You can wave your hand through air and feel almost nothing, yet air can fill a balloon, push a kite into the sky, and knock leaves across the ground. That seems strange at first. If you cannot see air, how can it be real? Science gives a powerful answer: air is matter, and matter is made of tiny pieces that can be far too small to see.
Matter is anything that has mass and takes up space. A rock is matter. Water is matter. Air is matter too. Sometimes students think only solid things count as matter because solids are easy to see and touch. But gases are matter even though they are harder to notice.
Scientists have learned that all matter can be divided into tiny pieces called particles. These particles are so small that we cannot see them with our eyes. Even when matter is broken into pieces too tiny to see, the matter still exists. It can still be detected in other ways.
Matter is anything that has mass and takes up space.
Gas is a form of matter whose particles are far apart and move freely.
Model is a science idea or representation that helps explain how something works.
Think about perfume, soup, or popcorn. You may smell it from across a room before you see it. That smell reaches your nose because tiny particles from the substance spread through the air. The particles are too small to see, but your nose detects them. This is evidence that invisible matter is still present.
Here is another clue. When you blow into a plastic bag, the bag puffs up. Something goes in, even though you cannot see it. That "something" is air. It takes up space inside the bag. If it takes up space, it is matter.
Breathing is also evidence. Your lungs fill with air, and then air comes back out. If you breathe onto a cold window, it fogs up. That tells you the breath contains matter, including tiny bits of water in the gas you exhale.
Your body uses gases all day long. You breathe in oxygen and breathe out carbon dioxide, written as \(CO_2\). Both are forms of matter, even though you usually cannot see them.
Scientists use a model to explain gases, and [Figure 1] shows the main idea clearly: gases are made of tiny particles that are spread out, with lots of empty space between them, and they move freely in all directions. We cannot watch these particles directly with our eyes, but this model helps explain what we observe.
