A forest can build massive trees from air, a pond can turn oxygen-rich at noon and oxygen-poor by night, and your own muscles can switch energy pathways in the middle of a sprint. These are not separate stories. They all reflect one big idea: energy drives the cycling of matter. Ecosystems are not just collections of organisms. They are dynamic systems in which atoms are constantly reused while energy is captured, transformed, transferred, and eventually released as heat.
To understand ecosystems deeply, it helps to separate two ideas that are often mixed together. Matter is anything that has mass and takes up space. The carbon in a leaf, the nitrogen in protein, the oxygen in water, and the phosphorus in DNA are all matter. Energy, by contrast, is the ability to do work or cause change. Organisms need energy to build molecules, move materials, grow, reproduce, and maintain internal balance.
Matter cycling means that atoms and molecules are repeatedly reused within and between living organisms and the nonliving environment.
Energy flow means that energy enters a system, is transformed and transferred, and eventually leaves as heat rather than being endlessly recycled in the same usable form.
In ecosystems, matter is conserved. A carbon atom in the air may become part of a sugar molecule in a plant, then part of an animal's muscle, then part of the atmosphere again as carbon dioxide. Energy behaves differently. Sunlight enters an ecosystem, some of it is captured by photosynthesis, and each energy transfer becomes less useful because some energy is dispersed as heat. That difference between cycling and flowing is one of the most important patterns in biology.
Every organism is part of a system that includes both biotic components, such as plants, animals, fungi, and microbes, and abiotic components, such as air, water, soil, minerals, and temperature. Life depends on interactions between these parts. Organisms cannot create atoms from nothing. They must obtain matter from their surroundings. They also cannot carry out life processes without energy transformations.
A useful analogy is a rechargeable battery. The materials in the device, such as metals and plastics, remain part of the object for a long time, though they may be rearranged or recycled later. But the battery's energy must be supplied again and again. Ecosystems work similarly, except on a much larger and more complex scale. The atoms are reused through cycles, while usable energy must keep entering, mostly from the Sun and, in a few special ecosystems, from chemical sources.
When scientists describe ecosystems as systems, they mean that different parts interact and affect one another. A change in oxygen levels in water can alter microbial respiration. That can change nutrient availability, which affects plant growth, which then changes how much matter is stored in biomass. In other words, energy transformations and matter cycling are linked at every level.
Atoms are not destroyed in ordinary biological processes. Instead, they are rearranged into new molecules. This idea from chemistry helps explain why matter can cycle through ecosystems again and again.
That is why a biological explanation must track both the path of atoms and the source and transformation of energy. If you only follow matter, you miss why reactions happen. If you only follow energy, you miss what is actually being built, broken down, and moved through the environment.
[Figure 1] Within an ecosystem, matter moves repeatedly among organisms and the physical environment through exchanges among air, water, soil, plants, animals, and decomposers. A plant takes in carbon dioxide, water, and mineral nutrients. It uses those materials to build carbohydrates, lipids, proteins, and other molecules. An herbivore eats the plant and incorporates some of that matter into its own body. Waste, dead tissue, and dead organisms are then processed by decomposers, returning atoms to the soil, water, and atmosphere.
The main biological roles in this movement are producers, consumers, and decomposers. Producers, such as plants and algae, convert inorganic substances into organic molecules using an energy source. Consumers obtain matter by eating other organisms. Decomposers, including many fungi and bacteria, break down dead organic material and wastes, releasing simpler substances back into the environment.
