A hospital uses X-rays to see broken bones, a kitchen uses microwaves to heat food, sunscreen blocks ultraviolet light, and a router sends information through radio waves. These technologies seem completely different, yet they all depend on one powerful idea: if you understand what happens at a very small scale, you can often predict what will happen in a much larger system. That is one of the most important habits in science and engineering.
In both natural systems and human-designed systems, cause and effect relationships are often too complex to observe all at once. Instead, scientists study smaller mechanisms inside the system. When matter absorbs electromagnetic radiation, the effects depend on details such as the radiation's frequency, the energy carried by its photons, and the structure of the material doing the absorbing. By connecting these smaller mechanisms, we can predict larger outcomes such as warming, glowing, chemical reactions, cell damage, data transfer, or medical imaging.
A system is a set of connected parts that influence one another. In a climate system, radiation from the Sun interacts with gases, land, water, and living things. In a communication system, electromagnetic waves move through air or optical fiber and are detected by receivers. In a human body, radiation can pass through tissue, be absorbed, and trigger biological responses. These are complex systems because many variables interact at once.
To make sense of complexity, scientists break the system into smaller pieces. They ask questions such as: Which particles absorb the energy? What kind of motion or change does that energy produce? How does that local change spread through the rest of the system? This approach turns a confusing big picture into a chain of understandable causes and effects.
Small-scale mechanism to large-scale effect
A small-scale mechanism is a process happening at the level of atoms, molecules, electrons, or tiny device components. A large-scale effect is what we can observe at the human scale, such as a material heating up, skin reddening, a sensor producing an image, or a signal carrying information. Scientific prediction becomes stronger when the observed effect matches a known mechanism.
This way of thinking also protects us from weak or misleading claims. If someone says a certain kind of radiation has a dramatic effect, the first scientific question is not just whether the claim sounds impressive. The first question is whether there is a believable mechanism connecting the radiation to the effect.
[Figure 1] Electromagnetic radiation is energy that travels as oscillating electric and magnetic fields. The entire spectrum forms one connected system, and changing the frequency changes wavelength and energy in predictable ways. Radio waves, microwaves, infrared, visible light, ultraviolet, X-rays, and gamma rays are all part of the same spectrum.
Two key properties are frequency and wavelength. Frequency is how many wave cycles pass a point each second. Wavelength is the distance from one crest to the next. For electromagnetic waves in a vacuum, they are related by the equation \[c = f\lambda\] where \(c\) is the speed of light, \(f\) is frequency, and \(\lambda\) is wavelength. If frequency increases, wavelength decreases.
Another crucial relationship connects frequency to photon energy: \(E = hf\), where \(E\) is photon energy, \(h\) is Planck's constant, and \(f\) is frequency. This means higher-frequency radiation carries more energy per photon. For example, if one photon has frequency \(5.0 \times 10^{14} \textrm{ Hz}\), then its energy is approximately \(E = (6.63 \times 10^{-34})(5.0 \times 10^{14}) \approx 3.3 \times 10^{-19} \textrm{ J}\). If the frequency doubles, the photon energy doubles too.
