A single drop of water, a steel bicycle frame, the oxygen you breathe, and the sugar in a cookie seem completely different. Yet all of them are built from tiny particles called atoms. That is one of the most powerful ideas in science: huge differences in the world around us can come from tiny building blocks combining in different ways.
Everything you can touch, hold, pour, stretch, or smell is made of matter. Matter includes solids, liquids, and gases, and all of it is built from particles too small to see with your eyes alone. Even though these particles are tiny, the way they are put together determines whether a substance is hard like diamond, bendable like plastic, slippery like oil, or invisible like air.
The number of naturally occurring types of atoms is limited, but the number of substances they can form is enormous. This is a bit like letters in an alphabet. There are only so many letters, yet they can be arranged into a huge number of words. In matter, the "letters" are atoms, and the "words" are the substances they form.
You may already know that matter has mass and takes up space. A deeper idea is that matter is not continuous throughout. It is made of tiny particles, and the identity and arrangement of those particles matter.
Scientists use models to understand these particles because atoms and molecules are far too small to observe directly in ordinary classroom situations. Models are not perfect copies, but they help us picture how matter is built.
An atom is the smallest unit of an element that still has the properties of that element. A element is a pure substance made of only one kind of atom. For example, gold is made of gold atoms, oxygen gas is made from oxygen atoms, and helium is made of helium atoms.
Each type of atom is different from other types. Oxygen atoms are not the same as carbon atoms. Carbon atoms are not the same as iron atoms. These differences matter because they affect how atoms behave and what substances they can form.
Scientists represent elements with chemical symbols such as \(\textrm{H}\) for hydrogen, \(\textrm{O}\) for oxygen, \(\textrm{C}\) for carbon, and \(\textrm{Fe}\) for iron. These symbols are useful because they give a quick way to describe matter and chemical changes.
Atom is the smallest unit of an element that keeps that element's identity. Molecule is a group of atoms joined together. Compound is a pure substance made from two or more different kinds of atoms chemically joined in a fixed ratio.
Atoms are incredibly small. A speck of dust contains an enormous number of them. You do not need to memorize just how many, but it helps to realize that every visible object is made from unimaginably tiny parts.
Atoms often do not stay alone. They can join together to form a molecule. Some molecules are very small, with just two atoms, while others contain hundreds, thousands, or even more atoms linked together, as [Figure 1] illustrates. This range in size helps explain why substances can be so simple or so complex.
For example, oxygen in the air is commonly found as \(\textrm{O}_2\), which means each oxygen molecule contains \(2\) oxygen atoms. Water is \(\textrm{H}_2\textrm{O}\), which means each water molecule has \(2\) hydrogen atoms and \(1\) oxygen atom. Carbon dioxide is \(\textrm{CO}_2\), which means each molecule has \(1\) carbon atom and \(2\) oxygen atoms.
Some molecules are much larger. Many plastics are made of long chains of repeating atoms. Proteins in your body are large molecules built from many smaller pieces joined together. DNA, the molecule that carries genetic information, contains thousands to millions of atoms.