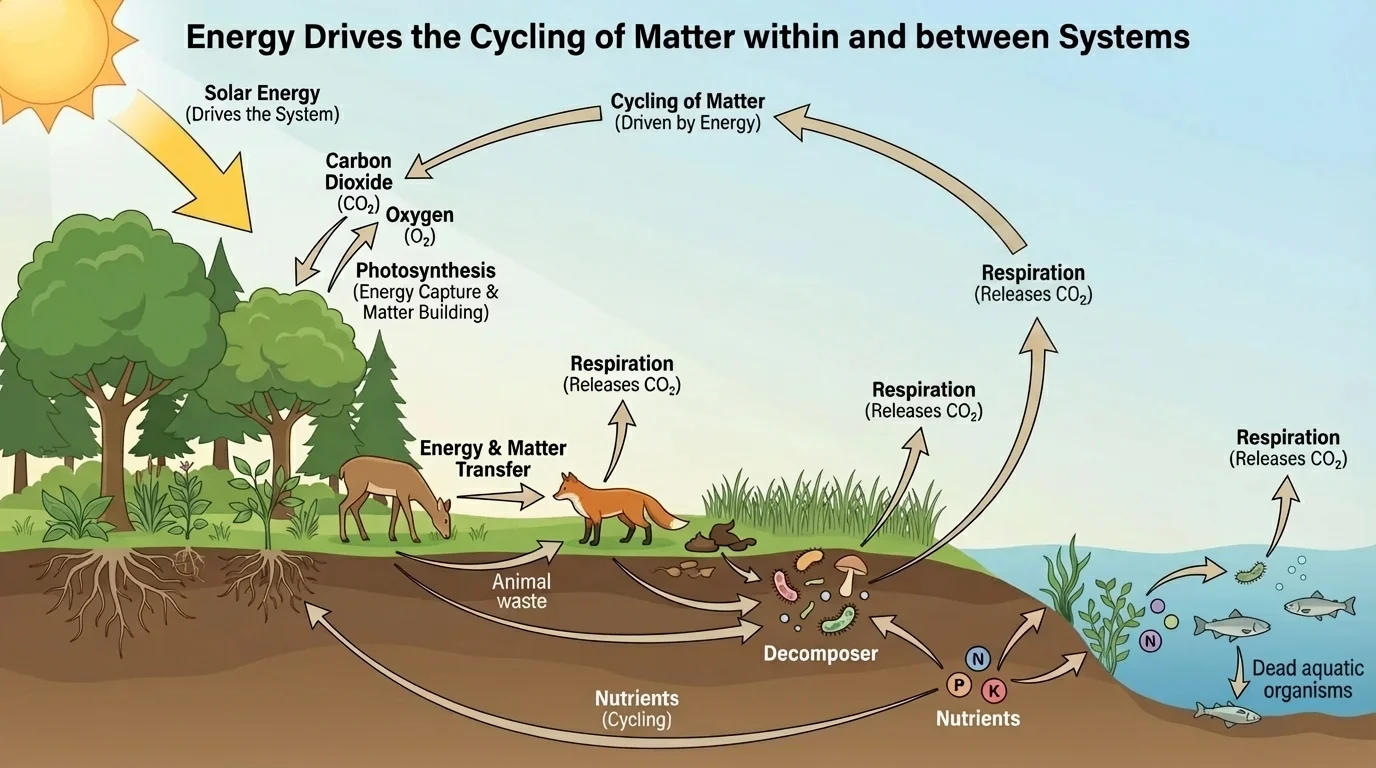
This cycling happens both within systems and between systems. For example, carbon moves within a forest ecosystem among trees, insects, birds, fungi, and soil microbes. But carbon also moves between the forest and the atmosphere as \(\textrm{CO}_2\) is absorbed and released. Water can evaporate from a lake, move through the atmosphere, and return as precipitation in a different ecosystem. Rivers transport dissolved nutrients from land to wetlands, lakes, and oceans.
One of the clearest examples is the carbon cycle. Carbon enters many ecosystems when producers take in \(\textrm{CO}_2\) during photosynthesis. It moves through food webs as organisms eat one another. It returns to the atmosphere through respiration, decomposition, and combustion. Some carbon is stored for long periods in soils, sediments, forests, and fossil fuels. The same atom may move through air, water, organisms, and rocks over very different timescales.