Unlike particles in many solids, gas particles are not locked into one place. They zoom around and bump into each other and into the sides of any container holding them. A container can be a bottle, a room, a tire, or a balloon.
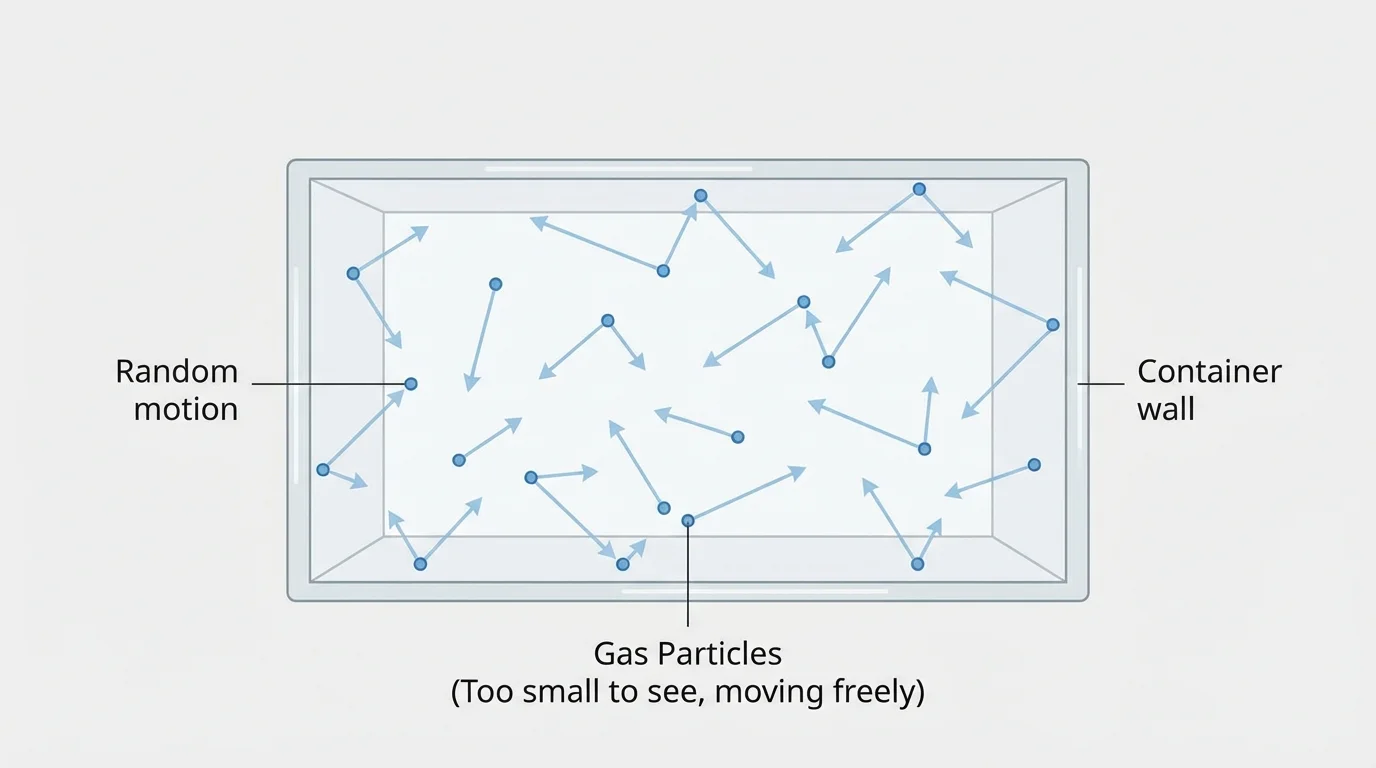
Because gas particles move freely, a gas does not stay in a little pile. It spreads out to fill the space available. If air is inside a bedroom, it spreads through the whole room. If air is inside a soccer ball, it spreads through the inside of the ball.
This particle model explains why gases can be squeezed more easily than solids or liquids. Since there is a lot of empty space between gas particles, the particles can be pushed closer together. If you press on a syringe filled with air while the tip is closed, the air inside can be compressed.
The model also explains why a smell travels. Tiny gas particles from food or flowers move through the air until some of them reach your nose. What seems like "nothing" in the air is actually many moving particles. The spreading of a smell makes more sense when we remember the moving particles we saw earlier in [Figure 1].
Why a model matters
A scientific model is not just a guess. It is a careful way of thinking that helps explain evidence. We use the gas particle model because it matches many observations: gases spread out, gases can be squeezed, smells travel through air, and air fills containers.
A balloon looks simple, but it is a great science tool. When you blow into a balloon, your breath adds more gas particles inside it. As [Figure 2] illustrates, these particles move freely and bump into the inside surface of the balloon again and again.
Each tiny collision is very small, but there are enormous numbers of particles. Together, these collisions push outward on the balloon's rubber. This push is called pressure. As more particles enter the balloon, there are more collisions, so the push increases.
The balloon stretches because the rubber can bend. It grows until the inward pull of the rubber balances the outward push from the gas particles. That is why the balloon inflates instead of staying flat.
