystems. By tracking oxygen production, carbon dioxide uptake, or biomass increase, they can estimate how much solar energy is being stored in living matter.
The rate of photosynthesis depends on environmental conditions, and [Figure 3] shows an important pattern: increasing one factor often raises the rate only until another factor becomes limiting. This idea is called a limiting factor. Once one resource runs short, adding more of something else may no longer help much.
Light intensity affects photosynthesis because light supplies the energy for the first stage. At low light intensity, increasing light usually increases the rate. But after a point, the rate levels off because the plant's enzymes, carbon dioxide supply, or temperature become the new limits.
Carbon dioxide concentration also matters because carbon dioxide supplies the carbon atoms for sugar production. In many situations, raising \(\textrm{CO}_2\) concentration increases photosynthesis up to a saturation point. Water availability matters too; without enough water, plants may close their stomata to reduce water loss, which also reduces carbon dioxide entry.
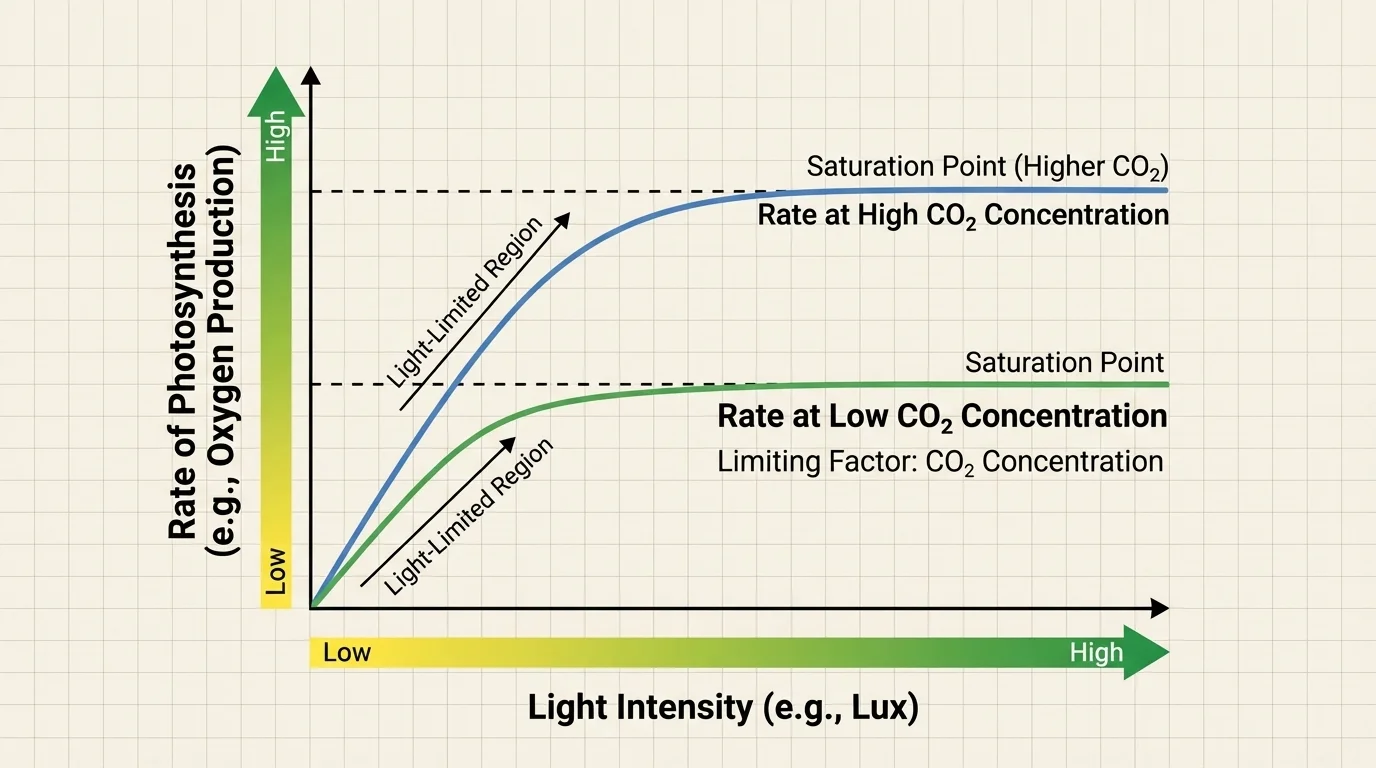
Temperature influences the enzymes that drive photosynthetic reactions. If temperature is too low, reactions proceed slowly. If temperature becomes too high, enzymes may lose effectiveness and the plant may experience water stress. The result is often a curved relationship, with an optimal range rather than "hotter is always better."