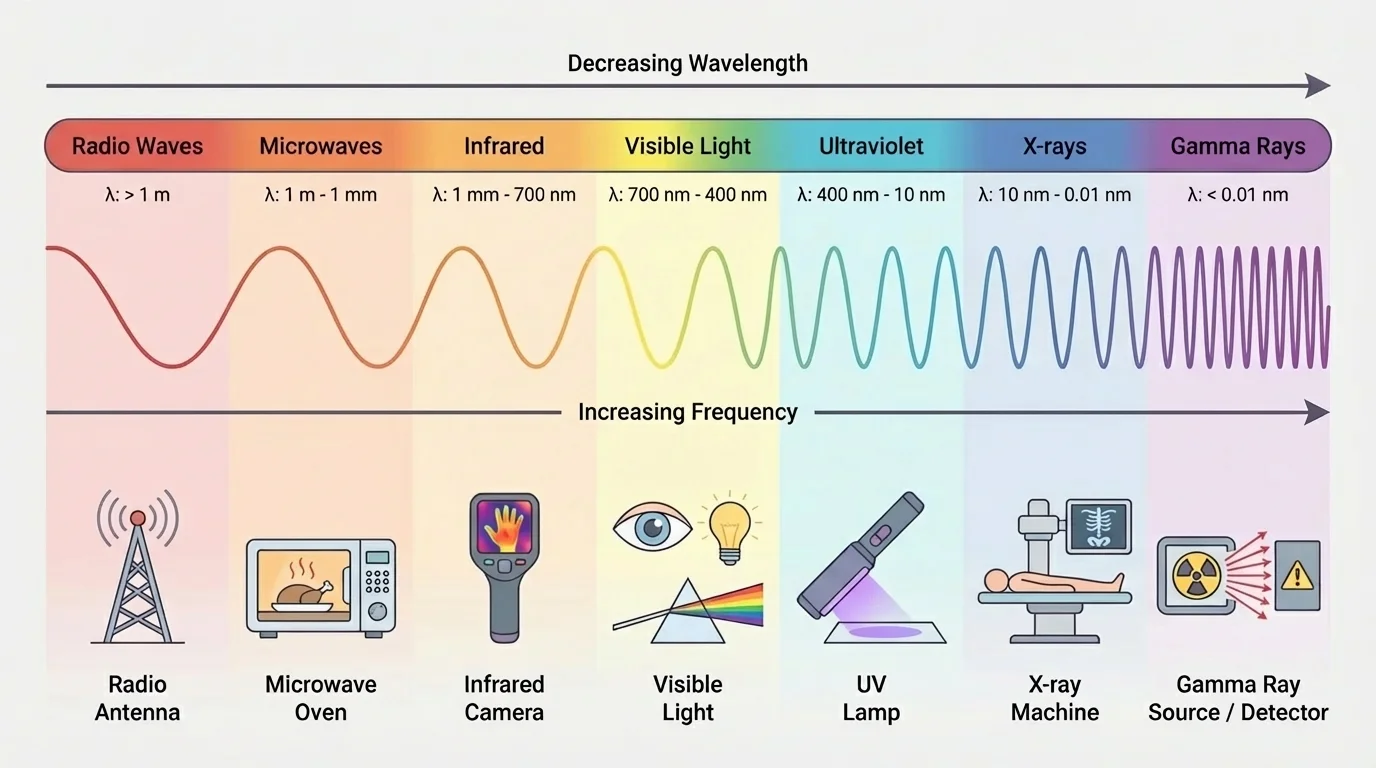
This relationship helps explain why different parts of the spectrum produce different effects. Low-frequency radio waves are excellent for communication because they can travel long distances and can be detected electronically, but their photons do not usually have enough energy to trigger major chemical changes in tissue. By contrast, ultraviolet and X-rays can affect electrons much more strongly, which is why they can be useful in medicine but also potentially harmful.
The spectrum is not a list of unrelated categories. It is a pattern. Once you know how energy changes with frequency, you can begin predicting what types of interactions are likely in matter. That is exactly how science moves from small mechanisms to large outcomes.
[Figure 2] When radiation meets matter, several things can happen: it can be transmitted, reflected, scattered, or absorbed. Absorption matters most for cause-and-effect analysis because that is when energy is transferred into the material. Different frequencies interact with matter in distinct ways through the progression from molecular motion to electron changes and ionization.
At lower frequencies, absorbed energy often changes the motion of particles without ionizing atoms. Microwaves can cause certain polar molecules, especially water molecules, to rotate back and forth. Infrared radiation often increases molecular vibration. Both of these mechanisms increase internal energy, which we observe as a rise in temperature.
Visible light can raise electrons in atoms or molecules to higher energy states. That absorbed energy may later be released as light, converted to heat, or used to drive chemical reactions. Ultraviolet light has more energy and can trigger stronger chemical changes, including damage to biological molecules such as DNA. X-rays and gamma rays have still higher photon energies and can remove electrons from atoms or molecules, a process called ionization.
