A hawk soaring above a field, a tree growing from sunlight, and a lizard warming itself on a rock may seem like very different events. But they are all connected by one powerful idea: energy is always moving and changing form. Scientists can track that movement through natural systems, from the Sun to plants, from plants to animals, and from warm ground to cool air. Once you learn to follow the energy, nature starts to make much more sense.
Every natural system depends on energy. A natural system is a group of parts in nature that interact with one another, such as a pond, forest, desert, ocean, or even the whole Earth. In each system, energy enters, moves, changes form, and leaves. Scientists track these changes to explain weather, food webs, body temperature, and many other processes.
One of the most important scientific ideas is that energy is conserved. This means energy is not created from nothing and does not simply disappear. Instead, it changes from one form to another and transfers from place to place. When a fox runs after a rabbit, the energy in the fox's muscles came from food. That food energy originally came from plants or from animals that ate plants. Those plants got their energy from sunlight.
"Energy cannot be created or destroyed, only transformed."
— First Law of Thermodynamics
Tracking energy helps scientists answer questions such as: Why are there fewer top predators than plants? Why does the ground cool after sunset? Why do animals use fur, feathers, fat, or burrows? Why do insulated bottles keep drinks warm or cold? These questions all involve energy transfer.
Energy appears in different forms. In natural systems, several forms are especially important. Radiant energy travels as light from the Sun. Chemical energy is stored in food, fuels, and living tissue. Kinetic energy is the energy of motion. Gravitational potential energy depends on an object's position, especially its height. Thermal energy is related to the motion of particles in matter.
Thermal energy is the total energy of moving particles within matter. A warmer object usually has particles moving faster on average than a cooler object.
Energy transfer happens when energy moves from one object, place, or part of a system to another.
Energy transformation happens when energy changes from one form into another, such as light energy becoming chemical energy in plants.
These forms often work together. Sunlight reaches a leaf as radiant energy. During photosynthesis, the plant transforms some of that energy into chemical energy stored in molecules such as glucose, \(\textrm{C}_6\textrm{H}_{12}\textrm{O}_6\). When an animal eats the plant, that chemical energy transfers into the animal's body. If the animal runs, some chemical energy becomes kinetic energy and thermal energy.
Not all energy stays in a form that living things can easily use. During almost every transfer, some energy spreads out as thermal energy into the surroundings. That does not mean the energy is gone. It means it has become less useful for doing certain jobs, such as building tissue or causing motion.
The sunlight that reaches Earth in about one hour contains more energy than humans use in a whole year. The challenge is not whether enough energy exists, but how to capture, transfer, and use it effectively.
Because energy changes form so often, scientists use diagrams, arrows, and measurements to trace where it comes from and where it ends up.
Most ecosystems get their energy from the Sun. [Figure 1] shows this pathway clearly with arrows. Plants, algae, and some bacteria capture sunlight and use it to make food. These organisms are called producers because they produce their own chemical energy-rich food.
Animals cannot capture sunlight directly in the same way, so they get energy by eating producers or by eating other animals. Herbivores eat plants. Carnivores eat other animals. Omnivores eat both. Decomposers, such as fungi and bacteria, break down dead organisms and waste. They also obtain energy from chemical compounds in this material.