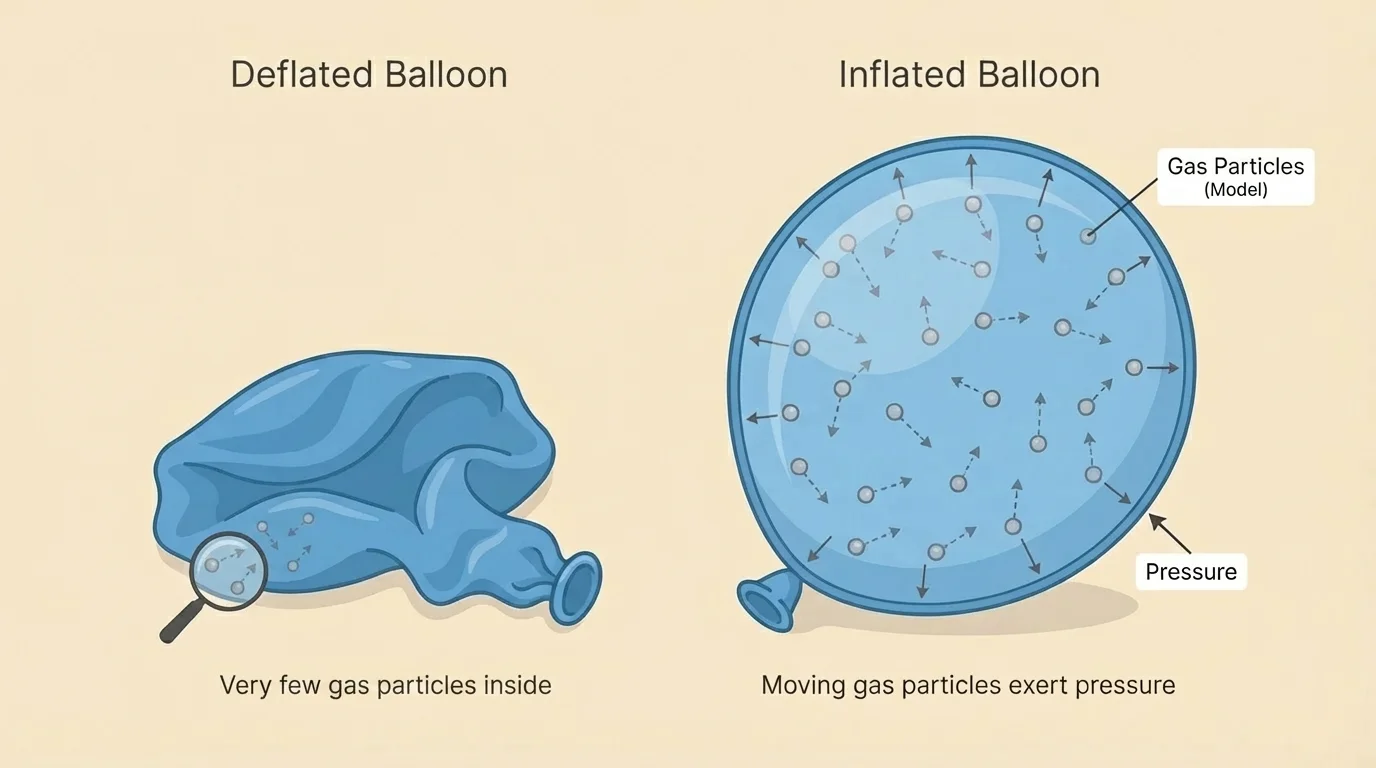
The shape of the balloon also makes sense with the gas particle model. Gas particles spread out to fill available space. If the balloon material can stretch evenly, the balloon becomes rounded because the gas pushes in many directions at once.
If you squeeze one side of a balloon, the gas particles still move everywhere inside, but the shape changes because the rubber changes shape. When you let go, the balloon often returns to a rounded form. The particles keep moving and spreading through the space inside.
This same idea works for basketballs, bicycle tires, and inflatable pool toys. Air inside pushes outward on the walls. The object's material holds the air in and gives the object its shape. The collisions shown earlier in [Figure 2] help explain why a ball feels firmer when it has more air inside.
Example: Why does a balloon get bigger when you blow more air into it?
Step 1: More air goes into the balloon.
That means more gas particles are added inside.
Step 2: The particles keep moving and colliding.
They bump into the inside of the balloon more often.
Step 3: The increased collisions create a stronger outward push.
This pressure stretches the rubber outward.
The balloon becomes larger because many tiny moving particles push on its inside surface.
Air may be invisible, but it can affect things we can see, and [Figure 3] shows several good examples. Moving air, which we call wind, can push on dust, leaves, flags, sails, and kites. That happens because air is matter made of moving particles.
When many air particles move in the same general direction, they collide with objects in their path. Each collision is tiny, but together they can create a strong push. That is why wind can swing branches or slam a door.
You can observe this with very small particles too. Dust can drift in the air. Pollen can blow from one place to another. Sand can move along a beach on a windy day. Air particles interact with these larger particles and carry them along.
