A cup of hot coffee cools on a desk, a car engine heats up while running, and an ice pack warms in your hand. These seem like ordinary events, but each one raises the same scientific question: what exactly counts as the system, and what lies outside it? The answer matters. If you do not define the system correctly, your explanation of what enters, what leaves, and why change happens can become confusing or completely wrong. In science, one of the first steps in investigating any process is deciding where the system begins and ends.
Scientists use systems thinking because real situations are often too complex to study all at once. A city, a cell, a weather pattern, or a beaker of water can all be treated as systems. Once a system is defined, scientists can identify its components, its interactions, and the transfers of energy or matter that affect it. This approach is especially important in chemistry and physics, where understanding energy transfer often depends on carefully defined boundaries.
When studying thermal energy, the system must be clearly described. For example, if a piece of hot metal is placed into cooler water, is the system just the metal? Just the water? Both together? What about the cup holding them? Each choice changes the analysis. A model only works well when the boundary of the system and the starting conditions are stated clearly.
A system is a set of interacting parts considered as a whole. Everything outside that chosen region is called the surroundings. This may sound simple, but it is one of the most powerful ideas in science. If you are studying a chemical reaction in a flask, you might define the reacting chemicals as the system and the flask plus room air as the surroundings. If you are studying a moving bicycle, the bicycle and rider might be the system, while the road and air are the surroundings.
System means the part of the universe chosen for study. Boundary is the real or imagined surface that separates the system from its surroundings. Initial conditions are the measured starting properties of the system, such as temperature, mass, pressure, speed, or composition.
Defining the system helps scientists ask better questions. What can cross the boundary? Is energy entering as heat, light, or electrical work? Is matter entering or leaving? Are there outside influences that should be controlled? Without these decisions, measurements can be misleading. For example, if a warm object cools down, that does not necessarily mean energy "disappeared." More likely, thermal energy crossed the boundary into the surroundings.
In many investigations, system definitions are chosen to simplify analysis. Good science is not about including every detail; it is about including the right details. A model becomes useful when it focuses on the most important interactions and ignores those too small to matter for the question being studied.
Not all boundaries behave the same way. As [Figure 1] illustrates, scientists often classify systems by whether matter or energy can cross the boundary. This classification helps predict what kinds of changes are possible and which conservation ideas can be applied most directly.