The movement of water also connects systems. Organisms contain a great deal of \(\textrm{H}_2\textrm{O}\), but water is constantly entering and leaving bodies through drinking, food, excretion, evaporation, transpiration, and respiration. A tree pulls water from soil and releases much of it through leaves into the atmosphere. That water can later condense into clouds and fall elsewhere. Matter is therefore local and global at the same time.
[Figure 2] The tight connection between photosynthesis and respiration is one of the best examples of how matter cycling depends on energy transformations. In photosynthesis, producers capture light energy and store some of it in chemical bonds. In cellular respiration, organisms release energy from those bonds to make ATP, the immediate energy carrier used by cells.
Photosynthesis can be summarized by the equation
\[6\textrm{CO}_2 + 6\textrm{H}_2\textrm{O} + \textrm{light energy} \rightarrow \textrm{C}_6\textrm{H}_{12}\textrm{O}_6 + 6\textrm{O}_2\]
This equation shows that carbon dioxide and water are rearranged into glucose and oxygen. Energy from sunlight is not matter, but it drives the reaction. For a numeric example, if a plant uses 6 molecules of \(\textrm{CO}_2\) and 6 molecules of \(\textrm{H}_2\textrm{O}\), it can form 1 molecule of \(\textrm{C}_6\textrm{H}_{12}\textrm{O}_6\) and 6 molecules of \(\textrm{O}_2\), assuming the reaction proceeds exactly as written.
Cellular respiration in aerobic conditions is often summarized as
\[\textrm{C}_6\textrm{H}_{12}\textrm{O}_6 + 6\textrm{O}_2 \rightarrow 6\textrm{CO}_2 + 6\textrm{H}_2\textrm{O} + \textrm{ATP} + \textrm{heat}\]
Here, glucose is broken down in the presence of oxygen. The carbon, hydrogen, and oxygen atoms are rearranged into carbon dioxide and water. Some of the chemical energy is transferred into ATP, and some is released as heat. For a simple numeric example, if 1 glucose molecule reacts with 6 oxygen molecules, the products are 6 carbon dioxide molecules and 6 water molecules in the balanced equation.
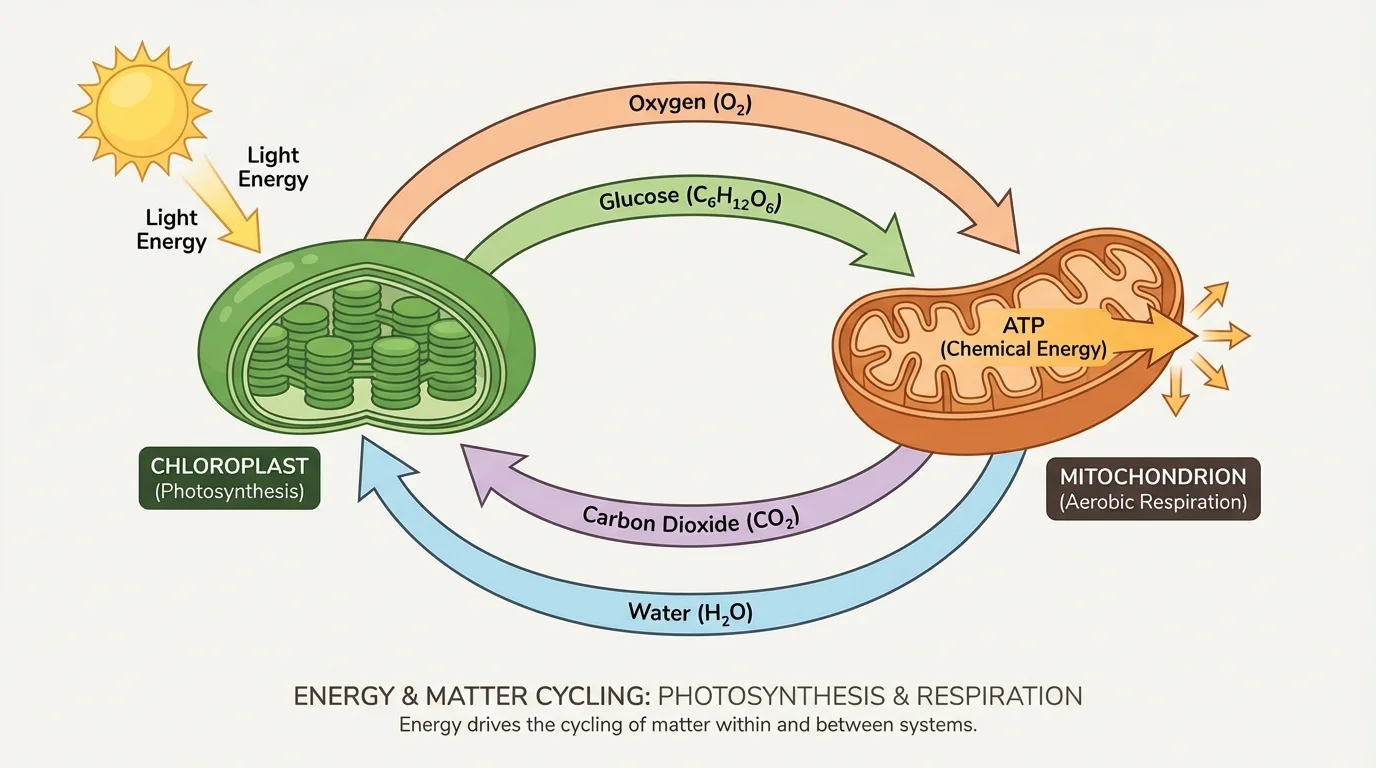
