Your body is giving off heat right now, even if you are sitting completely still. At the same time, your cells are building molecules, pumping ions across membranes, sending nerve signals, and keeping you alive. None of that happens because energy appears out of nowhere. It happens because energy is constantly being transferred and transformed. One of the most powerful ideas in science is that energy cannot be created or destroyed. It only moves: from food into cells, from cells into motion, from motion into heat, and from organisms into their environment.
When people say they are "out of energy," they do not mean energy has vanished from the universe. They mean their bodies have less readily available energy for the tasks they want to do. The total amount of energy is conserved, but its location and form change. This is true in physics, chemistry, and biology. A phone battery, a campfire, a solar panel, and a human muscle all follow the same rule.
In living things, this idea is especially important because life depends on controlled energy transfers. Organisms do not create energy. They obtain it from external sources, such as sunlight or food, and then transfer it through chemical reactions. In animals, much of that transfer happens through cellular respiration, the set of chemical reactions that breaks down food molecules and transfers some of their energy into a form cells can use.
"Energy cannot be created or destroyed, only transformed and transferred."
— First law of thermodynamics, stated in everyday language
This principle also helps explain why every process has consequences. If energy moves into one part of a system, it must come from somewhere else. If a cell gains chemical energy in ATP, that energy came from molecules such as glucose. If your muscles warm up during a workout, some transferred energy has become thermal energy.
Scientists often understand energy by defining a system, a part of the universe chosen for study. The system could be a cell, a whole organism, or even a mitochondrion. Everything outside it is the surroundings. Energy can cross the boundary between them, as [Figure 1] shows in the case of a human body during activity.
Energy may move between objects by direct contact, such as heat transfer from a warm hand to a cold metal railing. It may also move through fields. For example, sunlight reaches Earth through electromagnetic radiation, so energy moves from the Sun to living systems without needing matter to travel all the way from the Sun to your skin or to a leaf.
In biology, energy transfer often involves both matter and fields. You eat food, so matter enters your body. But the energy associated with chemical bonds in that food is then transferred through chemical reactions. Some becomes motion, some helps build or transport molecules, and some leaves as heat. This is why a runner needs food and oxygen, exhales carbon dioxide and water vapor, and releases heat to the air.