An open system allows both matter and energy to cross the boundary. A boiling uncovered pot is a good example. Water vapor leaves the pot, so matter crosses the boundary, and thermal energy enters from the stove. Open systems are common in daily life because most real objects interact continuously with their surroundings.
A closed system allows energy to cross the boundary, but not matter. A sealed bottle warming in sunlight is approximately a closed system. Light energy enters, thermal energy may later leave, but the mass inside remains the same. In many chemistry investigations, scientists try to create a closed system so they can focus on energy redistribution without losing material.
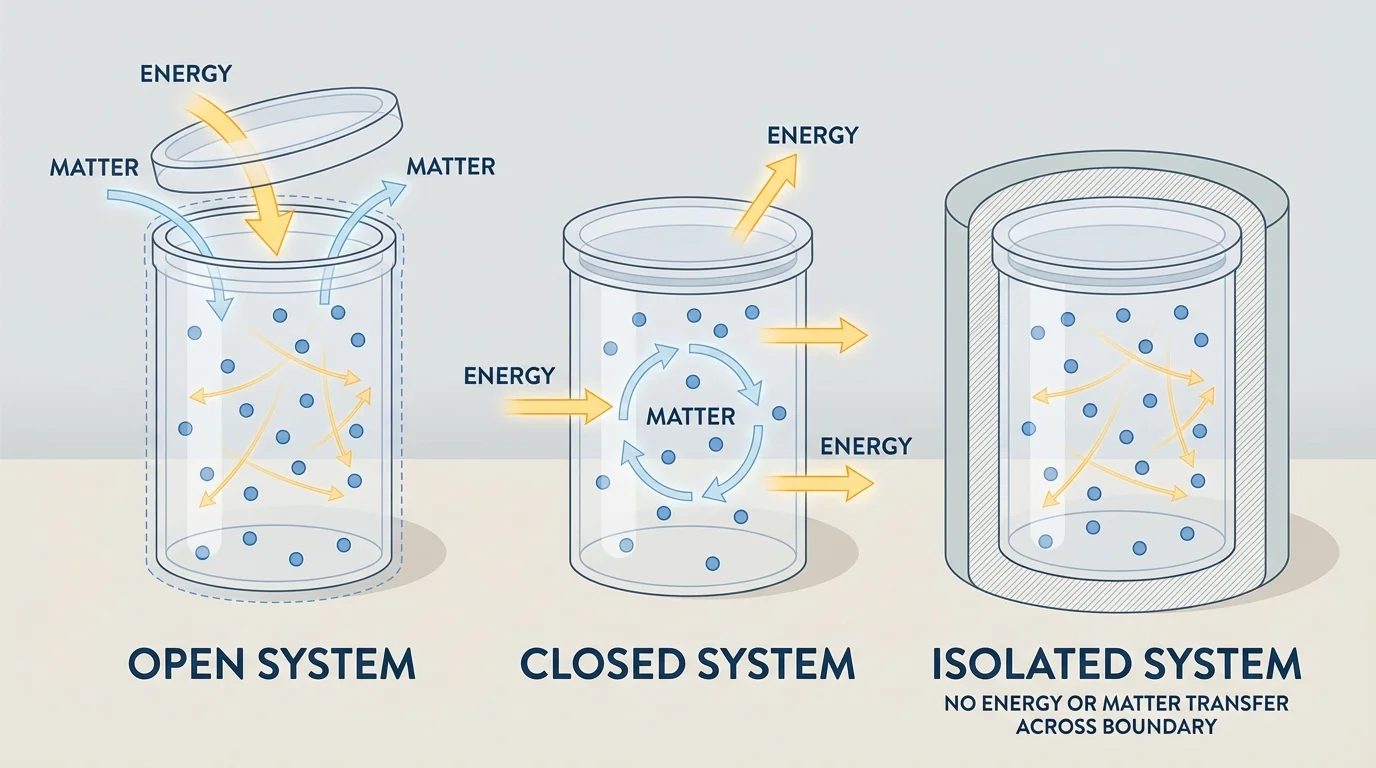
An isolated system allows neither matter nor energy to cross the boundary. Perfectly isolated systems are idealizations; in practice, they are extremely difficult to create. Even a well-insulated thermos still exchanges a small amount of energy with the surroundings over time. However, scientists often treat a system as isolated approximately if the transfers are small enough to ignore.
The type of boundary chosen affects the explanation. If you observe a temperature change in a closed system, you usually interpret it as energy being redistributed among parts of the system or crossing the boundary as energy only. In an open system, both material flow and energy flow may need to be tracked. That is why a well-defined boundary is the foundation of a good model.
Once the boundary is defined, the next step is to record the system's starting state. These starting values are called the initial conditions. In a thermal energy investigation, important initial conditions often include temperature, mass, phase, and the identity of the substances involved. A sample of water at \(20\,\textrm{°C}\) behaves differently from the same mass of water at \(80\,\textrm{°C}\), and liquid water behaves differently from ice because phase matters.
Initial conditions are essential because they influence the direction and amount of change. If two objects begin at different temperatures, thermal energy tends to transfer from the hotter object to the cooler one. If they begin at the same temperature, there is no net thermal energy transfer between them. This idea sounds obvious, but it is a powerful predictive tool.
Temperature is a measure related to the average kinetic energy of particles in a substance. Thermal energy refers to the total internal energy associated with the random motion and interactions of particles in a sample.
In many experiments, initial conditions must be measured carefully because small errors at the start can affect the final result. For example, if a metal sample is recorded as \(75\,\textrm{°C}\) when it is actually \(72\,\textrm{°C}\), the predicted final temperature after mixing with cooler water will be inaccurate. Good investigations depend not only on the right model but also on accurate initial data.