Notice what is cycling here: the atoms in \(\textrm{CO}_2\), \(\textrm{H}_2\textrm{O}\), \(\textrm{O}_2\), and glucose move through different forms. Notice what is flowing: energy enters as light, is stored in glucose, transferred into ATP, used for cellular work, and then partly released as heat. The matter can be reused. The usable energy cannot be reused indefinitely in the same way.
ATP links energy capture and energy use. Cells do not use sunlight or food energy directly for most tasks. They use ATP, which stores smaller, manageable amounts of energy that can be released to power transport, movement, synthesis, and signaling. That makes ATP a key bridge between ecosystem energy flow and cell-level activity.
Plants perform photosynthesis, but they also carry out cellular respiration. So do animals, fungi, protists, and many bacteria. Even a tree that makes glucose in daylight must respire continuously to power its cells. Ecosystems are therefore shaped by both building reactions and breakdown reactions happening at the same time.
[Figure 3] Not all environments contain the same amount of oxygen, and cells respond differently when oxygen is present or absent. Aerobic conditions involve oxygen. Anaerobic conditions involve little or no oxygen. These differences strongly affect how energy is released from organic matter and what products are formed.
In aerobic respiration, oxygen serves as the final electron acceptor in a chain of reactions that allows cells to extract a relatively large amount of energy from glucose. In many biology courses, this is described as yielding about 36 to 38 ATP per glucose in simplified textbook models, though actual values can vary. The key idea is that aerobic pathways release more usable energy than anaerobic ones.
When oxygen is unavailable, some cells switch to fermentation. In animal muscle cells, intense exercise can lead to lactate fermentation. In yeast, alcoholic fermentation produces ethanol and carbon dioxide. These pathways allow glycolysis to continue, but they produce much less ATP overall than aerobic respiration. A common simplified value is 2 ATP per glucose.