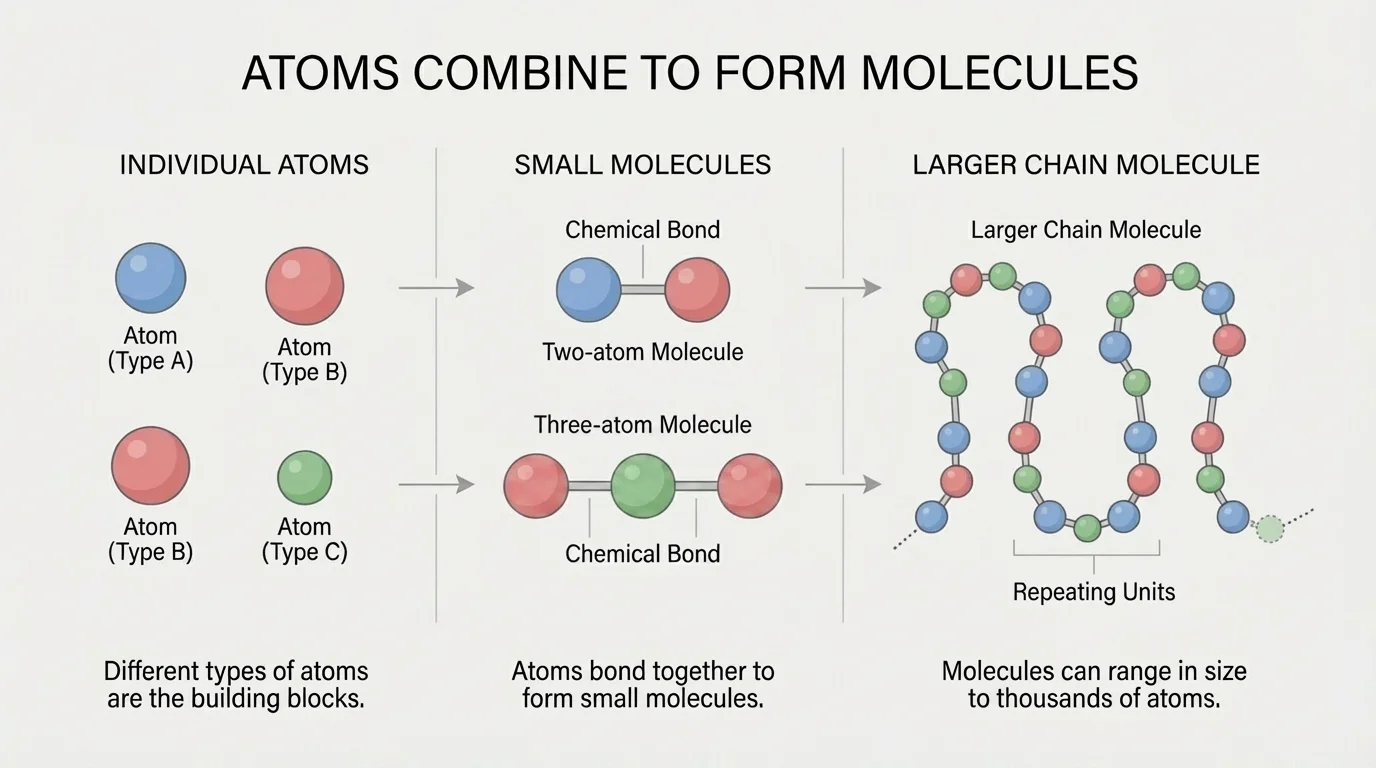
When atoms join, they form chemical bonds. You do not need to know every detail about bonding yet, but it is important to know that these bonds hold atoms together in stable groups. A molecule is not just atoms sitting nearby; the atoms are connected in an organized way.
Notice that a chemical formula tells both the types of atoms and how many of each are present. In \(\textrm{H}_2\textrm{O}\), the small \(2\) shows there are \(2\) hydrogen atoms. In \(\textrm{CO}_2\), the small \(2\) applies only to oxygen, so there is \(1\) carbon and \(2\) oxygen atoms.
Ozone and regular oxygen are both made only of oxygen atoms, but ozone is \(\textrm{O}_3\) while most oxygen gas is \(\textrm{O}_2\). Changing the number of atoms in each molecule changes the substance's properties.
The idea from [Figure 1] becomes especially important in biology and materials science. Tiny molecules such as \(\textrm{O}_2\) move easily through air, while very large molecules such as proteins fold into complex shapes that help living cells function.
Not all matter is organized in the same way. Particle models help us compare pure elements, pure compounds, and mixtures, as [Figure 2] shows. This comparison is one of the best ways to understand what a substance is made of.