Initial conditions can include more than numerical values. They can also include whether the system is at rest or moving, whether a reaction has started, or whether the container is insulated. In other words, the "starting state" includes both measurable properties and relevant setup conditions.
After defining the system and measuring initial conditions, scientists identify inputs and outputs. An input is anything entering the system, such as energy from a battery or matter flowing through a pipe. An output is anything leaving the system, such as waste heat, emitted light, or released gas. Inputs and outputs are not guesses; they must be analyzed based on the chosen boundary.
Suppose you are studying a rechargeable hand warmer. If the system is the chemical pouch only, thermal energy leaving the pouch is an output, while any mass leaving due to a leak would also be an output. If the system includes the pouch and your hand together, then the thermal transfer between them happens within the system instead of crossing the boundary. The science changes because the boundary changed.
Models often represent inputs and outputs with arrows. These arrows are not just visual decoration. They show the direction of transfer and help clarify cause and effect. In system models, arrows can represent forces, energy transfer, matter flow, or information flow, depending on the situation being studied.
Why models matter
A model is a simplified representation of a real system. It may be a diagram, a verbal description, a graph, a physical replica, or a mathematical equation. Models help scientists explain patterns, make predictions, and test whether evidence supports an idea. A good model clearly states what is included, what is excluded, and what assumptions are being made.
In secondary school and beyond, system models become especially important because many scientific phenomena involve interactions among multiple parts. A thermal energy investigation is a good example: the water, the metal, the container, and the air all matter, but they may not all be included in the same system model.
When two components at different temperatures are combined in a closed system, thermal energy tends to spread out more evenly among them, as [Figure 2] shows. This process leads toward a more uniform energy distribution. That trend is consistent with the second law of thermodynamics: energy spontaneously becomes more dispersed unless outside work maintains a difference.