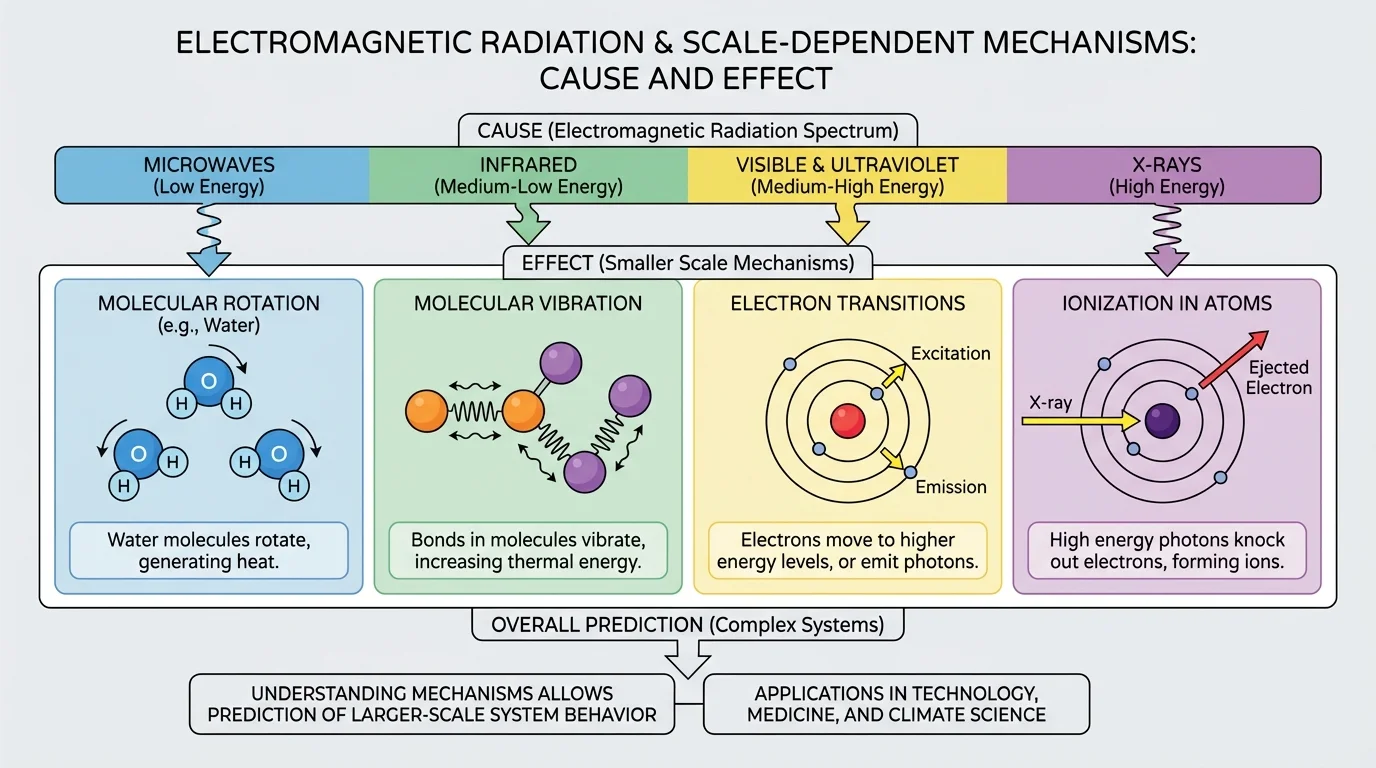
Absorption is the process in which matter takes in energy from electromagnetic radiation. Photon energy is the amount of energy carried by one packet of electromagnetic radiation and increases with frequency. Ionization is the removal of one or more electrons from an atom or molecule, creating charged particles.