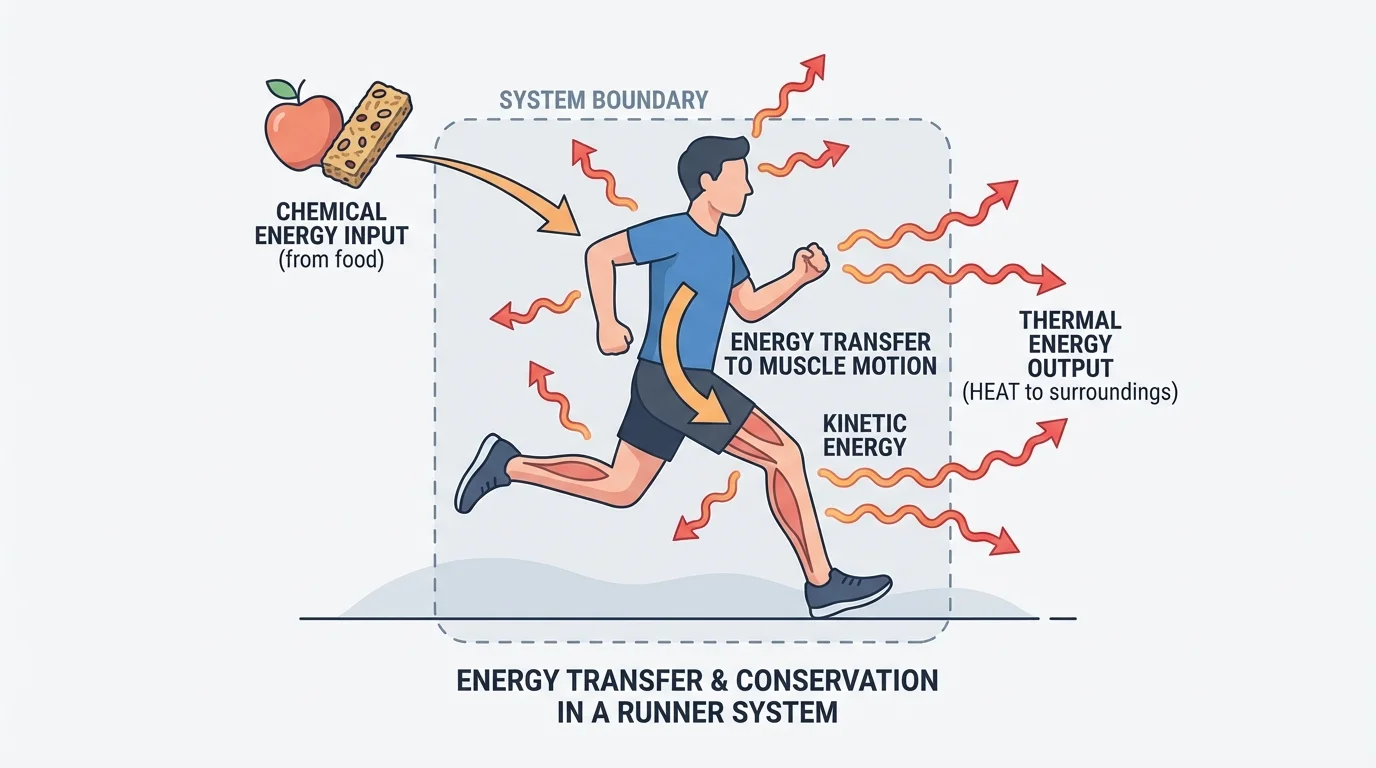
Thinking in terms of systems prevents a common mistake: saying energy is "lost." In careful scientific language, energy is not lost from the universe. It may become less useful for a particular purpose, especially when much of it spreads out as thermal energy, but it still exists.
Energy conservation means the total amount of energy remains constant in an isolated system. Energy transfer is movement of energy from one object, place, or system to another. Energy transformation is a change from one form of energy to another, such as chemical energy becoming kinetic energy or thermal energy.
These ideas become clearer when we zoom in from the whole body to the level of cells and molecules.
Food contains molecules whose atoms are arranged in ways that make energy transfer possible during reactions. A major example is glucose, \(\textrm{C}_6\textrm{H}_{12}\textrm{O}_6\). Glucose contains many bonds such as \(\textrm{C–H}\), \(\textrm{C–C}\), and \(\textrm{O–H}\). Cells can react glucose with oxygen, \(\textrm{O}_2\), to form carbon dioxide, \(\textrm{CO}_2\), and water, \(\textrm{H}_2\textrm{O}\).
It is tempting to think of chemical bonds as little containers full of energy. That picture is only partly helpful. A better idea is this: different arrangements of atoms have different potential energies. During a chemical reaction, old bonds are broken and new bonds are formed. Because the products have lower chemical potential energy than the reactants in cellular respiration, there is a net transfer of energy to the cell and surroundings.
Not all of the transferred energy becomes useful cellular work. Some of it becomes thermal energy. That is why metabolism helps maintain body temperature. If you have ever felt warm after eating or exercising, you have experienced this energy transfer directly.
Atoms are conserved in chemical reactions: they are rearranged, not created or destroyed. Energy is also conserved, but unlike matter, it can be transferred in many forms such as chemical, thermal, electrical, and kinetic.
Plants also make glucose, but mainly through photosynthesis, which captures energy from sunlight. Animals then obtain chemical energy by eating plants or other organisms. So the energy in your muscles during a sprint may have started as energy radiated from the Sun.
Cellular respiration is best understood through a model, and [Figure 2] traces the major inputs and outputs. In eukaryotic cells, most cellular respiration happens in mitochondria, organelles specialized for transferring energy from food molecules into ATP.