Consider hot metal placed into cooler water inside an insulated cup with a lid. If we define the system as the metal plus the water, and if we assume the cup is good enough that very little energy escapes, then the combined system is approximately closed to matter and nearly isolated for energy transfer during the short experiment. The hot metal cools, the cooler water warms, and both approach the same final temperature.
This final state is called thermal equilibrium. Thermal equilibrium does not mean particles stop moving. It means the system has reached the same temperature throughout the interacting components, so there is no longer a net flow of thermal energy between them. At that point, the energy distribution is more uniform than it was initially.
This process does not require "cold" to move into the hot object. Instead, energy transfers from higher-temperature parts of the system to lower-temperature parts. The direction matters: under ordinary conditions, net thermal energy transfer goes from hot to cold, not the reverse.
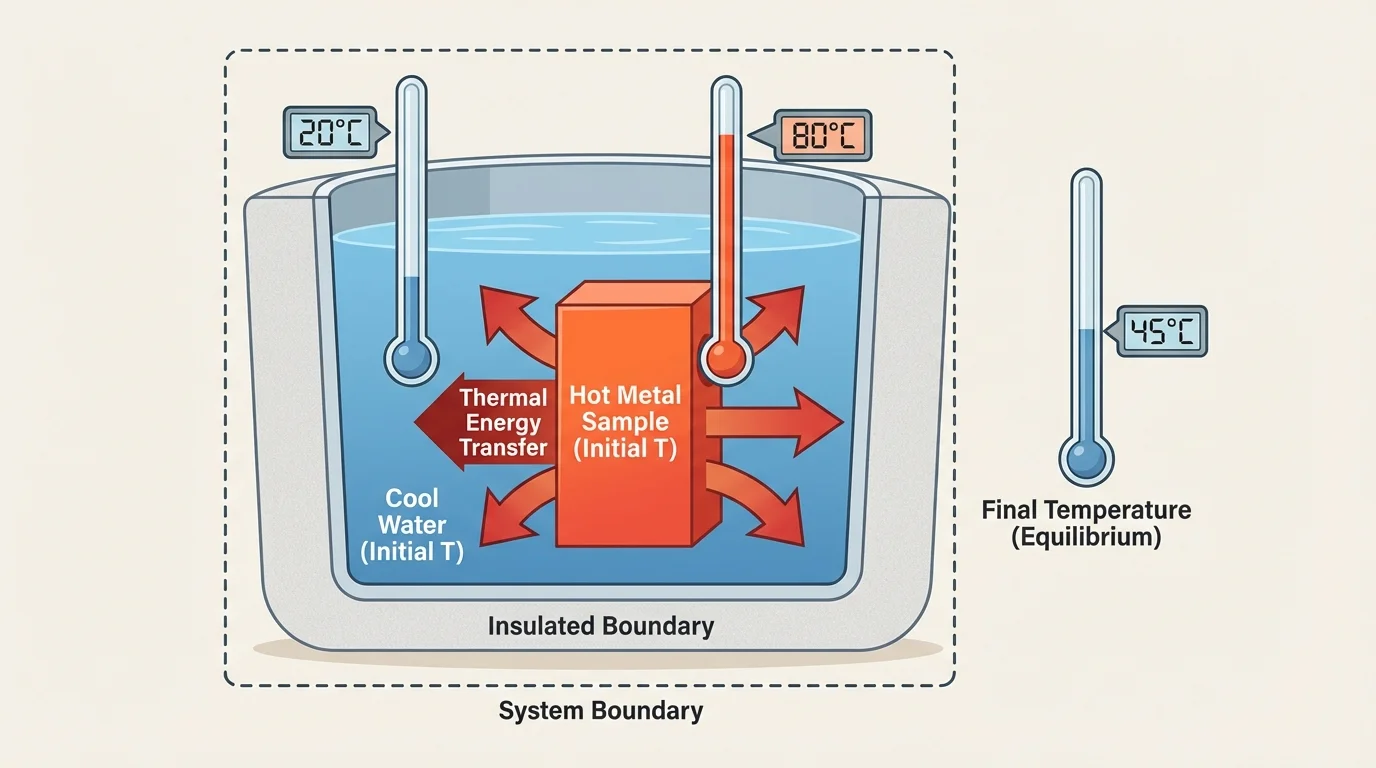
A common misunderstanding is that the final temperature should be halfway between the two initial temperatures. That is not always true. The actual final temperature depends on the masses of the materials and their specific heat capacities. Water usually changes temperature less than many metals for the same amount of transferred energy because water has a relatively high specific heat capacity.
This idea helps explain why oceans moderate coastal temperatures, why metal benches feel colder than wooden ones on the same day, and why burns can happen quickly when touching hot metal. The same systems reasoning used in a classroom calorimetry setup helps explain real thermal behavior outside the lab. This kind of energy redistribution is a small-scale version of what happens in countless natural and engineered systems.
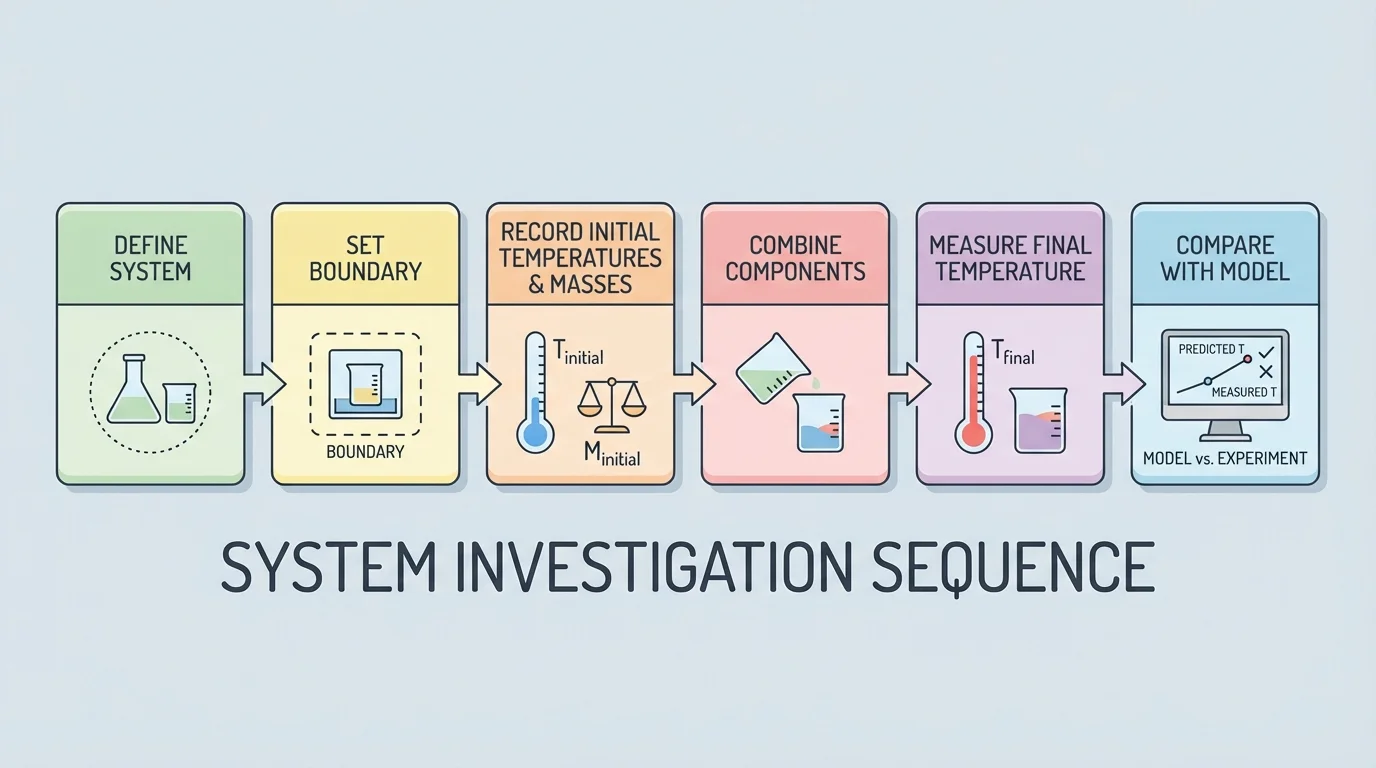
A strong investigation follows a systems-based sequence, as [Figure 3] illustrates: define the system, state the boundary, record initial conditions, identify expected inputs and outputs, collect data, and compare the data to the model. Without that order, students may gather numbers without understanding what those numbers mean.