Because absorption depends on structure, not every material absorbs every frequency equally well. Glass transmits much visible light but blocks much ultraviolet radiation. The atmosphere allows much visible light to pass but absorbs most X-rays. Dark clothing often absorbs more visible light than light-colored clothing and therefore may warm more in sunlight.
This is why a frequency alone does not tell the whole story. You must also ask what kind of matter is present, how much radiation is absorbed, and how the absorbed energy is redistributed. Those smaller details determine the larger effect.
Once matter absorbs radiation, the next step is tracing where the energy goes. Energy can spread through collisions between particles, trigger a chemical reaction, produce electrical signals in a detector, or damage a biological structure. The larger effect is a result of many tiny interactions added together.
Consider heating. If microwave radiation is absorbed by water-rich food, many molecules begin rotating more energetically. Through collisions, that energy spreads, increasing the food's thermal energy. The food feels hotter not because a single molecule became extremely energetic, but because enormous numbers of molecules shared the transferred energy.
We can describe thermal energy changes with the relationship \[Q = mc\Delta T\] where \(Q\) is the energy transferred as heat, \(m\) is mass, \(c\) is specific heat capacity, and \(\Delta T\) is temperature change. For example, if \(0.20 \textrm{ kg}\) of water in food absorbs \(16{,}720 \textrm{ J}\), using \(c \approx 4180 \textrm{ J/(kg }\cdot\textrm{ ^\circ C)}\), then \[\Delta T = \frac{Q}{mc} = \frac{16{,}720}{(0.20)(4180)} = 20^{\circ} \textrm{C}\]
Case study: Why microwaves heat food unevenly
A microwave oven is a human-designed system that depends on predictable interactions between waves and matter.
Step 1: The oven generates microwaves that enter the food.
Step 2: Water and some other polar molecules absorb the radiation and rotate.
Step 3: The rotating molecules collide with nearby particles, spreading energy as thermal motion.
Step 4: Because food has uneven water content, shape, and thickness, absorption is uneven, so some regions heat faster than others.
The large-scale effect, hot and cold spots, can be predicted from small-scale differences in absorption and energy transfer.
The same logic explains why infrared cameras detect warm objects. Warm surfaces emit more infrared radiation than cooler ones. A detector absorbs that radiation and converts the energy into electrical signals. Those signals are processed into a visible image. Tiny interactions in the sensor lead to useful information at the human scale.
[Figure 3] Natural systems often appear too complicated to predict, but the same cause-and-effect thinking works there too. In Earth's atmosphere and in living tissue, frequency-specific absorption allows scientists to predict which radiation reaches the ground and which frequencies create biological risks.
Ozone in the upper atmosphere absorbs much of the Sun's ultraviolet radiation. Because scientists understand the molecular absorption of ozone, they can explain why the atmosphere protects life from many high-energy photons. Greenhouse gases such as \(\textrm{CO}_2\) and water vapor absorb particular infrared frequencies, which helps explain why outgoing thermal radiation is partly trapped in the atmosphere.