An element contains only one kind of atom. It may exist as single atoms, like helium, or as molecules made of the same atom type, like \(\textrm{O}_2\) or \(\textrm{N}_2\). Even though \(\textrm{O}_2\) has two atoms in each molecule, it is still an element because both atoms are oxygen.
A compound contains atoms of different elements chemically joined in a fixed ratio. Water, \(\textrm{H}_2\textrm{O}\), is a compound. Salt, \(\textrm{NaCl}\), is a compound made from sodium and chlorine. Carbon dioxide, \(\textrm{CO}_2\), is another compound.
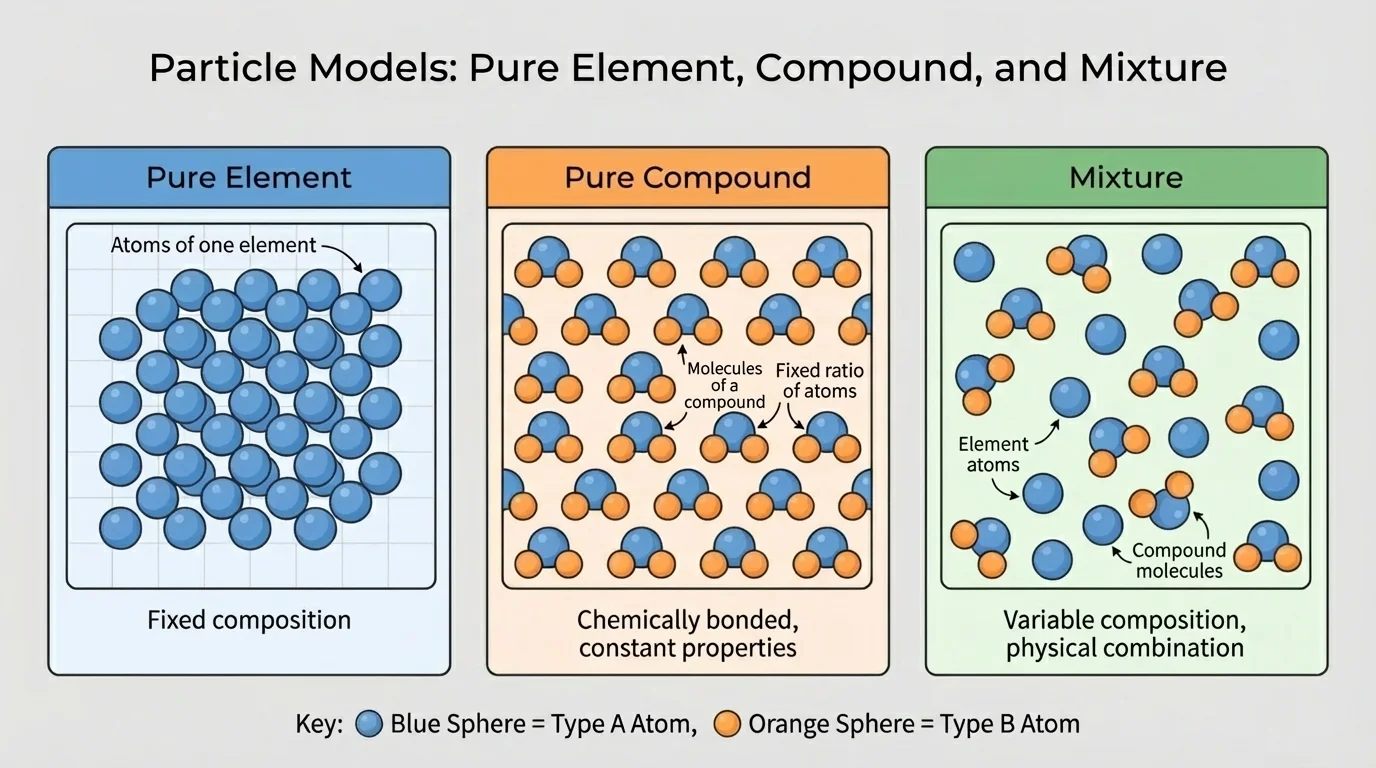
A mixture is different. In a mixture, substances are physically combined, but they are not chemically bonded into one new pure substance. Air is a mixture of gases, including nitrogen, oxygen, argon, and small amounts of carbon dioxide. Trail mix is also a mixture; the parts are together, but they still keep their own identities.