The overall reaction for cellular respiration is often written as:
\[\textrm{C}_6\textrm{H}_{12}\textrm{O}_6 + 6\textrm{O}_2 \rightarrow 6\textrm{CO}_2 + 6\textrm{H}_2\textrm{O} + \textrm{energy}\]
This equation shows conservation of matter clearly: the same kinds and numbers of atoms appear on both sides, rearranged into new compounds. It also shows that energy is not created from nothing. The reactants already have chemical energy. During the reaction, some of that energy is transferred into ATP and some becomes thermal energy.
The molecule ATP, short for adenosine triphosphate, acts as an immediate energy-transfer molecule in cells. Cells do not use the energy in a sandwich directly to contract a muscle fiber or move sodium ions across a membrane. Instead, energy from food is transferred through respiration into ATP, and ATP then participates in reactions that power cellular work.
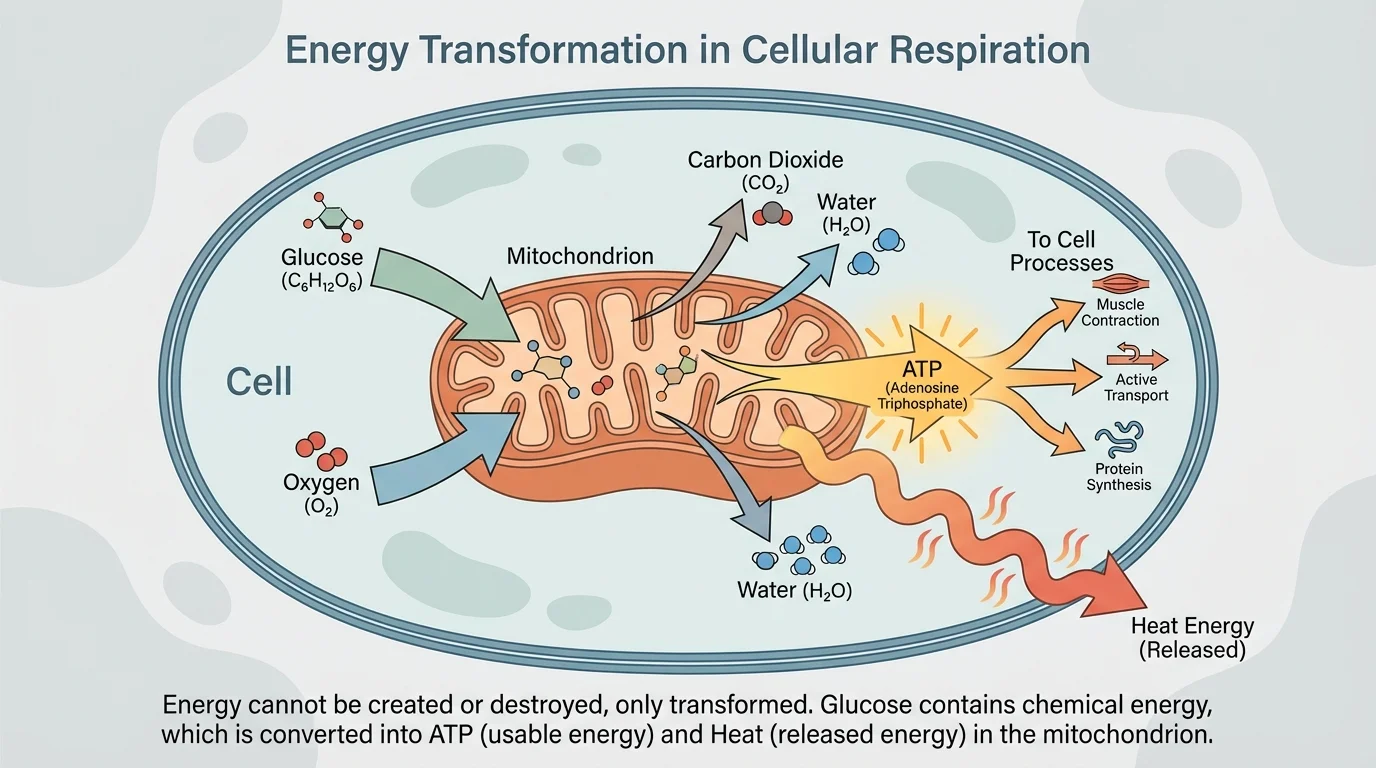
Cellular respiration includes several stages, including glycolysis, the citric acid cycle, and oxidative phosphorylation. For this topic, the key idea is not memorizing every intermediate compound but recognizing the pattern: bonds in food molecules are broken, bonds in new compounds are formed, and there is a net transfer of energy.