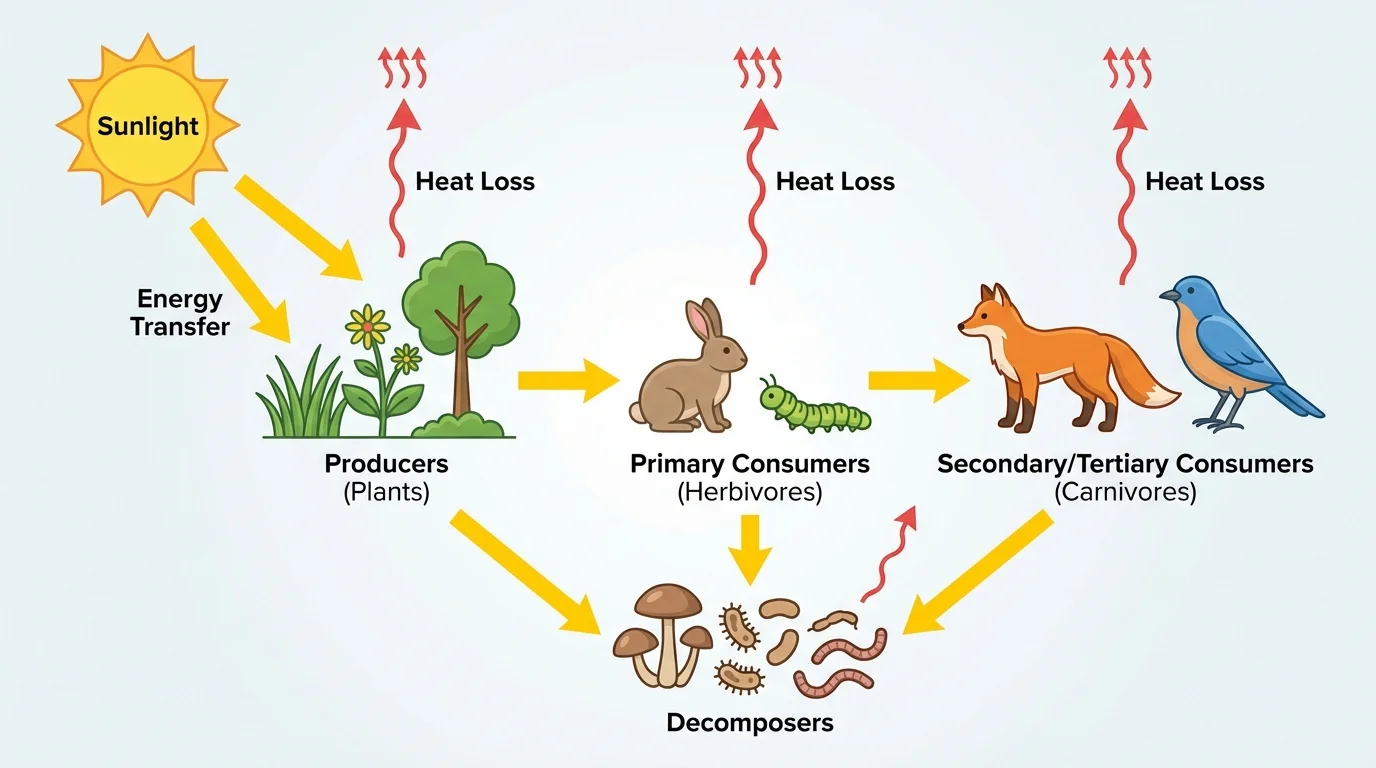
As energy moves from one feeding level to the next, less of it remains available in the form of chemical energy stored in living tissue. A large amount is used for life processes and released as thermal energy. That is why a field can support many plants, fewer rabbits, and even fewer hawks.
Photosynthesis is a major energy transformation in ecosystems. In a simplified form, it can be represented as
\[6\textrm{CO}_2 + 6\textrm{H}_2\textrm{O} + \textrm{light energy} \rightarrow \textrm{C}_6\textrm{H}_{12}\textrm{O}_6 + 6\textrm{O}_2\]
This equation shows that plants use carbon dioxide, water, and light energy to make glucose and oxygen. The energy from sunlight becomes stored in the chemical bonds of glucose.
Later, organisms release some of that stored energy during cellular respiration. A simplified equation is
\[\textrm{C}_6\textrm{H}_{12}\textrm{O}_6 + 6\textrm{O}_2 \rightarrow 6\textrm{CO}_2 + 6\textrm{H}_2\textrm{O} + \textrm{energy}\]
The energy released can power body processes, movement, growth, and repair. The same energy pathway seen earlier in [Figure 1] helps explain why energy flows through ecosystems while matter, such as carbon and water, cycles through them.