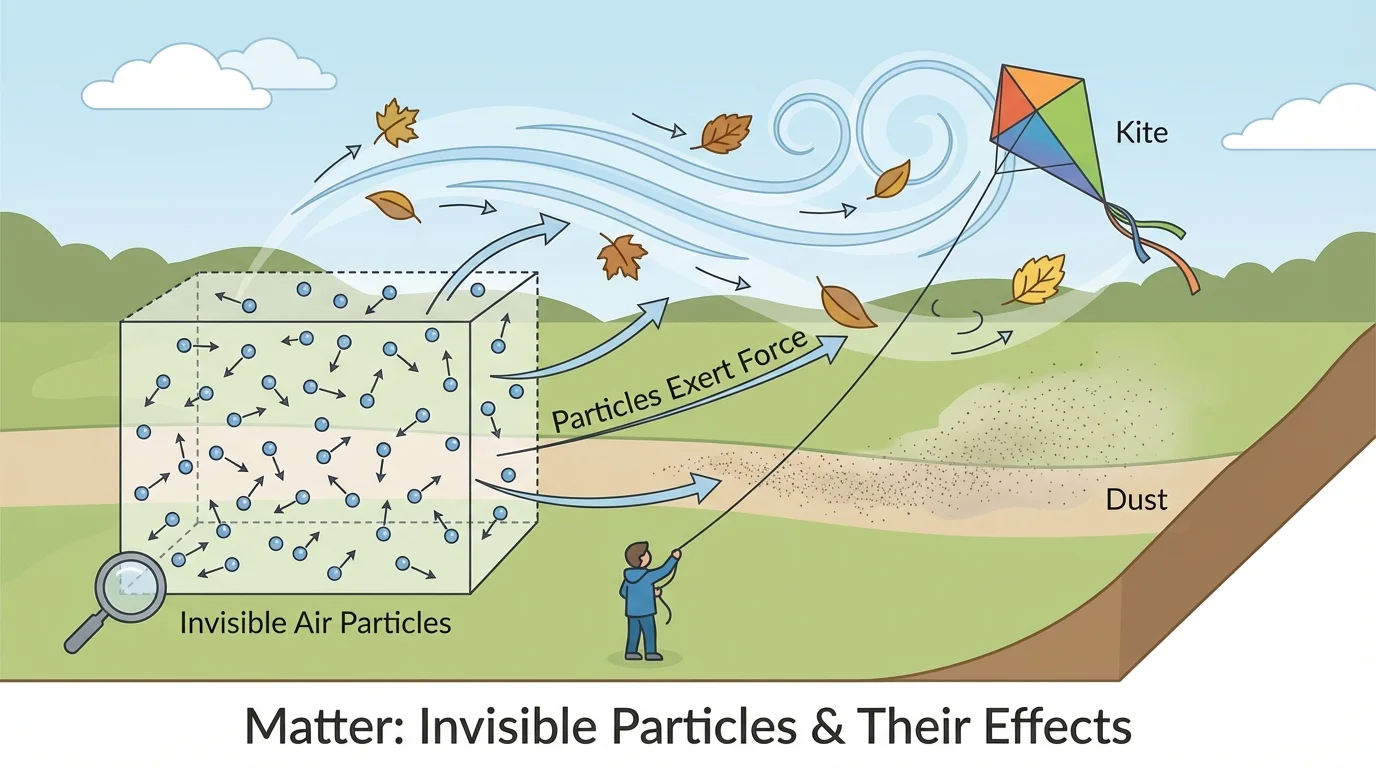
Kites are a great example of air pushing on a larger object. As wind moves against the kite, the air pushes on the kite's surface. If the push is strong enough and the kite is angled correctly, the kite rises.
Parachutes work in a related way. The wide parachute catches air, and the air pushes upward against it. This slows the person or object falling downward. We cannot see the air itself, but we can see what it does.
The same thinking helps explain why a fan cools you. The fan moves air toward your skin. That moving air carries heat away more quickly and can also help sweat evaporate faster. Invisible particles are causing a very noticeable effect.
When you watch leaves blowing or a kite lifting, the gas particle model from [Figure 1] is still useful. Air seems empty, but it is full of moving particles that can push on larger objects.