Suppose you want to test the claim that combining two components of different temperatures in a closed system leads to a more uniform energy distribution. One practical setup is to place a known mass of warm water and a known mass of cooler water in an insulated container, then measure the final temperature after mixing. Another setup uses hot metal and cool water. In either case, the investigation should include careful mass measurements, accurate thermometers, and a clear statement of assumptions.
The independent variable could be the initial temperature difference or the mass of one component. The dependent variable could be the final equilibrium temperature. Controlled variables might include the container type, insulation level, timing of measurements, and stirring method. These controls matter because they reduce unwanted influences from the surroundings.
Investigation example: mixing warm and cool water
A student mixes \(100\,\textrm{g}\) of water at \(60\,\textrm{°C}\) with \(100\,\textrm{g}\) of water at \(20\,\textrm{°C}\) in a well-insulated cup.
Step 1: Define the system
The system is both water samples together inside the cup. The cup is treated as part of the surroundings or ignored if its effect is very small.
Step 2: State the initial conditions
Masses are equal, and initial temperatures are \(60\,\textrm{°C}\) and \(20\,\textrm{°C}\).
Step 3: Predict the result
Thermal energy transfers from the warmer water to the cooler water until both reach the same final temperature. Because the masses and substances are the same, the final temperature is expected to be close to \(40\,\textrm{°C}\).
Step 4: Interpret the evidence
If the final measured temperature is near \(40\,\textrm{°C}\), the data support the model of energy redistribution toward uniform temperature.
Evidence from this kind of investigation supports a broader principle: temperature differences within a system tend to decrease unless external energy is added or removed in a way that maintains the difference. That is why insulated systems approach equilibrium, while refrigerators and air conditioners must continuously do work to keep regions at different temperatures.