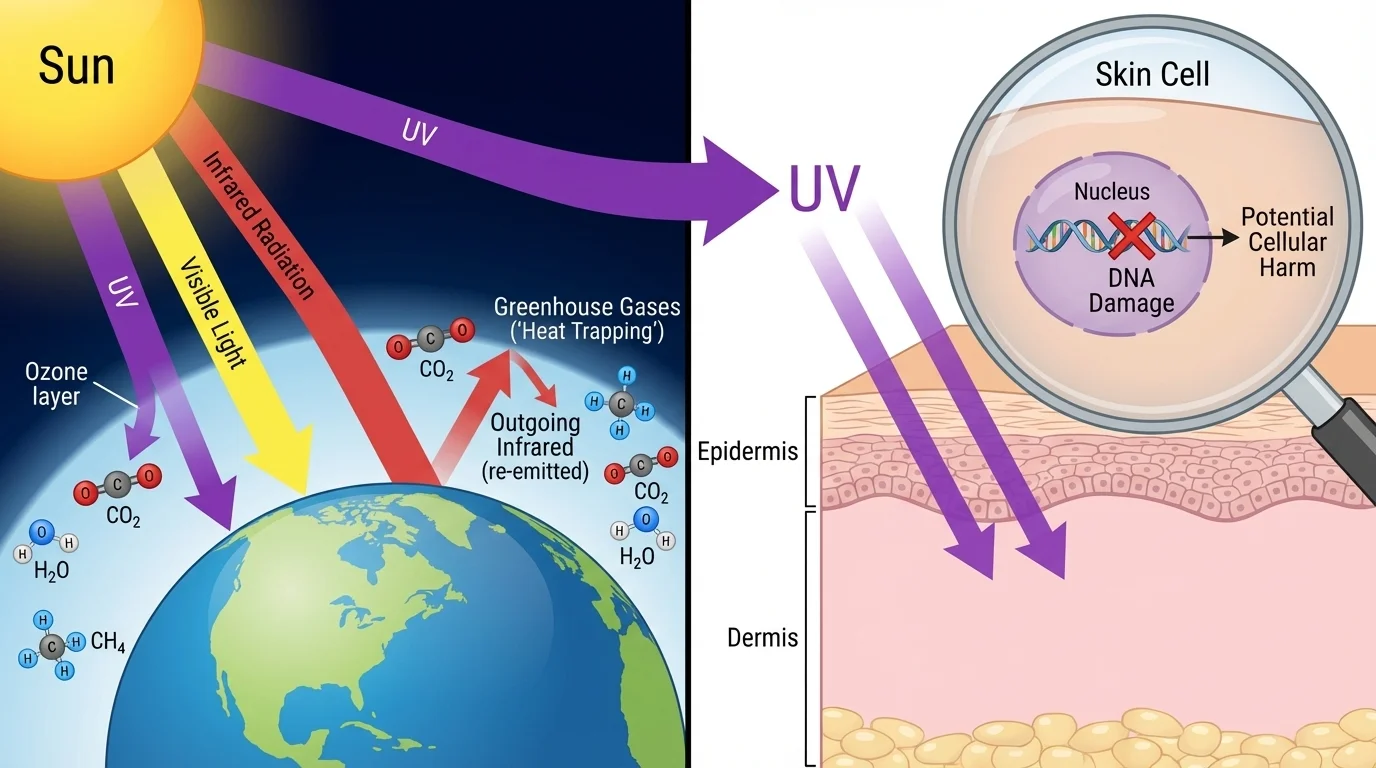
In the human body, skin and eye tissues absorb different frequencies differently. Visible light usually allows us to see because it is detected by light-sensitive molecules in the retina. Too much ultraviolet radiation can damage skin cells by altering molecular bonds and harming DNA. X-rays can pass through soft tissue more easily than bone, which is why they are useful for imaging skeletons, but they can also ionize atoms and damage cells if exposure is too high.
The same basic ultraviolet radiation can be beneficial in one context and harmful in another. Limited exposure can help the body produce vitamin D, but excessive exposure can increase the risk of skin damage and cancer because the underlying molecular mechanisms involve changes to biological molecules.
These examples show that prediction does not come from memorizing isolated facts. It comes from knowing what molecules absorb, what energies are involved, and what those absorbed energies do next.
[Figure 4] Engineers choose frequencies strategically because they know how those frequencies interact with matter. That design logic is visible in communication, medicine, industry, and household technologies. A technology works well when its chosen frequency matches the desired effect and avoids unwanted effects.
Radio systems use lower-frequency electromagnetic waves because they can carry information over long distances and be generated and detected efficiently. Fiber-optic communication often uses visible or near-infrared light because those frequencies travel effectively through specially designed glass fibers with low loss. In both cases, the design depends on understanding absorption, transmission, and signal detection at the small scale.