It is helpful to track energy through real natural systems. In a pond, sunlight warms the water and also powers photosynthesis in algae and aquatic plants. Tiny animals eat the algae. Small fish eat the tiny animals. Larger fish or birds may eat the small fish. At every step, some energy transfers to the next organism, and some spreads into the water and air as thermal energy.
Case study: Energy in a pond
Step 1: Sunlight reaches the pond surface.
Some radiant energy is absorbed by water and mud, increasing thermal energy.
Step 2: Producers store energy.
Algae and pond plants transform light energy into chemical energy in food molecules.
Step 3: Consumers transfer energy through feeding.
Insects, tadpoles, and fish obtain chemical energy by eating producers or other consumers.
Step 4: Decomposers continue the pathway.
Bacteria and fungi break down dead matter and release thermal energy during their life processes.
The pond system shows that energy flow continues even when organisms die.
In a forest, trees are major producers. Deer may eat leaves, and wolves may prey on deer. Birds, insects, fungi, and soil organisms are also part of the energy pathway. Fallen leaves do not keep their energy forever; decomposers use the chemicals in them. Meanwhile, sunlight also heats the forest floor, tree bark, and air.
In a desert, energy flow follows the same basic rules, but the conditions are different. Strong sunlight heats rocks and sand quickly during the day. Cacti store chemical energy through photosynthesis. Insects, reptiles, birds, and mammals obtain energy through feeding. Because deserts often cool quickly at night, thermal energy transfer is a major part of how desert organisms survive.
[Figure 2] helps show that thermal energy moves in three main ways, and natural scenes often include all three at once. These methods are conduction, convection, and radiation.
Conduction is the transfer of thermal energy through direct contact. If a lizard lies on a warm rock, energy transfers from the rock into the lizard's body. Metals usually conduct thermal energy well, while materials such as foam, wool, and air conduct it poorly.
Convection is the transfer of thermal energy by the movement of fluids, which means liquids or gases. Warm air rises because it is less dense, while cooler air sinks. This movement creates convection currents. In lakes and oceans, warm and cool water can also move in convection patterns.
