A phone battery dies, a car brakes to a stop, a campfire fades, and a star shines for billions of years. In each case, energy seems to appear, vanish, or be "used up." But physics says something more surprising: energy never disappears. It moves, spreads out, and changes form. Once you start tracking where energy goes, many everyday events make more sense—from why machines heat up to how the Sun can power life on Earth.
People often say that energy is "made" by a power plant or "used up" by a light bulb. Those phrases are common, but they are not literally correct. A power plant does not create energy from nothing. Instead, it transfers energy stored in fuel, flowing water, wind, or atomic nuclei into electrical energy. A light bulb does not destroy energy. It transfers electrical energy into light and thermal energy.
This idea becomes clearer when we define a system. A system is the part of the universe we choose to study. Everything outside it is the surroundings. If the system is a bouncing basketball, we might track energy within the ball, between the ball and the floor, and into the surrounding air. If the system is a car engine, we track the chemical energy of the fuel as it is transformed during combustion, the motion of parts, heat flowing away, and sound leaving the system.
Earlier science courses may have introduced matter conservation in physical and chemical changes. That idea still matters here: careful accounting is one of the central habits of science. In energy studies, we do the same kind of accounting, but we track energy flows and transformations rather than simply counting substances.
The reason this topic fits under flows, cycles, and conservation is that understanding a system means following what crosses its boundaries and what changes inside it. If you lose track of energy leaving as heat, sound, or radiation, the system seems mysterious. If you account for all transfers, the behavior becomes understandable.
The law of conservation of energy states that the total energy of an isolated system remains constant. Energy can be transferred from one object to another or transformed from one form to another, but it cannot be created or destroyed.
Energy is the capacity to cause change, such as motion, heating, or radiation.
Transfer means energy moves from one place, object, field, or system to another.
Transformation means energy changes from one form to another within a system.
Isolated system means a system that exchanges neither matter nor energy with its surroundings.
Real systems are often not isolated. A moving bicycle slows down because energy is transferred to the surroundings as thermal energy in the tires, road, and air, and some as sound. The total energy is still conserved, but if you only watch the bicycle's motion, it may seem that energy has disappeared. It has not. It has just spread into forms that are less useful for the purpose you care about.