This difference matters because compounds and mixtures behave differently. A compound has properties that can be very different from the elements that form it. Water does not behave like hydrogen gas or oxygen gas. In a mixture, however, the separate substances usually keep most of their original properties.
| Type of matter | What it contains | Example |
|---|---|---|
| Element | One kind of atom | \(\textrm{Au}\), \(\textrm{He}\), \(\textrm{O}_2\) |
| Compound | Different atoms chemically joined in a fixed ratio | \(\textrm{H}_2\textrm{O}\), \(\textrm{CO}_2\), \(\textrm{NaCl}\) |
| Mixture | Two or more substances physically combined | Air, saltwater, soil |
Table 1. Comparison of elements, compounds, and mixtures.
Later, when you study methods of separating matter, this distinction becomes useful. Mixtures can often be separated by physical methods such as filtering or evaporation, while compounds must be broken apart by chemical changes.
Atoms do not combine randomly. They join in specific patterns and ratios, and those patterns strongly affect the substance that forms. The relationship between ratio and arrangement is central to chemistry, as [Figure 3] demonstrates.
Take carbon and oxygen as an example. Carbon monoxide is \(\textrm{CO}\), with a ratio of \(1:1\). Carbon dioxide is \(\textrm{CO}_2\), with a ratio of \(1:2\). Because the ratios are different, these are different substances with different properties.
Arrangement matters too. Carbon atoms can be arranged in different structures. In graphite, the carbon atoms form layers that can slide past one another, making graphite soft enough to leave marks in pencil "lead." In diamond, carbon atoms form a strong three-dimensional network, making diamond extremely hard.
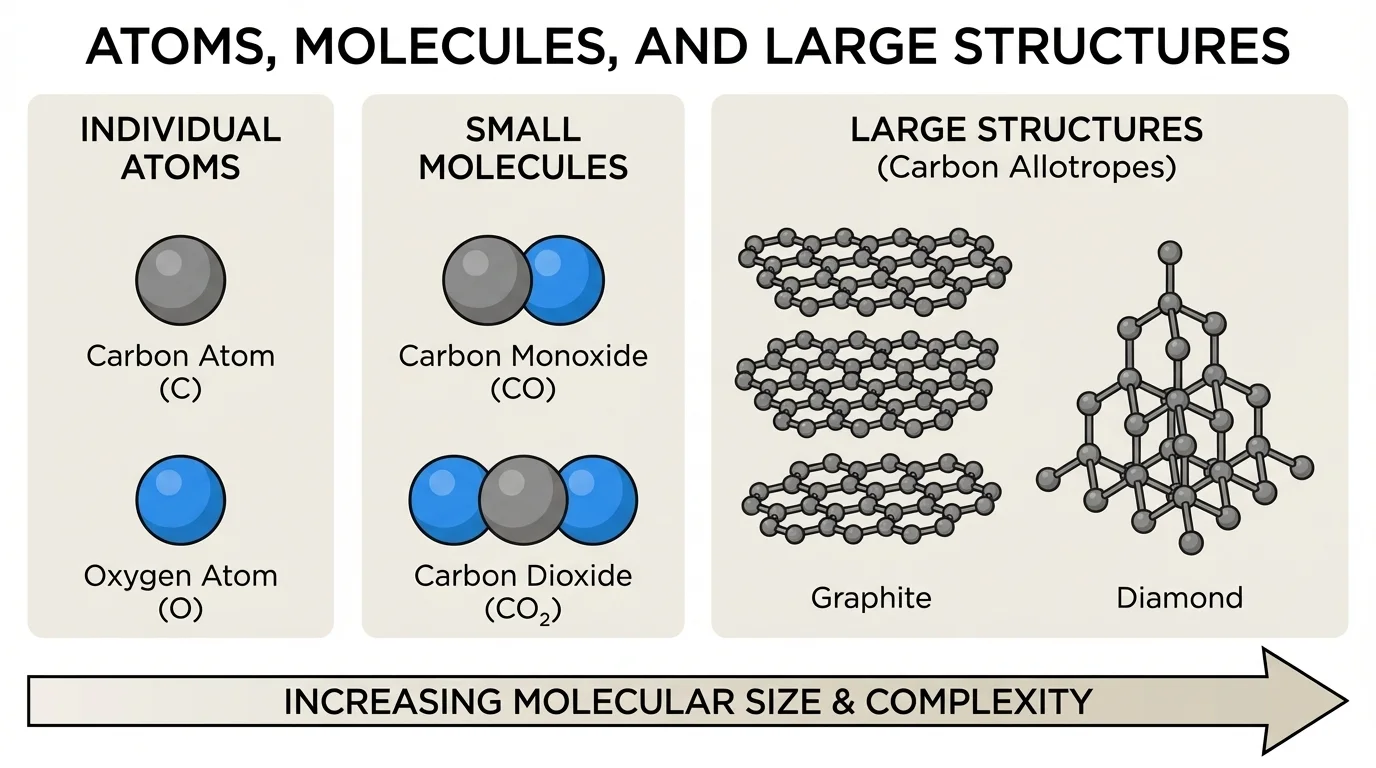
This is a remarkable idea: the same kinds of atoms can produce different substances either because the ratio changes or because the arrangement changes. Scientists must pay attention not only to which atoms are present but also to how many there are and how they are connected.
Why structure matters
The properties of a substance come from both composition and structure. Composition tells which atoms are present and in what ratio. Structure tells how those atoms are arranged. Two substances can contain the same element and still behave very differently if their atoms are connected differently.