These ideas are important in agriculture. Farmers and greenhouse managers adjust light, water, temperature, and carbon dioxide to improve plant growth. In a greenhouse, increasing \(\textrm{CO}_2\) from a lower level to a more favorable one can raise photosynthetic productivity, but only if light, nutrients, and temperature are also suitable.
Example: A limiting-factor situation
A plant in dim light receives more carbon dioxide, but its growth changes very little. Why?
Step 1: Identify the possible limiting factor
If light intensity is very low, the light-dependent reactions cannot make ATP and NADPH fast enough.
Step 2: Connect that limit to the whole process
Even with more \(\textrm{CO}_2\), the Calvin cycle cannot speed up much because it depends on products made from light energy.
The main limit in this case is light intensity, not carbon dioxide.
Later in a different context, the pattern in [Figure 3] also helps explain why plant growth in nature can vary so strongly with season, cloud cover, drought, or atmospheric conditions.
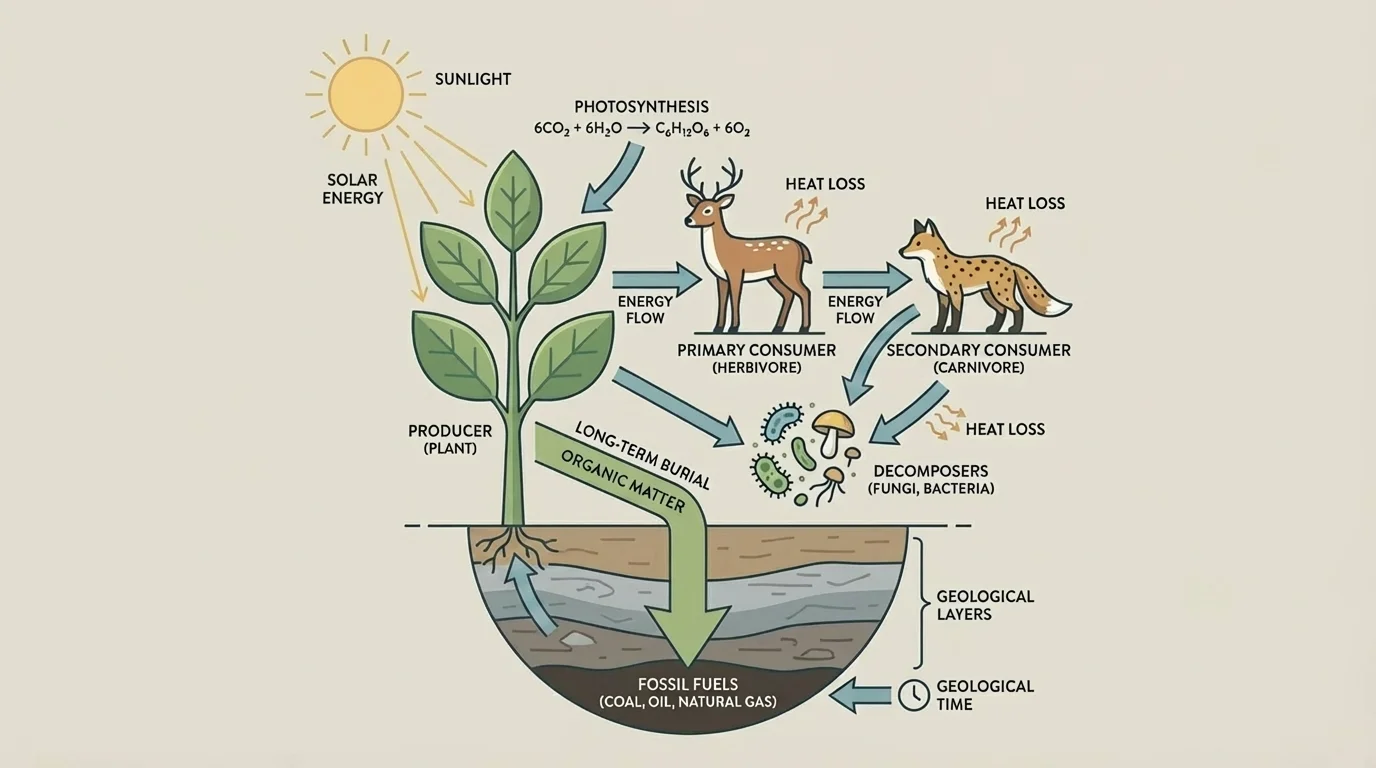
Nearly all ecosystem energy begins with photosynthesis. Producers such as plants and algae capture light energy and convert it into chemical energy. Herbivores obtain some of that stored energy by eating producers, and predators obtain some by eating other consumers.
As [Figure 4] shows, not all captured energy remains available at each step. Organisms use much of it for their own metabolism, movement, repair, and growth. That is why food webs usually support fewer organisms at higher levels. Still, the original stored energy came primarily from photosynthesis at the base of the system.