You already know that a push or a pull is a force. Air can create forces too. Wind is not magic—it is moving matter pushing on objects.
Science often depends on evidence we can observe. We may not see the particles themselves, but we can detect their effects. This is how scientists learn about things too small to see directly.
One kind of evidence is using touch to detect air. Put your hand near a fan. You feel moving air. Put your hand over the end of an inflated balloon as air rushes out. You feel the fast-moving gas.
Another kind of evidence is hearing. If you let air out of a balloon, you hear a sound. The escaping gas causes the balloon to vibrate and the surrounding air to vibrate. Your ears detect those vibrations.
A syringe with its tip covered provides strong evidence too. If you push the plunger, the trapped air resists being squeezed smaller. If there were truly nothing inside, there would be no resistance. The trapped gas particles press back.
Example: A simple observation with a syringe
Step 1: Pull air into a syringe and cover the tip.
Now gas particles are trapped inside.
Step 2: Push the plunger gently.
The air gets squeezed into a smaller space.
Step 3: Notice the resistance.
The trapped gas pushes back because its particles are still moving and colliding inside the syringe.
This shows that even invisible matter still exists and takes up space.
Smell is another way to detect matter. When someone peels an orange on one side of a room, the scent slowly spreads. Tiny particles from the orange move through the air until they reach people farther away. Their noses detect matter too small to see.
If you blow soap bubbles, you can detect gas by sight in a different way. The bubble film outlines the space taken up by the trapped gas. The gas itself is hard to see, but the bubble's shape reveals where the gas is.
Matter can be a solid, a liquid, or a gas. In a solid, particles are packed closely and stay in fixed positions more often. In a liquid, particles are still close together but can slide past one another. In a gas, particles are much farther apart and move freely.
That means the same basic idea—matter is made of particles—works for all three forms. The difference is how the particles are arranged and how they move. Gases are special because they spread out much more than solids and liquids.
| Form of matter | Particle spacing | Particle motion | Shape behavior |
|---|---|---|---|
| Solid | Very close together | Vibrate in place | Keeps its own shape |
| Liquid | Close together | Slide past each other | Takes shape of container |
| Gas | Far apart | Move freely in all directions | Fills available space |
Table 1. Comparison of particle spacing, motion, and shape behavior in solids, liquids, and gases.
Water gives an easy example. Ice, liquid water, and water vapor are all the same substance, \(H_2O\), but the particles behave differently in each form. When water becomes water vapor, it is still matter. It has not disappeared. Its particles are simply much farther apart and moving freely as a gas.
Invisible does not mean nonexistent
Many gases are colorless, but that does not mean they are nothing. We know they exist because they can be trapped, moved, compressed, smelled, measured, and used. Science often studies things we cannot see directly by looking at the evidence they leave behind.
The particle model of gases helps explain many ordinary things. A car tire works because air inside pushes outward on the tire. A soccer ball bounces because the air inside compresses and pushes back. Breathing works because gases move into and out of your lungs.
Weather also depends on moving air. Wind forms when air moves from one place to another. Storms involve huge amounts of moving gas in Earth's atmosphere. The same basic idea applies whether you are talking about a tiny balloon or a giant weather system: gas particles are moving and interacting.
Helium balloons are another interesting example. Helium is a gas, written as \(He\). Like other gases, it is made of tiny moving particles. A helium balloon has gas particles inside that push on the balloon, giving it shape. Because helium is lighter than the surrounding air, the balloon can rise.
Air is not just one gas. It is a mixture, mostly nitrogen and oxygen, along with smaller amounts of other gases. Even though the mixture is invisible, it still has mass and takes up space.
Engineers and doctors also depend on gas behavior. Air pumps fill tires and sports equipment. Scuba tanks store compressed gas for divers. Oxygen tanks help patients breathe. These technologies work because gases are matter made of tiny moving particles.
Even safety devices use this science. Air bags in cars fill quickly with gas to protect people during a crash. The gas spreads out and pushes against the bag material, just as air pushes on a balloon. The explanation for both depends on the moving particles and pressure we discussed earlier in [Figure 2].