System models can be qualitative, but they can also be mathematical. One important equation for thermal energy transfer is
\[Q = mc\Delta T\]
where \(Q\) is the thermal energy transferred, \(m\) is mass, \(c\) is specific heat capacity, and \(\Delta T\) is the change in temperature.
In a nearly isolated mixing experiment, the energy lost by the hotter component is approximately equal to the energy gained by the cooler component. That idea can be written as
\[Q_{\textrm{lost}} = Q_{\textrm{gained}}\]
For example, suppose \(200\,\textrm{g}\) of water cools from \(50\,\textrm{°C}\) to \(35\,\textrm{°C}\). Using water's specific heat capacity of about \(4.18\,\textrm{J/(g}\cdot\textrm{ °C)}\), the thermal energy lost is
\[Q = mc\Delta T = (200)(4.18)(15) = 12{,}540\,\textrm{J}\]
The magnitude of the lost energy is \(12{,}540\,\textrm{J}\). If the system is well insulated, another component in the system gains about that same amount of energy.
Worked example: hot metal in cool water
A \(100\,\textrm{g}\) metal sample at \(80\,\textrm{°C}\) is placed into \(200\,\textrm{g}\) of water at \(20\,\textrm{°C}\). The final temperature is \(24\,\textrm{°C}\). Find the energy gained by the water.
Step 1: Identify the known values
For the water, \(m = 200\,\textrm{g}\), \(c = 4.18\,\textrm{J/(g}\cdot\textrm{ °C)}\), and \(\Delta T = 24 - 20 = 4\,\textrm{°C}\).
Step 2: Substitute into the equation
Using \(Q = mc\Delta T\), we get \(Q = (200)(4.18)(4)\).
Step 3: Calculate
\[Q = 3{,}344\,\textrm{J}\]
The water gains \(3{,}344\,\textrm{J}\) of thermal energy, so the metal loses approximately that amount if heat loss to the surroundings is negligible.
Quantitative models help test whether observations match theory. If the measured final temperature differs significantly from the predicted value, scientists do not immediately reject the theory. They first ask whether the system boundary was chosen correctly, whether the cup absorbed some energy, or whether measurement error occurred.
Every model leaves something out. A calorimetry model may ignore heat absorbed by the container, heat exchange with the air, evaporation, or nonuniform temperature immediately after mixing. These simplifications are not flaws by themselves; they are choices made to focus on the main pattern.
However, it is important to know when a model becomes too simple. If a container is poorly insulated, the "closed system" assumption becomes weak because energy is escaping rapidly. If a lid is not used and steam escapes, matter also crosses the boundary, making the system more like an open system. The labels open, closed, and isolated are useful, but they often describe approximations rather than perfection.
Space is often described as cold, but a spacecraft's thermal system is complicated because heat transfer by conduction and convection is limited in vacuum. Engineers must carefully control radiation and internal energy flow to keep equipment working.
Measurement uncertainty also matters. A thermometer with a precision of only \(\pm 1\,\textrm{°C}\) may hide small differences between prediction and observation. Good scientists report limitations and discuss whether the evidence still supports the model within reasonable uncertainty.