This is one of the most important scientific habits: always ask where did the energy go? That question turns a confusing event into a traceable process.
"Energy cannot be created or destroyed, only transformed or transferred."
— Statement of the law of conservation of energy
In many situations, energy also moves through fields. For example, when a magnet pulls on iron or when Earth pulls an object downward through gravity, energy can be transferred without direct contact. Electrical, magnetic, and gravitational fields allow interactions across space.
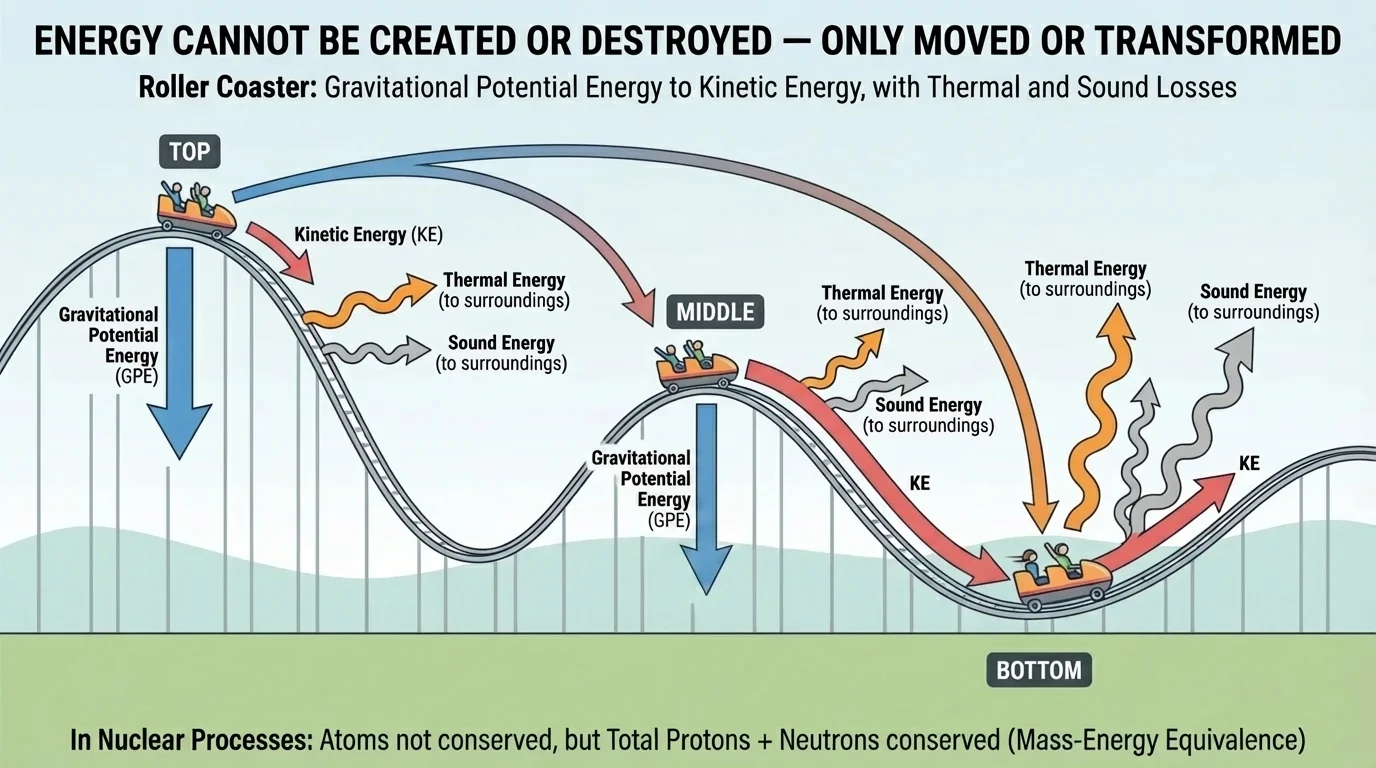
Energy appears in many forms: kinetic energy of motion, gravitational potential energy due to height, elastic potential energy in stretched or compressed materials, thermal energy associated with particle motion, chemical energy in bonds, electrical energy, radiant energy carried by electromagnetic waves, and nuclear energy associated with atomic nuclei. In a roller coaster, energy changes form continuously as the car moves from the top of a hill to the bottom and back up again.
[Figure 1] At the top of a hill, a coaster has a large amount of gravitational potential energy. As it descends, that energy is transformed into kinetic energy. At the bottom, its speed is greatest, so its kinetic energy is largest. But even then, not all the original energy remains in the motion. Friction in the wheels and air resistance transfer some energy to the surroundings as thermal energy and sound.
Energy can move in several main ways. One is by work, when a force acts through a distance. Another is by heating, when energy is transferred because of a temperature difference. Another is by radiation, such as visible light, infrared, ultraviolet, X-rays, or radio waves. Energy also moves through electrical currents and through waves in materials, such as sound waves.
A speaker system is a good modern example. Electrical energy from an amplifier transfers to the speaker cone, causing it to vibrate. That vibration transfers energy to the air as sound waves. Some energy also becomes thermal energy in the wires and speaker materials. No energy is lost from the universe; it is redistributed.
The Sun sends energy to Earth across nearly empty space. That transfer does not need air or any other material medium because radiant energy travels as electromagnetic waves.
The same logic applies to food and muscles. Chemical energy stored in molecules from food is transformed by the body into motion, thermal energy, and electrical activity in nerves. Athletes do not "create" energy when they sprint. Their bodies transfer and transform chemical energy already present in their cells.