Medical technologies also depend on frequency choice. X-ray imaging works because bones absorb more X-rays than surrounding soft tissue, creating contrast on a detector. Ultraviolet sterilization works because UV photons can damage the genetic material of microbes, reducing their ability to reproduce. Infrared thermometers work by detecting thermal radiation emitted by surfaces.
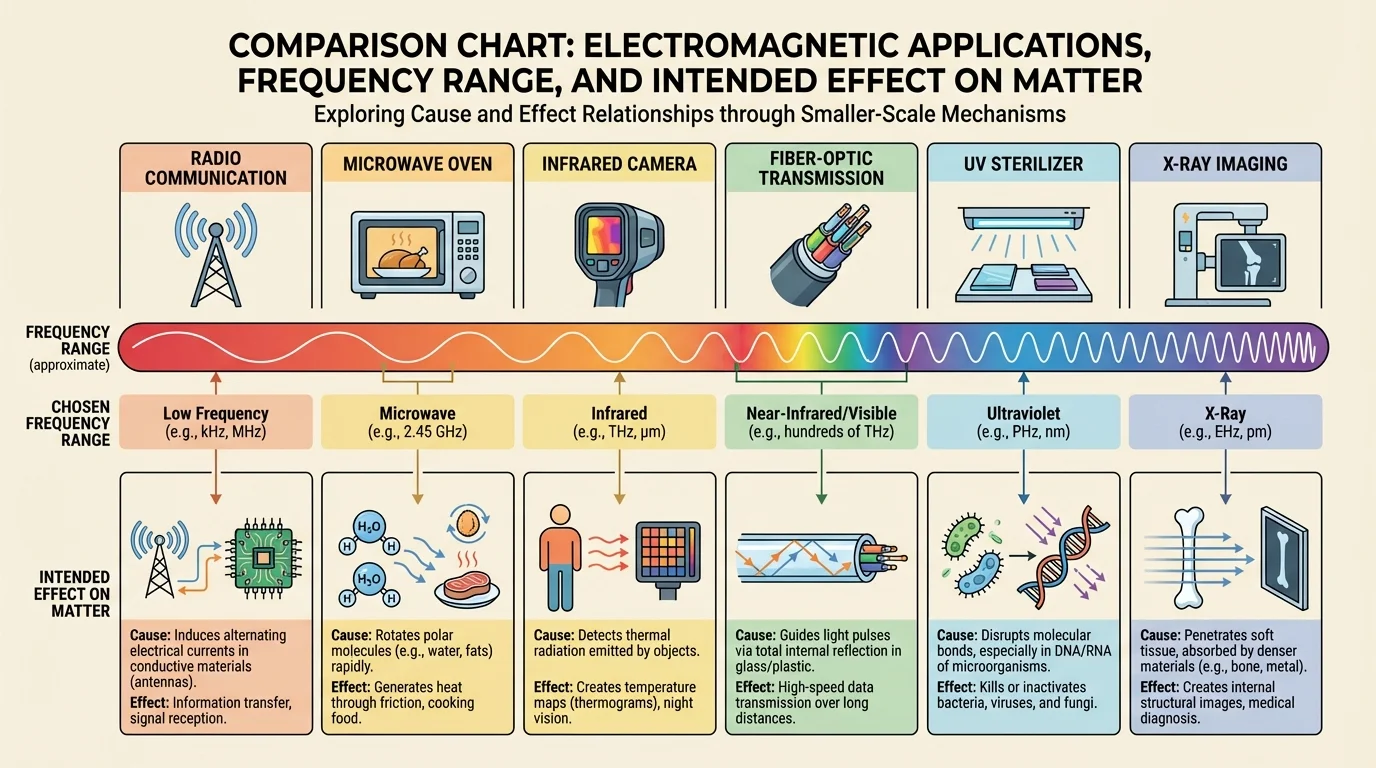
This comparison helps explain why radiation at one frequency can be useful in one device and dangerous in another context. A microwave oven uses shielding so the radiation stays inside the appliance, while a medical X-ray system uses controlled exposure, protective barriers, and strict timing. Design always includes control of cause and effect.