Radiation is the transfer of energy by electromagnetic waves. Sunlight warms Earth through radiation. Unlike conduction and convection, radiation does not require matter, so solar energy can travel through space.
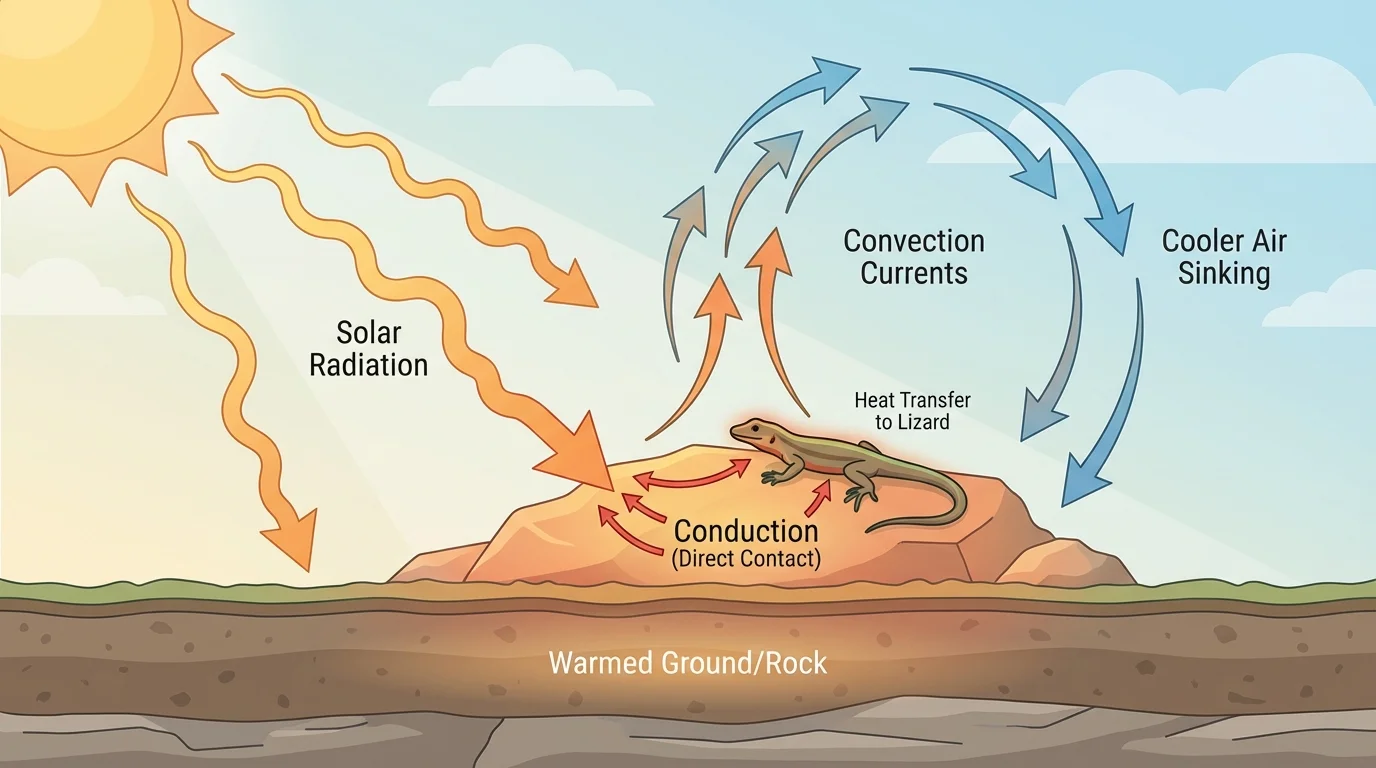
These three transfer methods often interact. Sunlight warms the ground by radiation. The ground then transfers thermal energy to nearby air by conduction. The warmed air rises, creating convection currents. This chain of transfers affects weather, climates, and local temperatures.
Animals and plants are strongly affected by thermal energy transfer. Fur traps air and reduces conduction and convection. Dark surfaces absorb more radiant energy than light surfaces. Some animals bask in sunlight to gain thermal energy, while others hide in shade or burrow underground to reduce energy gain. The scene in [Figure 2] helps connect these ideas to what happens outdoors every day.
Temperature and thermal energy are related but not identical. Temperature measures the average kinetic energy of particles, while thermal energy depends on both particle motion and the amount of matter present.
For example, a bathtub of warm water can contain more thermal energy than a cup of hot water, even if the cup has a higher temperature.
Scientists and engineers use what they know about thermal energy to design devices that either reduce or increase energy transfer. This is where science connects directly to solving real problems. A device does not need to be complicated. A lunch cooler, a greenhouse, an oven mitt, a solar cooker, and a thermos all involve controlling thermal energy transfer.
[Figure 3] illustrates how a design can minimize thermal energy transfer by using insulating materials, trapped air, reflective surfaces, or seals that reduce air movement. Foam coolers work because foam contains many tiny pockets of air, and air is a poor conductor. A tight lid reduces convection by slowing air exchange. Reflective materials can reduce energy transfer by radiation.
To maximize thermal energy transfer, a design might use dark surfaces, metal parts, larger surface area, or direct sunlight. A solar cooker often uses shiny reflectors to direct more radiant energy toward a dark cooking container. The dark container absorbs more solar energy and becomes hotter.