Why ATP matters
ATP is useful because it couples energy-releasing processes to energy-requiring ones. When ATP is converted to ADP and inorganic phosphate, energy can be transferred to drive muscle contraction, active transport across membranes, synthesis of proteins, and many other cellular processes. ATP is not an energy source separate from food; it is an energy-transfer molecule produced from the energy originally stored in food or captured from sunlight.
A healthy human body continuously turns over large amounts of ATP each day. Yet the mass of ATP in the body at any one moment is relatively small because ATP is constantly regenerated. That makes ATP more like a rechargeable energy-transfer currency than a long-term storage form.
One of the most important chemistry ideas in biology is that breaking chemical bonds requires energy, while forming chemical bonds releases energy. Students often hear that "energy is in the bonds" and conclude that simply breaking a bond releases energy. That is incorrect.
During cellular respiration, energy must be supplied to break bonds in glucose and oxygen. However, when new bonds form in carbon dioxide and water, even more energy is released. Because the energy released in forming the product bonds is greater than the energy required to break the reactant bonds, there is a net energy transfer out of the reacting molecules and into ATP production and thermal energy.
This is a net change idea. Suppose, in a simplified example, breaking old bonds requires \(2{,}800 \textrm{ kJ}\) per mole of glucose, while forming new bonds releases \(3{,}000 \textrm{ kJ}\). The net transferred energy would be \(3{,}000 - 2{,}800 = 200 \textrm{ kJ}\). The reaction does not create \(200 \textrm{ kJ}\); it transfers that amount because the products are at a lower chemical potential energy than the reactants.
Numeric example: net energy transfer in a reaction
A simplified reaction requires \(950 \textrm{ kJ}\) to break reactant bonds and releases \(1{,}120 \textrm{ kJ}\) when product bonds form. Find the net energy transferred out of the reacting molecules.
Step 1: Identify the energy in and energy out.
Energy required to break bonds: \(950 \textrm{ kJ}\)
Energy released when bonds form: \(1{,}120 \textrm{ kJ}\)
Step 2: Subtract the required energy from the released energy.
Net transferred energy is \(1{,}120 - 950 = 170 \textrm{ kJ}\).
The reaction transfers a net of \(170 \textrm{ kJ}\) to the surroundings and any energy-capturing molecules in the system.
This same logic helps explain why some reactions are good energy sources for organisms and others are not. The usefulness of a food molecule depends on how its atoms can be rearranged and what the energy difference is between reactants and products.
Tracing energy through a model is one of the best ways to avoid vague statements. The pathway in [Figure 3] follows energy from food into ATP, then into specific kinds of cellular work, and finally into thermal energy that leaves the body.
A cell takes in glucose and oxygen. Through cellular respiration, it transfers some of the glucose molecule's chemical energy into ATP. Then ATP transfers energy to processes such as active transport, biosynthesis, and movement. For example, sodium-potassium pumps in nerve cells use ATP to maintain ion gradients, and muscle cells use ATP to help actin and myosin slide past each other during contraction.
At each stage, some energy becomes thermal energy. This does not violate conservation of energy. It is exactly what conservation predicts: if energy leaves one form or location, it appears in another. The body does not perfectly channel every transfer into useful biological work.