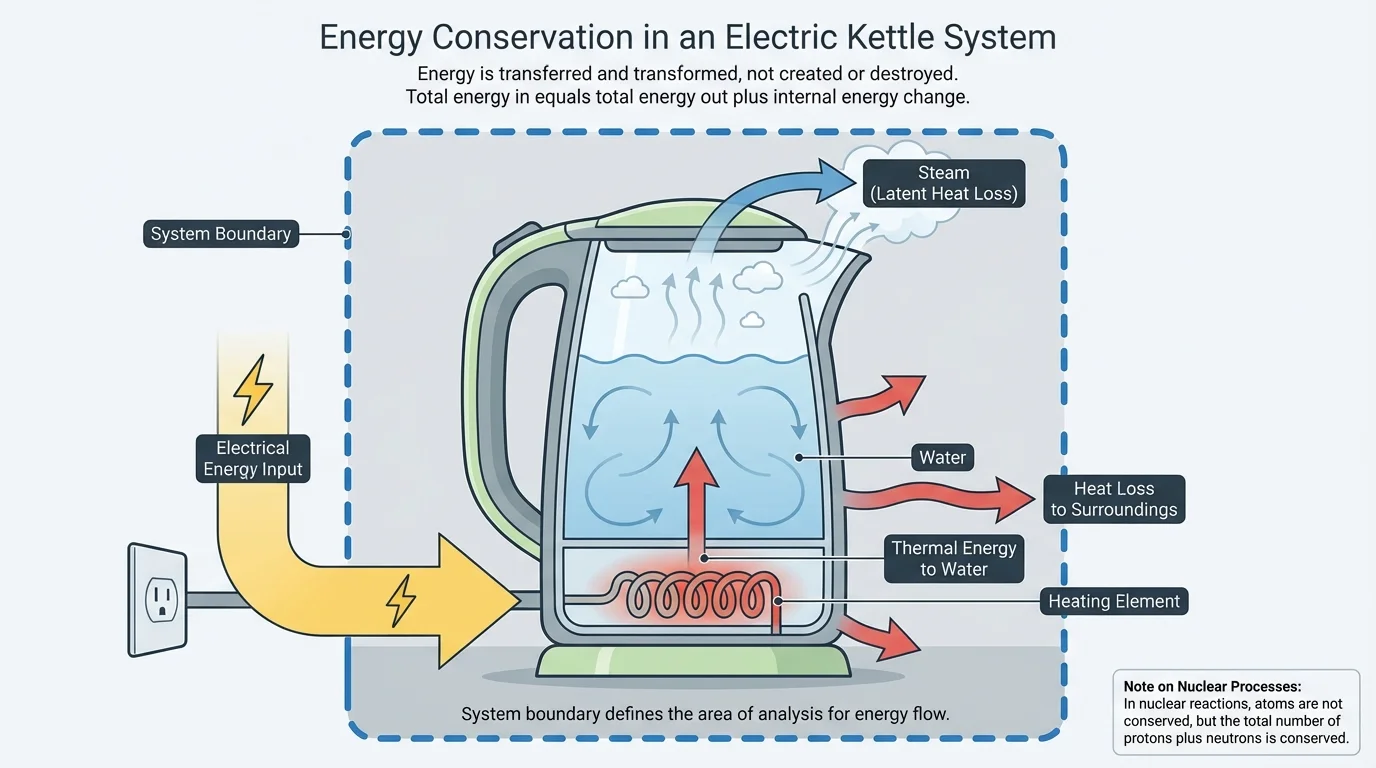
Scientists and engineers often begin by drawing a system boundary, because the answer depends on what is included inside the system. If the system is an electric kettle alone, electrical energy enters, thermal energy accumulates in the metal and water, and some heat leaves to the room. If the system is the kettle plus the room, that same "lost" heat is no longer leaving the system; it is staying within it.
[Figure 2] This is why tracking fluxes into, out of, and within systems helps us understand possibilities and limitations. A machine can only be as efficient as the energy transformations inside it allow. If large amounts of energy flow away in unwanted forms, such as heat or sound, less remains for the intended task.
Consider a power station. Chemical or nuclear energy is transformed into thermal energy, then into mechanical energy of spinning turbines, and finally into electrical energy. At each stage, some energy is transferred to the surroundings. Engineers try to reduce those unwanted transfers, but they can never make them zero in real devices.
When energy becomes more spread out, especially as low-temperature thermal energy, it is often less useful for doing work. That does not violate conservation. It explains why every real system has limits. A battery can power a phone only until the stored chemical energy has been transformed and redistributed. A car engine cannot turn all fuel energy into forward motion because some energy inevitably transfers to the engine block, exhaust gases, tires, and air.
Tracking energy also helps with environmental questions. If a building loses thermal energy rapidly through its walls and windows, it requires more energy input for heating. Better insulation does not create energy; it reduces energy transfer to the surroundings.
Some formulas help us calculate energy changes in simple situations. For motion, the kinetic energy of an object is
\[E_k = \frac{1}{2}mv^2\]
where \(m\) is mass and \(v\) is speed. Gravitational potential energy near Earth's surface is
\(E_p = mgh\)
where \(g \approx 9.8 \textrm{ m/s}^2\) and \(h\) is height.