The pattern seen in [Figure 3] also appears in biology. Large molecules in living things often contain mostly carbon, hydrogen, oxygen, and nitrogen, but small differences in arrangement can completely change what the molecule does.
When people hear the word molecule, they sometimes picture a tiny cluster of just a few atoms. That is only part of the story. Some molecules really are small, like \(\textrm{H}_2\), \(\textrm{O}_2\), or \(\textrm{H}_2\textrm{O}\). But other substances involve very large molecular chains or giant networks of atoms.
Plastics are a great example. Many plastics are made of repeating units linked into long chains called polymers. A single polymer molecule can include thousands of atoms. The length of the chain and the way chains interact help determine whether a plastic is stretchy, rigid, transparent, or tough.
Living things depend on giant molecules as well. Proteins help build muscles, speed up reactions, and send signals in cells. Starch and cellulose are large molecules built from many repeating sugar units. Even though these molecules are huge, they are still made of atoms joined in organized ways.
Real-world example: why plastics can differ so much
Step 1: Start with the idea of repeating units.
Many plastics are built by linking small molecules over and over into long chains.
Step 2: Compare chain behavior.
If the chains can slide easily, the plastic may be flexible. If the chains are strongly connected or packed tightly, the plastic may be harder and more rigid.
Step 3: Connect structure to use.
A soft plastic bag and a hard plastic chair both contain atoms arranged in large molecules, but the details of those arrangements help explain their different properties.
Not every large atomic structure is described best as a separate small molecule. Some solids, such as many crystals, are better thought of as giant arrangements of atoms repeated in a pattern. This is another reason matter can show such a wide variety of behaviors.
The properties of a substance depend on the particles that make it up and how those particles are arranged and move. This particle-level view explains why different materials act differently.