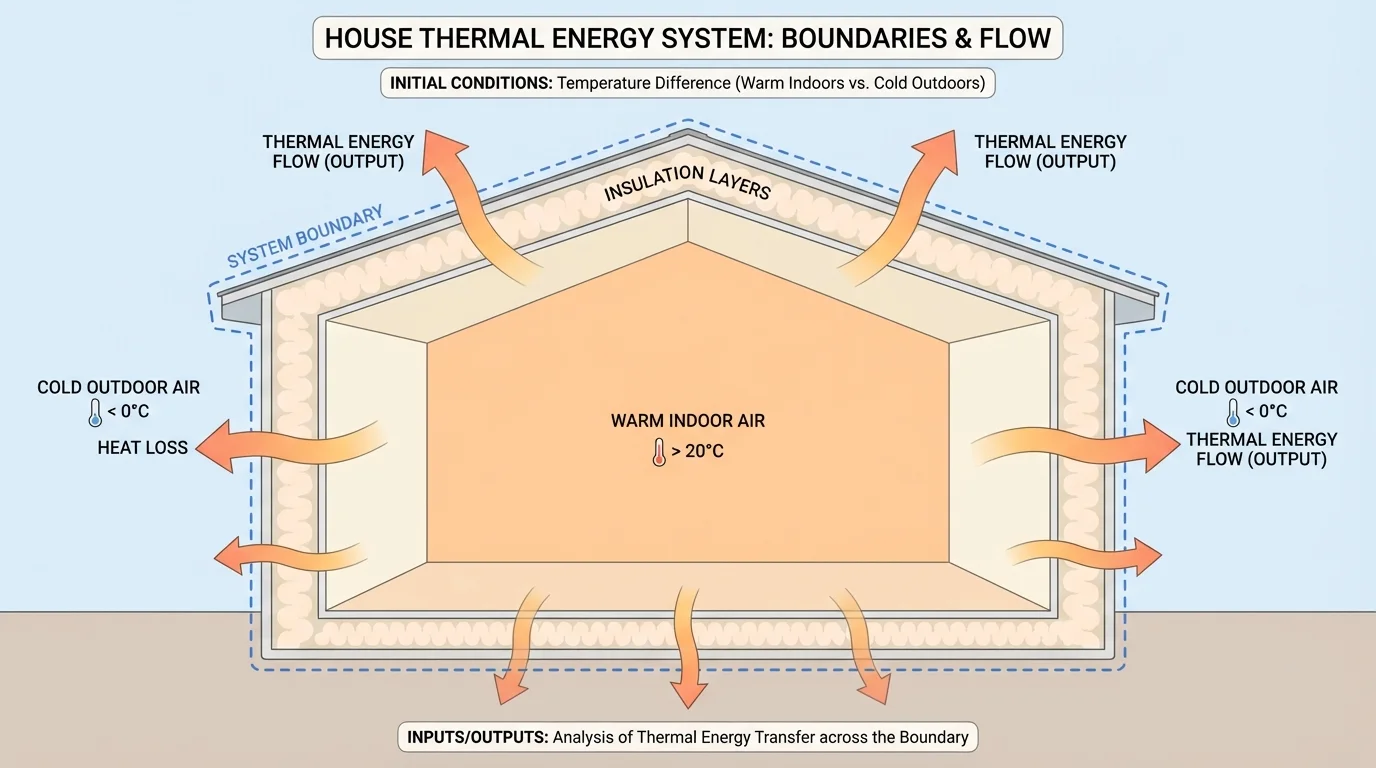
System boundaries are not just classroom ideas. In engineering design, medicine, and environmental science, the choice of boundary can determine whether a model is useful. In a house, for example, the boundary may be the insulated walls, roof, floor, windows, and doors. Thermal energy enters and leaves through those boundaries, and engineers analyze those flows to improve efficiency, as [Figure 4] shows.
If the indoor air is warmer than the outdoor air, thermal energy tends to flow outward. Better insulation reduces that transfer. In this case, the house is not an isolated system because energy crosses its boundary. But by changing materials and structure, engineers can reduce outputs and maintain a desired internal condition with less energy input.
In medicine, body temperature regulation can also be analyzed as a system. The human body exchanges energy with the environment through radiation, conduction, convection, and evaporation. When doctors use cooling blankets or warmed intravenous fluids, they are intentionally changing inputs and outputs to move the body toward a safer internal state.
In climate science, Earth can be treated as a system receiving energy from the Sun and releasing energy to space. The atmosphere, oceans, land, and ice interact as subsystems. The same logic applies: define the system, state the boundaries, identify inputs and outputs, and use models to understand change over time.
Even chemical reactions can be analyzed this way. An exothermic reaction in a sealed container transfers energy to its surroundings while keeping matter inside the container. An endothermic process absorbs energy from the surroundings. To explain either one correctly, the boundary and the initial conditions must be known.
Across all of these examples, the core scientific habit stays the same: define what is being studied, describe its starting state, track what crosses the boundary, and use a model to explain the evidence. That habit turns messy reality into a system that can be investigated clearly and rigorously.