Human-designed systems are not separate from natural laws. They are built by taking advantage of them. Engineers do not invent how matter absorbs radiation; they learn the mechanism and use it.
[Figure 5] Published claims about electromagnetic radiation can sound convincing, especially when they use scientific words. A reliable evaluation method begins with a checklist: identify the frequency, the dose, the material absorbing it, the proposed mechanism, and the quality of the evidence. Without those pieces, a claim is weak.
First, ask whether the claim matches known physics. If a source claims that low-energy radiation produces effects that normally require ionization, that should immediately raise questions. Since photon energy follows \(E = hf\), not all frequencies can cause the same kinds of changes. Frequency matters because mechanism matters.
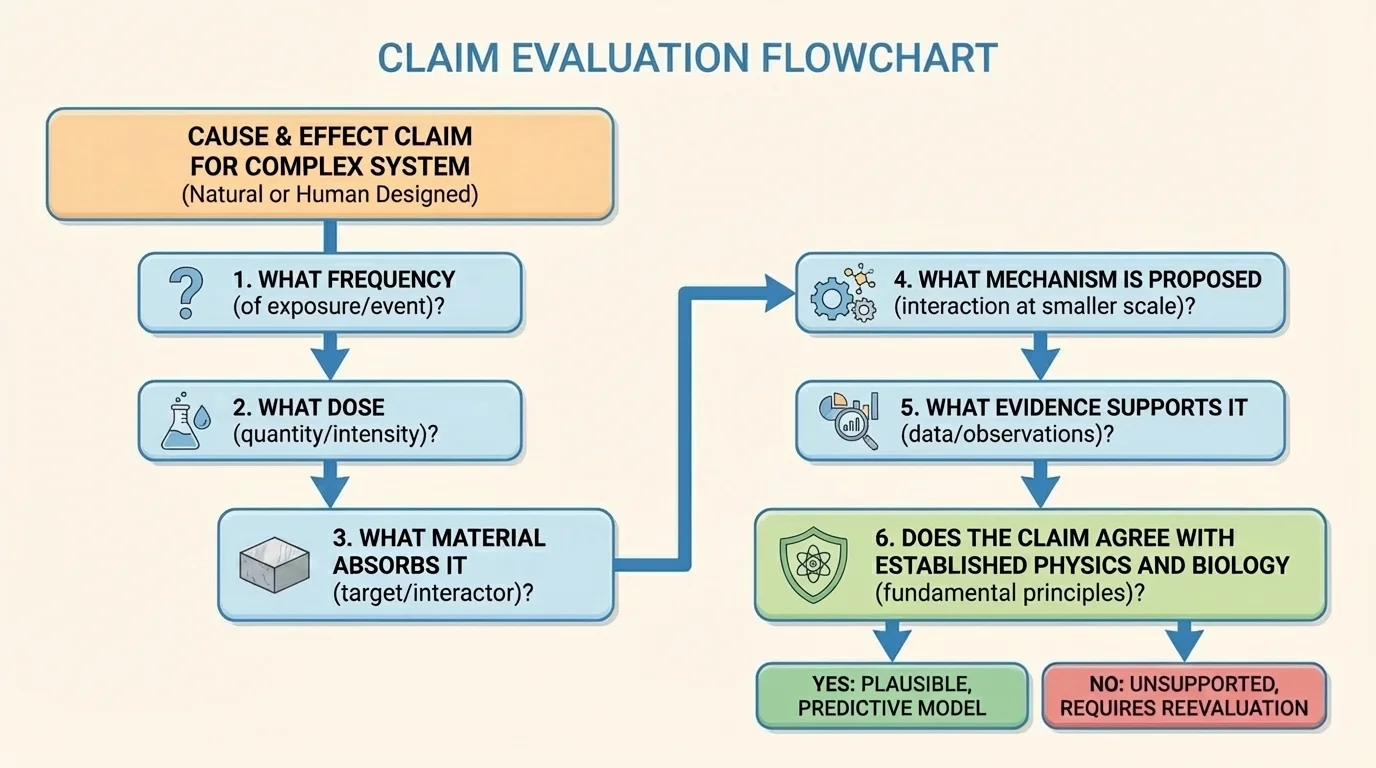
Second, examine the validity of the evidence. Validity asks whether the evidence really measures what the claim is about. For example, if a study on tissue heating uses much higher power than a normal consumer device, the results may not apply well to everyday exposure. A valid test must match the actual conditions being discussed.