For instance, water is liquid at room temperature, while oxygen is a gas. Both are made from atoms, but their molecules are different in size, shape, and attraction. Salt forms crystals because its particles arrange in a repeating pattern. Metals conduct electricity well because of how their atoms are organized and how electrons can move through them.
A material's hardness, melting point, solubility, and conductivity are not random traits. They come from its atomic composition and structure. That means if scientists understand the particles, they can design new materials for specific jobs, such as stronger phone screens, lighter bicycles, or medicines that target certain molecules in the body.
Graphite in pencils and diamond in jewelry are both made entirely of carbon atoms. Their different properties come from different arrangements of the same type of atom.
This is one of the biggest ideas in science: what you observe on a large scale is connected to what is happening on a tiny scale. The visible world depends on invisible structure.
When matter changes, the atoms do not vanish. In a chemical change, atoms are rearranged into new combinations, as [Figure 4] shows. This is why new substances can form while the total atoms of each element are conserved.
For example, hydrogen and oxygen can react to form water: \[2\textrm{H}_2 + \textrm{O}_2 \rightarrow 2\textrm{H}_2\textrm{O}\] On the left side, there are \(4\) hydrogen atoms and \(2\) oxygen atoms. On the right side, there are also \(4\) hydrogen atoms and \(2\) oxygen atoms. The atoms are the same before and after; they are just connected differently.
This idea helps explain burning, rusting, cooking, digestion, and many other everyday processes. A chemical reaction does not create matter from nothing or destroy it. Instead, it changes how atoms are arranged.
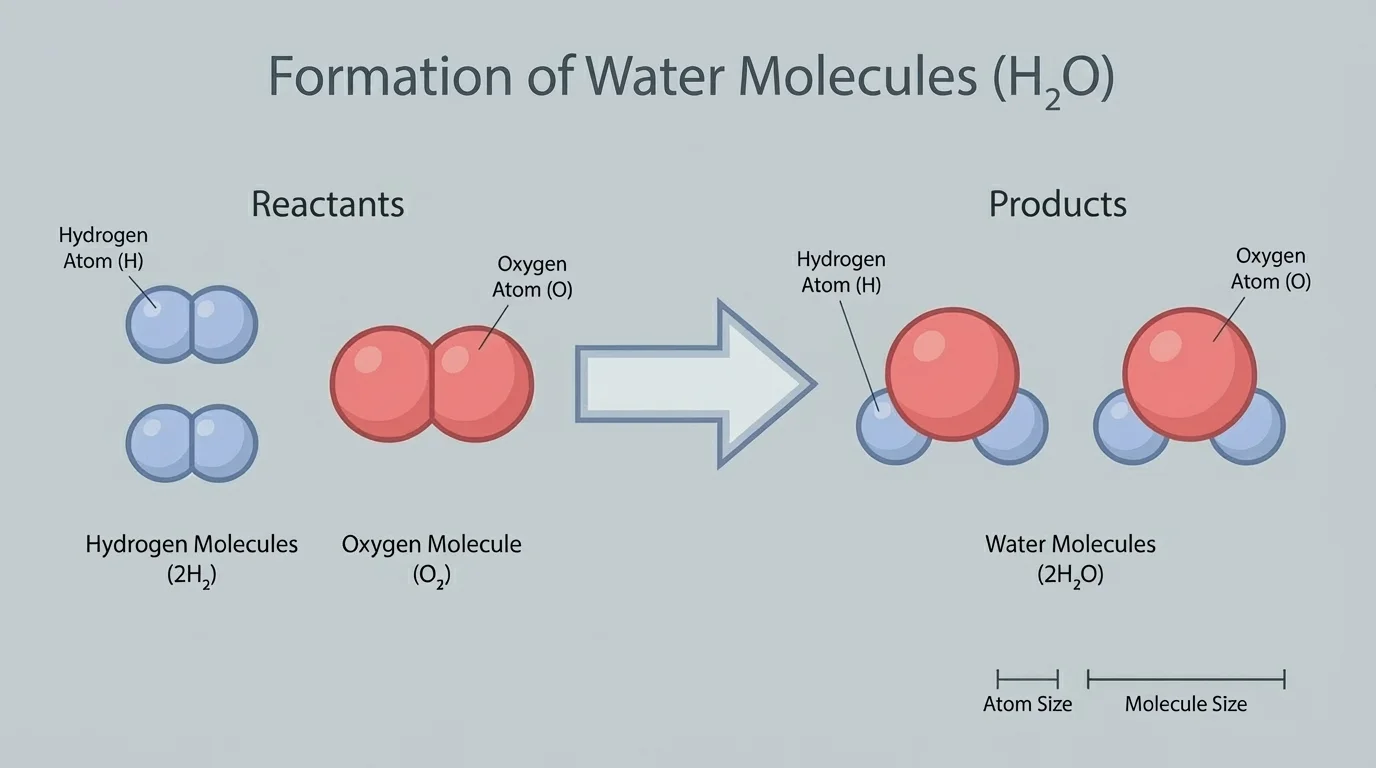
A physical change is different. If ice melts into liquid water, the substance is still \(\textrm{H}_2\textrm{O}\). The molecules do not become a different substance; they simply move differently and spread out more. In a chemical change, the atoms are regrouped to make something new.
The reaction model in [Figure 4] is useful because it shows both change and conservation at the same time. Molecules can break apart and form new ones, but the atoms themselves remain part of the system.
Real-world example: cooking an egg
Step 1: An uncooked egg contains large protein molecules folded into particular shapes.
Step 2: Heating changes those arrangements and interactions.
Step 3: The egg white turns from clear to opaque because the molecules are reorganized, changing the material's properties.