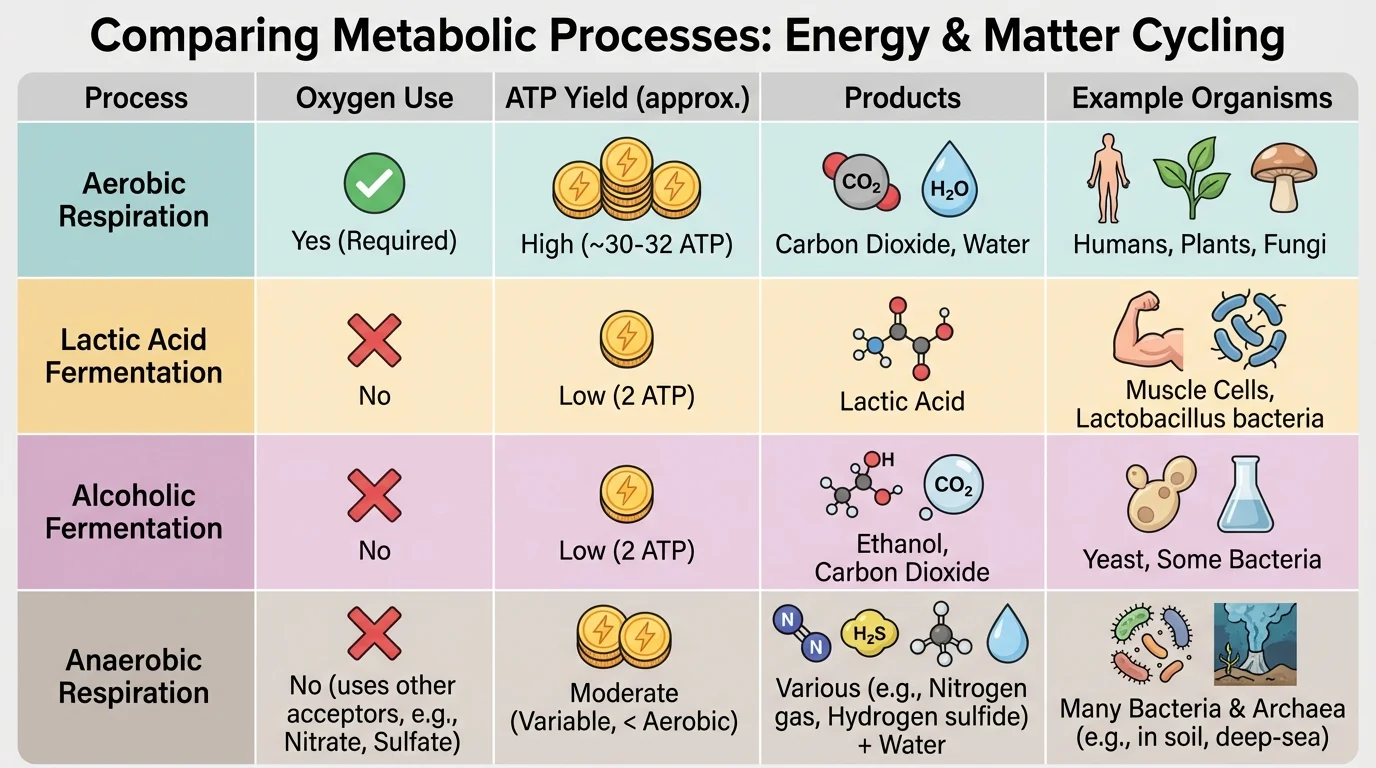
Microorganisms also perform forms of anaerobic respiration that use substances other than oxygen as final electron acceptors. Some bacteria use nitrate, sulfate, or carbon dioxide in their metabolic pathways. These processes are especially important in waterlogged soils, deep sediments, swamps, animal intestines, and poorly oxygenated water.
A pond provides a good ecological example. Near the surface, algae and aquatic plants produce oxygen by photosynthesis during daylight. Fish and aerobic microbes use that oxygen for respiration. But deeper in muddy sediments, oxygen may be quickly used up. There, anaerobic bacteria continue breaking down organic matter, producing substances such as methane, hydrogen sulfide, or ammonium depending on the pathway. The same ecosystem can therefore contain both aerobic and anaerobic zones.
Your own body also illustrates this shift. During rest or moderate exercise, muscle cells rely mostly on aerobic respiration. During a short, intense sprint, oxygen delivery may not keep up with demand, so fermentation helps maintain ATP production for a limited time. This is one reason muscles can become painful and fatigued during maximal effort. The chemistry inside your cells reflects the same principles that govern wetlands and sediments.