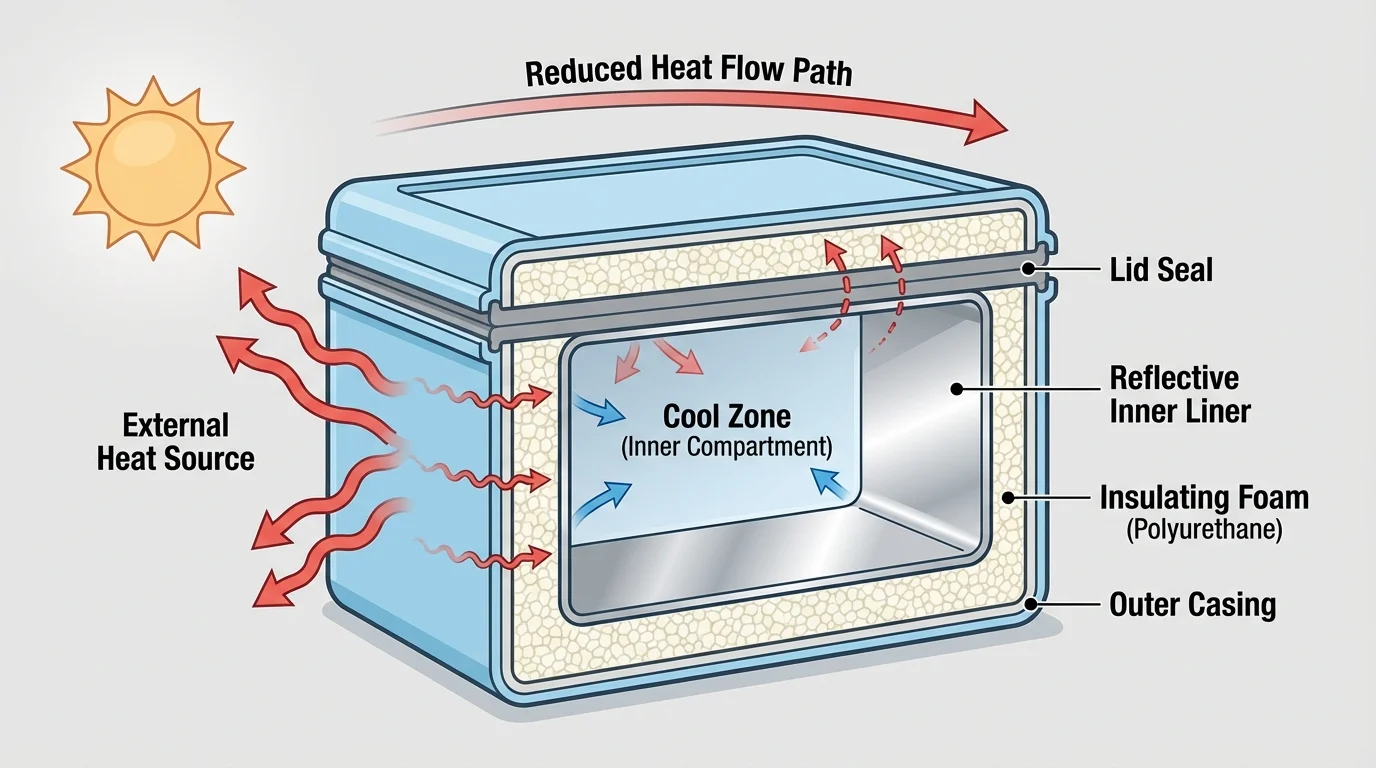
Good design depends on the goal. If the goal is to keep soup warm, the device should slow energy leaving the soup. If the goal is to heat water with sunlight, the device should increase energy entering the water and reduce energy escaping to the surroundings.
Designing with constraints
Engineers do not choose materials based only on science. They also consider cost, mass, safety, strength, and environmental impact. The best design is usually a balance between strong scientific performance and practical limits.
This design thinking also appears in nature. Polar bears have thick fur and fat that reduce heat transfer. Desert plants may have waxy coatings or shapes that reduce heating and water loss. Human-made devices often copy solutions already found in living systems.