Photosynthesis is also central to agriculture. Crop yield depends on the ability of plants to capture light, take in carbon dioxide, absorb water and minerals, and convert these into biomass. Plant breeders and agricultural scientists study photosynthesis to improve food production, especially as climates change and populations grow.
Forests and oceans matter greatly in the global carbon cycle because photosynthetic organisms remove carbon dioxide from the atmosphere. Some of that carbon is quickly returned through respiration and decomposition, while some is stored longer in wood, soils, sediments, or eventually fossil fuels. This does not make photosynthesis a complete solution to climate change, but it is a major natural pathway for carbon uptake.
Human technology is increasingly inspired by this process. Solar panels capture light energy directly as electricity, while photosynthesis stores it chemically. Researchers study artificial photosynthesis and bioengineered algae in hopes of producing fuels or useful chemicals more sustainably.
Marine phytoplankton, tiny photosynthetic organisms in oceans, contribute a large fraction of Earth's oxygen production. Vast atmospheric effects can come from organisms too small to see individually.
This energy pathway also helps explain why fossil fuels are really ancient stored solar energy. They are not being replenished on human timescales, which is why burning them releases carbon that was stored over millions of years.
Photosynthesis and cellular respiration are closely connected. Photosynthesis stores energy by building glucose, while cellular respiration releases energy from glucose in a controlled way that cells can use. The overall respiration equation is often written as the reverse of the photosynthesis equation:
\[\textrm{C}_6\textrm{H}_{12}\textrm{O}_6 + 6\textrm{O}_2 \rightarrow 6\textrm{CO}_2 + 6\textrm{H}_2\textrm{O} + \textrm{energy}\]
Even though the equations appear opposite, the processes are not simply mirror images in every mechanistic detail. They use different pathways, enzymes, structures, and purposes. Photosynthesis captures and stores energy; respiration transfers energy from food molecules into forms the cell can readily use, especially ATP.
Plants do both. A common mistake is thinking that plants only photosynthesize and animals only respire. In fact, plant cells carry out cellular respiration as well, because plants also need usable energy day and night. Photosynthesis mainly occurs when light is available, but respiration continues in living cells at all times.
One way to observe photosynthesis directly is with aquatic plants. In bright light, some release visible oxygen bubbles. While counting bubbles is not a perfect measurement, it gives a rough indication of photosynthetic rate. Scientists use much more precise tools, such as oxygen sensors, carbon dioxide probes, and satellite measurements of plant productivity.
In crop science, understanding photosynthesis helps explain why leaf area, spacing, irrigation, and nutrient supply affect harvests. In ecology, photosynthesis helps explain why deserts have lower productivity than tropical rainforests, and why seasonal changes strongly affect temperate ecosystems.
Example: A simple observation setup
An aquatic plant is placed under a lamp at \(10\) centimeters and then at \(20\) centimeters. In many cases, the plant releases fewer oxygen bubbles per minute at \(20\) centimeters. Why?
Step 1: Identify the changing variable
The lamp is farther away, so the light intensity reaching the plant is lower.
Step 2: Connect light to photosynthesis
Lower light intensity usually reduces the rate of the light-dependent reactions.
Step 3: Link to the observation
With a lower photosynthetic rate, less oxygen is produced per unit time, so fewer bubbles are observed.
This is a simplified indicator of how light can affect photosynthetic output.
Modern climate science also depends on understanding photosynthesis. Satellites can detect patterns of vegetation and estimate how actively plants are absorbing carbon dioxide. These measurements help scientists study droughts, wildfire recovery, deforestation, and changing growing seasons.
Several misconceptions about photosynthesis are very common. One is that plants "get their food from the soil." Soil provides water and minerals, but most of a plant's mass comes from carbon fixed from atmospheric \(\textrm{CO}_2\). Another misconception is that oxygen released in photosynthesis comes from carbon dioxide. In fact, the released oxygen gas comes from water that is split in the light-dependent reactions.
Another misunderstanding is that sunlight itself becomes matter. It does not. Light provides energy, while atoms from carbon dioxide and water are rearranged into glucose and other molecules. Energy and matter are related in the process, but they are not the same thing.
Finally, photosynthesis is sometimes treated as something only important to plants. In reality, it is a foundation of Earth's energy system. Food webs, atmospheric oxygen, biomass growth, much of the carbon cycle, and even many human energy resources depend on it.
Energy can be transferred, stored, and transformed. Photosynthesis is a major example of transformation: radiant energy from sunlight becomes chemical potential energy stored in organic molecules.
When you look at a forest, a field of corn, or green algae floating on water, you are seeing one of the most important energy-capture systems on the planet. Through photosynthesis, Earth does not merely receive sunlight; living things convert part of that sunlight into stored chemical energy that powers ecosystems and supports life.