Comparing ATP yield in two conditions
Suppose one cell breaks down 3 glucose molecules aerobically, while another breaks down 3 glucose molecules by fermentation using the common simplified values above.
Step 1: Aerobic estimate
If aerobic respiration yields about 36 ATP per glucose, then \(3 \times 36 = 108\) ATP are produced.
Step 2: Fermentation estimate
If fermentation yields about 2 ATP per glucose, then \(3 \times 2 = 6\) ATP are produced.
Step 3: Compare
The aerobic cell produces \(108 - 6 = 102\) more ATP molecules than the fermenting cell in this simplified comparison.
This large difference helps explain why oxygen availability changes organism activity and ecosystem processes so strongly.
The products also matter ecologically. Carbon dioxide released by respiration can be reused in photosynthesis. Organic acids, alcohols, methane, and other products of anaerobic pathways influence soil chemistry, atmospheric composition, and the survival of other organisms.
When organisms die or release waste, the matter in their bodies does not disappear. It becomes the raw material for decomposers and detritivores. Fungi secrete enzymes that break complex molecules into smaller ones. Bacteria continue the process, releasing carbon dioxide, water, and mineral nutrients. Without decomposition, nutrients would remain locked in dead material, and ecosystems would quickly run short of usable resources.
This is why a fallen log is not just rotting wood. It is an active site of transformation. Carbon compounds are being broken down. Nitrogen in proteins can be converted into ammonium and then into other forms by microbes. Phosphorus from nucleic acids and membranes can return to soil solution. New plants can then absorb those nutrients and use them to build new tissue.
As seen earlier in [Figure 1], decomposers connect the living and nonliving parts of ecosystems by returning matter to the environment in forms that other organisms can use. In forests, grasslands, agricultural soils, and compost piles, this recycling supports future growth.
Some wetlands store huge amounts of carbon because waterlogged, low-oxygen conditions slow decomposition. When those wetlands are drained, decomposition speeds up and much of that stored carbon can return to the atmosphere.
The nitrogen cycle is especially important because organisms need nitrogen to make amino acids and nucleic acids, but most cannot use atmospheric \(\textrm{N}_2\) directly. Certain bacteria convert \(\textrm{N}_2\) into biologically useful forms. Other bacteria transform nitrogen compounds among forms such as ammonium, nitrite, and nitrate. Plants absorb these ions, animals obtain nitrogen by eating plants or other animals, and decomposers return nitrogen to the environment.
Oxygen conditions affect nitrogen transformations too. Some microbial steps happen mainly in oxygen-rich soil, while others occur more readily in low-oxygen conditions. That means moisture, aeration, temperature, and microbial communities all influence how rapidly nutrients cycle.
[Figure 4] Although matter can be reused again and again, energy transfer becomes less efficient at each step in a food chain. Organisms use much of the energy they obtain for metabolism, movement, repair, transport, and maintenance. A substantial portion is released as heat. Because of this, ecosystems require a continual energy input.
Trophic levels are feeding positions in a food chain or food web. Producers form the base because they capture energy directly, usually from sunlight. Primary consumers eat producers. Secondary consumers eat primary consumers, and so on. At each transfer, only part of the energy becomes biomass available to the next level.