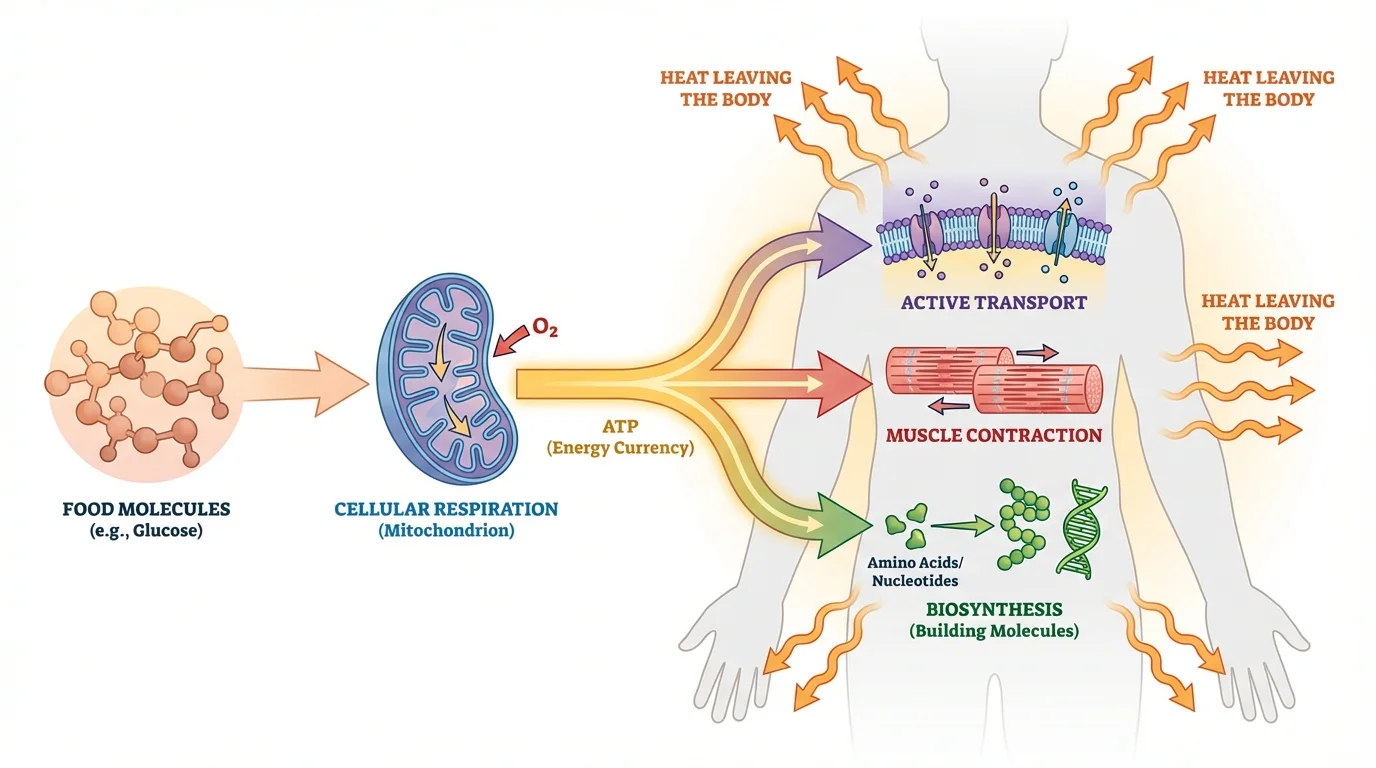
We can also describe this model as a chain of systems. Food is one energy-containing system. A cell is another. ATP molecules participate within the cell, and the surrounding environment is another system that receives thermal energy. Later, when thinking about exercise or ecosystems, we can return to the same model and simply change the scale of the system.
Your brain uses a large fraction of the body's energy at rest, even though it makes up only a small fraction of body mass. Much of that energy supports ion pumping needed for nerve signaling.
Models do not include every detail, but they help us answer a key question: where did the energy come from, and where did it go? In science, that question is often more important than memorizing the names of every molecule involved.