Example 1: Falling backpack
A backpack with mass \(2.0 \textrm{ kg}\) falls from a shelf \(1.5 \textrm{ m}\) high. Find its gravitational potential energy relative to the floor before it falls.
Step 1: Choose the formula.
Use \(E_p = mgh\).
Step 2: Substitute the values.
\(E_p = (2.0)(9.8)(1.5)\)
Step 3: Calculate.
\(E_p = 29.4 \textrm{ J}\)
The backpack has gravitational potential energy of \(29.4 \textrm{ J}\) relative to the floor.
If air resistance is small, that potential energy is transformed mainly into kinetic energy as the backpack falls. Just before impact, its kinetic energy is approximately the same amount, about \(29.4 \textrm{ J}\).
Example 2: Moving skateboard
A skateboarder and board have a total mass of \(60 \textrm{ kg}\) and move at \(4.0 \textrm{ m/s}\). Find the kinetic energy.
Step 1: Use the kinetic energy formula.
\(E_k = \dfrac{1}{2}mv^2\)
Step 2: Substitute the values.
\(E_k = \dfrac{1}{2}(60)(4.0)^2\)
Step 3: Calculate carefully.
\((4.0)^2 = 16\), so \(E_k = 30 \cdot 16 = 480 \textrm{ J}\)
The skateboarder has kinetic energy of \(480 \textrm{ J}\).
Efficiency tells us how much input energy becomes useful output energy:
\[\textrm{efficiency} = \frac{\textrm{useful output energy}}{\textrm{total input energy}} \times 100\%\]
Example 3: Lamp efficiency
A lamp receives \(100 \textrm{ J}\) of electrical energy and produces \(15 \textrm{ J}\) of visible light. The rest becomes mostly thermal energy. Find the efficiency for light production.
Step 1: Write the formula.
Efficiency \(= \dfrac{\textrm{useful output}}{\textrm{input}} \times 100\%\)
Step 2: Substitute.
Efficiency \(= \dfrac{15}{100} \times 100\%\)
Step 3: Simplify.
Efficiency \(= 15\%\)
The lamp is \(15\%\) efficient at producing visible light.
These calculations are simple, but the deeper idea is powerful: numbers let us verify that energy is being accounted for, not magically appearing or vanishing.
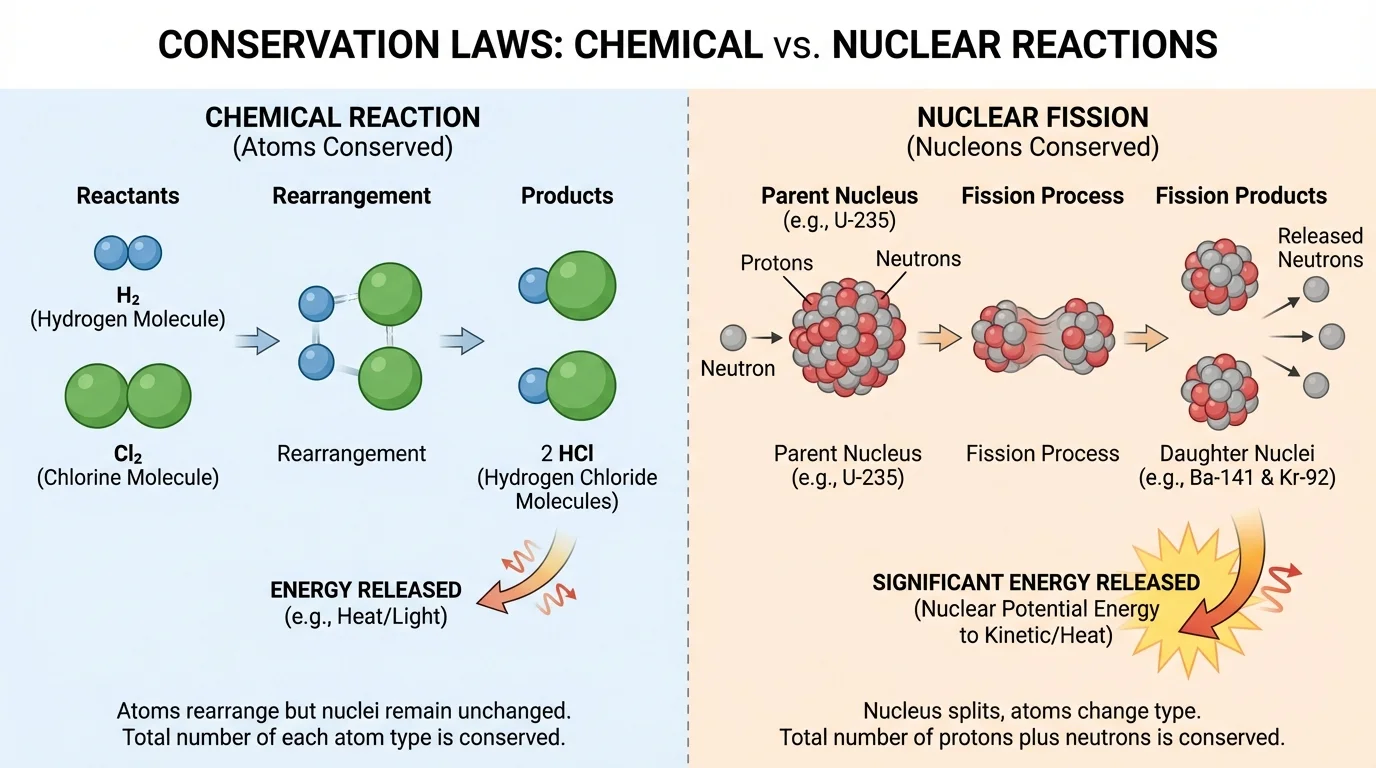
In ordinary physical and chemical processes, atoms are conserved. Ice melting into water keeps the same water molecules, \(\textrm{H}_2\textrm{O}\). In a chemical reaction such as
\[2\textrm{H}_2 + \textrm{O}_2 \rightarrow 2\textrm{H}_2\textrm{O}\]
the atoms are rearranged, but the same numbers of hydrogen and oxygen atoms are present before and after. Chemical reactions change electron arrangements and bonds, not the identities of atomic nuclei.
This is why balancing chemical equations works by keeping atom counts equal on both sides. Matter is not being created or destroyed; it is being reorganized.