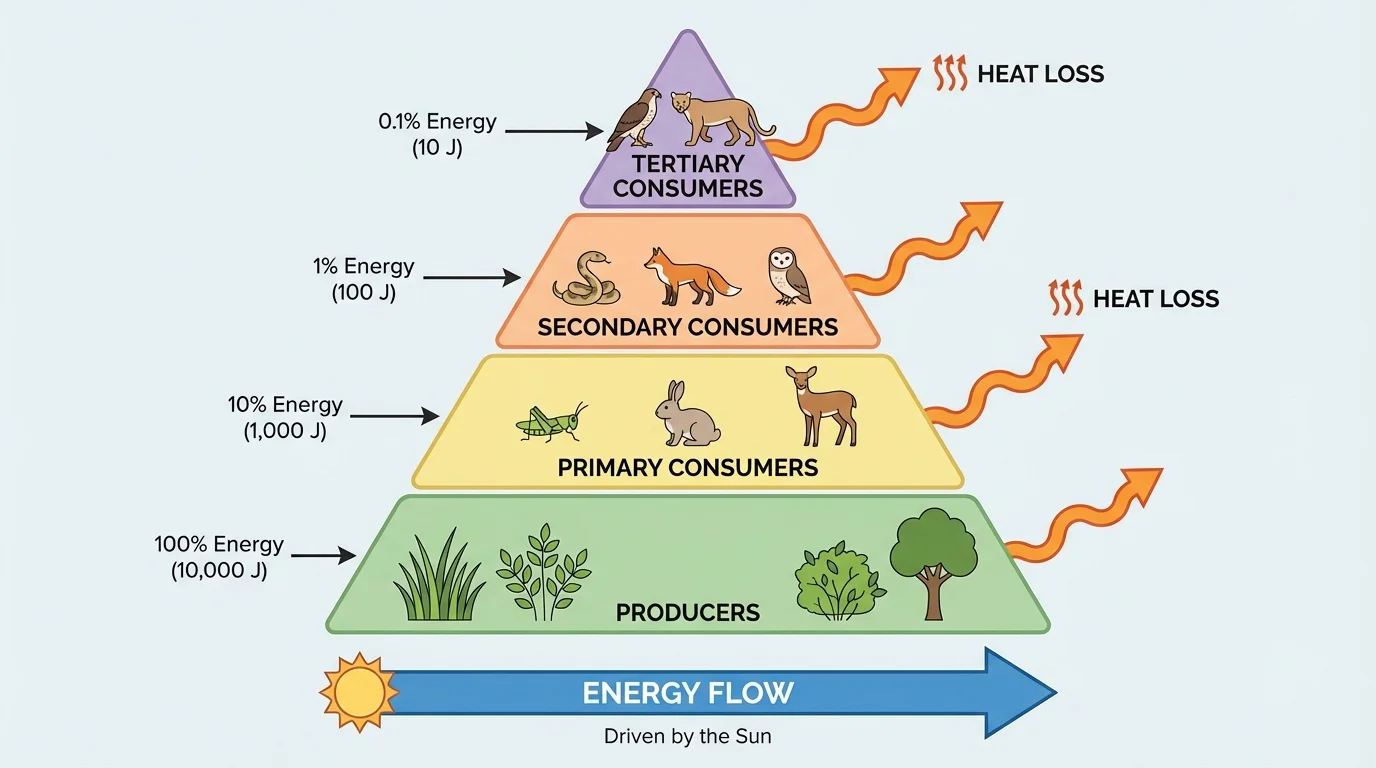
A common rough estimate is the 10 percent rule, which says that only about \(10\%\) of energy at one trophic level becomes biomass at the next. This is a simplification, but it is useful for understanding why food chains are usually short.
For a numeric example, if producers in a meadow store 10,000 units of chemical energy, then primary consumers may store about 1,000 units, secondary consumers about 100 units, and tertiary consumers about 10 units. In simplified form, that progression is \(10{,}000 \rightarrow 1{,}000 \rightarrow 100 \rightarrow 10\). The steep drop explains why there are typically fewer top predators than producers.
| Level | Main source of matter | Main source of energy | What happens to matter | What happens to energy |
|---|---|---|---|---|
| Producers | \(\textrm{CO}_2\), \(\textrm{H}_2\textrm{O}\), minerals | Sunlight or chemical sources | Built into biomass | Stored in chemical bonds |
| Consumers | Food from other organisms | Chemical energy in food | Rebuilt into body tissues or released as waste | Transferred, used, and partly lost as heat |
| Decomposers | Dead organisms and wastes | Chemical energy in detritus | Returned to soil, water, and air | Transferred, used, and partly lost as heat |
Table 1. Comparison of how matter and energy move through major biological roles in ecosystems.