This helps explain why many chemical and physical changes are really changes in particle arrangement.
Understanding rearrangement also helps scientists write chemical equations. The equation must show the same number of each kind of atom on both sides, because atoms are not lost; they are redistributed.
The idea that substances are made from atoms and molecules is not just classroom science. It helps people solve real problems. Doctors design medicines by studying how specific molecules fit with parts of cells. Engineers create stronger and lighter materials by changing atomic structure. Environmental scientists monitor gases such as \(\textrm{CO}_2\) and methane because the kinds and amounts of molecules in the atmosphere affect climate.
Cooking is full of molecular changes. Bread rises because gases form and get trapped in dough. Sugar dissolves in water because water molecules pull sugar particles apart and spread them out. Soap cleans because its molecules have one end attracted to water and another attracted to oils and grease.
Your own body is also a molecular system. Breathing brings in \(\textrm{O}_2\). Digestion breaks large food molecules into smaller ones. Cells then use atoms from food and oxygen to build new molecules and release energy for life processes.
"The world is made of stories, but also of atoms."
— Adapted from a famous scientific idea
Modern technology depends on this understanding too. Batteries, fertilizers, water treatment, medicines, synthetic fabrics, and computer chips all involve careful control of atoms and molecules. Once scientists understand how matter is built, they can redesign materials to do useful things.
One misunderstanding is that a molecule must contain different kinds of atoms. That is not true. \(\textrm{O}_2\) is a molecule made of only oxygen atoms. Another misunderstanding is that if something is too small to see, it must not have structure. In fact, atoms and molecules are highly organized.
Another mistake is thinking that a compound is just any combination of substances. A compound must have atoms chemically bonded in a fixed ratio. Saltwater is not a compound; it is a mixture of water and dissolved salt.
Some students also think chemical reactions destroy atoms. They do not. Reactions rearrange atoms into new substances. If you keep track of the atoms carefully, the count of each element stays the same before and after the reaction.
Finally, bigger is not always better for understanding matter. A large visible object can still be made of repeating microscopic patterns. Whether a substance is a tiny drop or a huge rock, the same core idea applies: matter is built from atoms.