Third, examine reliability. Reliability asks whether similar results appear when measurements are repeated, when different researchers test the same idea, or when larger samples are used. A single dramatic result is less convincing than repeated evidence from multiple studies.
Fourth, look for missing variables. Was the material the same in every trial? Were exposure time and intensity controlled? Was there a comparison group? Were other causes ruled out? Complex systems can mislead us if we ignore interacting factors.
| Question | Why it matters |
|---|---|
| What frequency is involved? | Different frequencies have different photon energies and mechanisms of interaction. |
| How much energy is absorbed? | Effects depend on dose, not just presence of radiation. |
| What matter absorbs it? | Different materials absorb different frequencies differently. |
| What mechanism is proposed? | A strong claim should connect to known atomic, molecular, or biological processes. |
| Is the evidence repeatable? | Reliable claims can be reproduced by others. |
| Does the test match real conditions? | Validity depends on whether the experiment actually represents the real-world situation. |
Table 1. Questions that help evaluate the validity and reliability of claims about electromagnetic radiation.
When students learn to ask these questions, they are doing more than checking facts. They are using cause-and-effect reasoning to judge whether a published claim deserves trust.
Cause and effect in radiation systems becomes clearer when you track energy. Energy is not created from nowhere. It is transferred and transformed. A source emits radiation, matter absorbs some of it, particles change motion or electronic state, and then larger effects follow.
"To understand a system, follow the energy."
— A guiding principle of physical science
This idea connects strongly to conservation of energy. If a surface warms when it absorbs infrared radiation, the increase in thermal energy comes from the radiation source. If a detector produces an electrical signal after absorbing visible light, the electrical response traces back to absorbed photon energy. The path may be complicated, but the chain is still there.
For example, suppose radiation transfers \(250 \textrm{ J}\) of energy to a sensor system, and \(190 \textrm{ J}\) becomes thermal energy while \(60 \textrm{ J}\) becomes electrical output. The total is conserved because \(190 + 60 = 250\). Following the energy helps verify whether an explanation is physically reasonable.
Good predictions are powerful, but they are not magical. In complex systems, many variables interact. A published claim may be partly true in one material and false in another. The same frequency may have different effects depending on intensity, duration, distance, shielding, and the internal structure of the absorbing material.
For example, the absorption of infrared radiation by one greenhouse gas molecule is a small-scale mechanism, but predicting climate change also requires considering clouds, oceans, land surfaces, feedback loops, and human activity. Likewise, predicting biological effects from radiation requires considering tissue type, repair mechanisms, and cumulative dose. Small-scale mechanisms are the foundation, but larger systems may add complications.
Still, prediction becomes far more reliable when it is rooted in known mechanisms. That is why scientists and engineers study atoms, molecules, and energy transfers so carefully. As we saw earlier in [Figure 1], the electromagnetic spectrum follows clear physical patterns, and as [Figure 2] shows, different frequencies lead to different interactions with matter. Those patterns allow us to make justified predictions rather than guesses.
Earlier physical science ideas remain essential here: energy can be transferred between systems, waves carry energy, and matter is made of atoms and molecules. This lesson extends those ideas by showing how the specific frequency of radiation helps determine the type of energy transfer and its effects.
Whether the system is a cell, an atmosphere, a medical scanner, or a wireless network, the same scientific habit applies: examine the small-scale mechanism, trace the energy transfer, and test whether the proposed cause really matches the observed effect. That is how science explains the world and how engineering designs technologies that work.