Scientists often measure temperature to help track thermal energy transfer. If the temperature of an object changes, energy has likely entered or left the object. One useful relationship is
\[Q = mc\Delta T\]
In this formula, \(Q\) is the thermal energy transferred, \(m\) is mass, \(c\) is a material constant called specific heat, and \(\Delta T\) is the change in temperature. Middle school students do not always need to calculate with this formula often, but it shows an important idea: the amount of energy transfer depends on how much material there is and how much its temperature changes.
Numeric example: Comparing warming of water
A sample of water has mass \(m = 200 \textrm{ g}\), specific heat \(c = 4.18 \textrm{ J/g}^\circ\textrm{C}\), and warms by \(\Delta T = 5^\circ\textrm{C}\).
Step 1: Write the formula.
Use \(Q = mc\Delta T\).
Step 2: Substitute the numbers.
\(Q = 200 \cdot 4.18 \cdot 5\).
Step 3: Calculate.
\(200 \cdot 4.18 = 836\), and \(836 \cdot 5 = 4{,}180\).
The water gains \(Q = 4{,}180 \textrm{ J}\) of thermal energy.
This example shows that even a small temperature increase can involve a large energy transfer. If students test two containers holding the same amount of warm water, the container that loses temperature more slowly is better at reducing thermal energy transfer.
Scientists also compare designs by observing which one works best under the same conditions. For example, if one solar cooker heats water from \(20^\circ\textrm{C}\) to \(50^\circ\textrm{C}\) while another only reaches \(35^\circ\textrm{C}\) in the same time and sunlight, the first design transfers more solar energy into the water.
| Design goal | Helpful features | Why they work |
|---|---|---|
| Keep heat in | Foam, fabric, trapped air, lids | Reduce conduction and convection |
| Reflect thermal radiation | Shiny surfaces | Reduce absorption or redirect radiation |
| Absorb solar energy | Dark surfaces, direct sunlight | Increase radiant energy gain |
| Transfer heat quickly | Metal, large contact area | Increase conduction |
Table 1. Features that help devices minimize or maximize thermal energy transfer.
People constantly interact with natural energy flows. Houses are designed to stay warm in winter and cool in summer. Roof color affects radiant energy absorption. Windows can let solar energy in or out. Trees planted near buildings can provide shade, lowering heating by radiation. City materials such as asphalt and concrete absorb and store thermal energy, contributing to urban heat island effects.
Farmers use greenhouses to trap thermal energy and create better growing conditions. Campers use insulated sleeping bags to reduce body heat loss. Refrigerators move thermal energy from the inside to the outside. Even clothing is a kind of thermal device. In cold weather, layered clothing traps air and slows heat transfer. In hot weather, light-colored clothing reflects more sunlight.
The insulated design in [Figure 3] connects directly to these real-world examples. Whether the goal is protecting food, saving energy in buildings, or keeping people safe in extreme temperatures, the same science applies.
Students sometimes mix up energy flow with matter cycling. Energy and matter are related, but they do not behave in exactly the same way. Matter, such as water, oxygen, carbon, and minerals, is recycled through natural systems. Energy, however, usually enters a system, moves through it, changes form, and eventually spreads out, often as thermal energy.
For example, the carbon atoms in \(\textrm{CO}_2\) taken in by a plant may later become part of glucose, then part of an animal, and eventually return to the air. But the light energy that helped make that glucose does not cycle back as sunlight. It moves through the system and is transformed along the way.
This is why scientists say energy flows through ecosystems, while matter cycles. Understanding this difference makes it easier to read food webs, explain weather patterns, and design devices that control energy transfer.
When you look at nature through the idea of energy transfer, many separate facts connect into one clear pattern. The Sun powers photosynthesis, food provides chemical energy, organisms release thermal energy, and engineering uses these same rules to solve problems. Tracking energy through a natural system reveals both the unity of science and the usefulness of scientific thinking.