ATP transfer can be discussed qualitatively, but a simple numerical example makes the idea sharper. A commonly used approximate value is that hydrolysis of one mole of ATP to ADP and phosphate transfers about \(30.5 \textrm{ kJ/mol}\) under standard conditions:
\[\textrm{ATP} + \textrm{H}_2\textrm{O} \rightarrow \textrm{ADP} + \textrm{P}_i + 30.5 \, \textrm{kJ/mol}\]
If a cellular process uses \(2\) moles of ATP, the approximate energy transferred is \(2 \times 30.5 = 61.0 \textrm{ kJ}\). Again, this energy is not being created by ATP. ATP is participating in a transfer process.
Numeric example: energy transferred by ATP use
A cell process uses \(4\) moles of ATP. Estimate the energy transferred if each mole transfers about \(30.5 \textrm{ kJ}\).
Step 1: Write the multiplication.
\(\textrm{Energy transferred} = 4 \times 30.5 \textrm{ kJ}\)
Step 2: Calculate.
\[4 \times 30.5 = 122.0 \, \textrm{kJ}\]
The cell process transfers approximately \(122.0 \textrm{ kJ}\).
Another useful idea is efficiency. If a process transfers \(100 \textrm{ kJ}\) from food molecules and \(38 \textrm{ kJ}\) ends up in ATP while the rest becomes heat, the efficiency is:
\[\textrm{efficiency} = \frac{38}{100} \times 100\% = 38\%\]
This does not mean \(62\%\) of the energy disappeared. It means \(62\%\) was transferred into other forms, mostly thermal energy, instead of being captured in ATP.
Numeric example: efficiency of energy capture
A reaction transfers \(250 \textrm{ kJ}\) from food molecules. If \(95 \textrm{ kJ}\) is captured in ATP, what percent is captured?
Step 1: Use the efficiency formula.
Efficiency \(= \dfrac{95}{250} \times 100\%\)
Step 2: Compute the fraction.
\[\frac{95}{250} = 0.38\]
Step 3: Convert to a percent.
\[0.38 \times 100\% = 38\%\]
The process captures \(38\%\) of the transferred energy in ATP.
These simple calculations support the central idea: energy accounting matters in biology just as much as in chemistry or physics.
In sports science, understanding energy transfer helps explain why sprinting and marathon running feel so different. Sprinting depends heavily on rapid ATP supply over short time periods, while endurance events depend on sustained cellular respiration and oxygen delivery. The same conservation principle applies in both cases: food energy is transferred into motion, heat, and biochemical work.
In medicine, problems with mitochondria can reduce a cell's ability to transfer energy efficiently. This can affect tissues with high energy demands, such as muscles and the nervous system. Doctors and researchers study mitochondrial disorders partly by tracing how energy transfer through cellular respiration is disrupted.
In ecology, the energy stored in food webs also follows conservation. Plants capture a tiny fraction of incoming solar energy and store some of it in chemical bonds. Herbivores and carnivores then obtain energy by eating other organisms. At every transfer, some energy becomes thermal energy and disperses to the surroundings, which is one reason food chains usually have limited length.
Even technology borrows these ideas. Battery chemistry, fuel cells, and bioengineering all depend on understanding how reactions transfer energy and how much of that transfer can be captured for useful work.
One misconception is that organisms "make" energy. Organisms do not make energy; they transfer and transform it. Plants capture light energy and store some of it chemically. Animals transfer chemical energy from food into ATP and other forms.
A second misconception is that energy is destroyed when it becomes heat. Thermal energy is still energy. It may simply be less concentrated and less useful for a particular task, such as muscle contraction.
A third misconception is that cellular respiration is only about breathing. Breathing supplies oxygen and removes carbon dioxide, but cellular respiration is the chemical process in cells that transfers energy from food molecules. Breathing supports respiration, but it is not the same thing.
Finally, some students think ATP contains energy as a magical substance. A more accurate statement is that ATP participates in reactions that transfer energy. Biology becomes much clearer when you focus on energy movement, not on "energy stuff" being stored and used up like a liquid.
By returning to the system view from [Figure 1] and the cellular model in [Figure 2], we can connect the scale of the whole organism to the scale of molecules. The same conservation law applies at every level.