This difference between matter and energy is essential. A carbon atom from a dead leaf may later become part of soil organic matter, a bacterial cell, a grass root, or atmospheric \(\textrm{CO}_2\). But the energy once stored in that leaf does not endlessly circulate in the same concentrated form. Much of it eventually becomes dispersed heat.
Human activities alter both matter cycles and energy flow. Burning fossil fuels transfers carbon from long-term underground storage to the atmosphere as \(\textrm{CO}_2\). Deforestation reduces the amount of carbon captured by photosynthesis. Fertilizer runoff adds nitrogen and phosphorus to waterways, often causing algal blooms. When those algae die, decomposition by microbes can consume dissolved oxygen and create low-oxygen or anaerobic zones.
This chain of events is not just a textbook process. It affects fisheries, drinking water, and coastal ecosystems. In severe cases, low dissolved oxygen leads to hypoxia, a condition in which oxygen levels are too low for many aquatic organisms to survive. The matter from fertilizer increases production at first, but the later energy demands of decomposition can destabilize the system.
Wastewater treatment plants intentionally use both aerobic and anaerobic microbes. In some stages, oxygen is supplied so aerobic bacteria can rapidly break down organic matter. In other stages, anaerobic microbes help process sludge and may produce methane, which can be captured as an energy source. This is a practical example of managing biological pathways by controlling environmental conditions.
Composting also depends on these principles. In a well-aerated compost pile, aerobic decomposers dominate and break down organic material efficiently. If the pile becomes too compacted and oxygen falls, anaerobic conditions increase, often producing slower decomposition and stronger odors. Gardeners, farmers, and engineers all rely on the same biology when they manage oxygen, moisture, and temperature.
"Nothing in nature is wasted; it is transformed."
— A principle reflected in ecosystem cycling
Climate change connects strongly to these ideas as well. Warmer temperatures can speed decomposition in some regions, releasing more carbon dioxide. Thawing permafrost may expose previously frozen organic matter to microbial breakdown, including anaerobic processes that generate methane. Small cellular reactions, multiplied across vast landscapes, can influence the global atmosphere.
Scientific explanations about matter cycling and energy flow are built from evidence, not guesswork. Scientists measure gas concentrations, biomass, nutrient levels, temperature, soil chemistry, dissolved oxygen, and rates of photosynthesis and respiration. They compare conditions, track changes over time, and test how systems respond to disturbances.
For example, if dissolved oxygen in pond water falls overnight while carbon dioxide rises, that evidence supports the idea that respiration is dominating over photosynthesis in darkness. If oxygen is near zero in sediment but methane is being released, that supports the presence of anaerobic microbial processes. If adding oxygen speeds the breakdown of organic waste in a treatment system, that supports a role for aerobic decomposition.
Scientists can also use isotopes to trace matter. Carbon atoms labeled in \(\textrm{CO}_2\) can later be detected in plant sugars, herbivore tissues, or respired gases, revealing the movement of matter through a system. Measurements of ATP production, heat release, and oxygen consumption help reveal how energy is being transformed.
The linked pathways in [Figure 2] and the comparative processes in [Figure 3] help explain why different environmental conditions produce different evidence patterns. Oxygen-rich settings tend to support more energy-rich aerobic pathways, while oxygen-poor settings favor fermentation or other anaerobic pathways with different products and ecological effects. Likewise, the decreasing energy available across trophic levels in [Figure 4] helps explain observed limits on biomass at higher consumer levels.
Good scientific explanations are also revisable. If new data show that an ecosystem has hidden low-oxygen zones, overlooked microbial pathways, or unexpected nutrient inputs, the explanation must change. Biology is not a set of isolated facts. It is an evidence-based effort to understand how energy transformations drive the movement and reuse of matter within and between systems.