| Process type | What changes | What is conserved |
|---|---|---|
| Physical change | State or arrangement | Atoms, mass, energy |
| Chemical reaction | Bonds and molecular structure | Atoms of each element, total energy |
| Nuclear process | Nuclei and sometimes element identity | Total energy, charge, total protons plus neutrons |
Table 1. Comparison of what changes and what is conserved in physical, chemical, and nuclear processes.
Nuclear reactions are different from chemical reactions. In a nuclear process, the nucleus changes. That means one element can change into another. So atoms are not conserved in nuclear processes in the same way they are in chemistry. However, an important count is conserved: the total number of protons plus neutrons.
[Figure 3] The combined number of protons and neutrons in nuclei is often called the nucleon number or mass number. In nuclear equations, the total nucleon number before and after a reaction must balance. Electric charge must also balance.
For example, uranium-235 can undergo fission after absorbing a neutron. A simplified fission equation is
\[{}^{235}_{92}\textrm{U} + {}^{1}_{0}\textrm{n} \rightarrow {}^{141}_{56}\textrm{Ba} + {}^{92}_{36}\textrm{Kr} + 3{}^{1}_{0}\textrm{n} + \textrm{energy}\]
Check the conservation: on the left, the total nucleon number is \(235 + 1 = 236\). On the right, it is \(141 + 92 + 3 = 236\). Charge also balances because \(92 = 56 + 36\).
In fusion, small nuclei combine to form a larger nucleus. In stars, hydrogen nuclei fuse into helium. One simplified fusion reaction is
\[4{}^{1}_{1}\textrm{H} \rightarrow {}^{4}_{2}\textrm{He} + \textrm{energy}\]
The detailed process in stars involves several steps, but the key idea remains: the particles are rearranged at the nuclear level, energy is released, and conservation laws still hold.
Radioactive decay is another nuclear process. In alpha decay, a nucleus emits a helium nucleus, \({}^{4}_{2}\textrm{He}\). In beta decay, a neutron can change into a proton or a proton into a neutron, with other particles emitted. The parent atom changes into a different atom, so atom identity is not conserved. But total energy, charge, and nucleon accounting remain consistent with the reaction type.
The difference from chemistry is huge. Burning coal changes carbon and oxygen atoms into \(\textrm{CO}_2\); the atoms remain carbon and oxygen. Nuclear reactions can actually change which element is present.
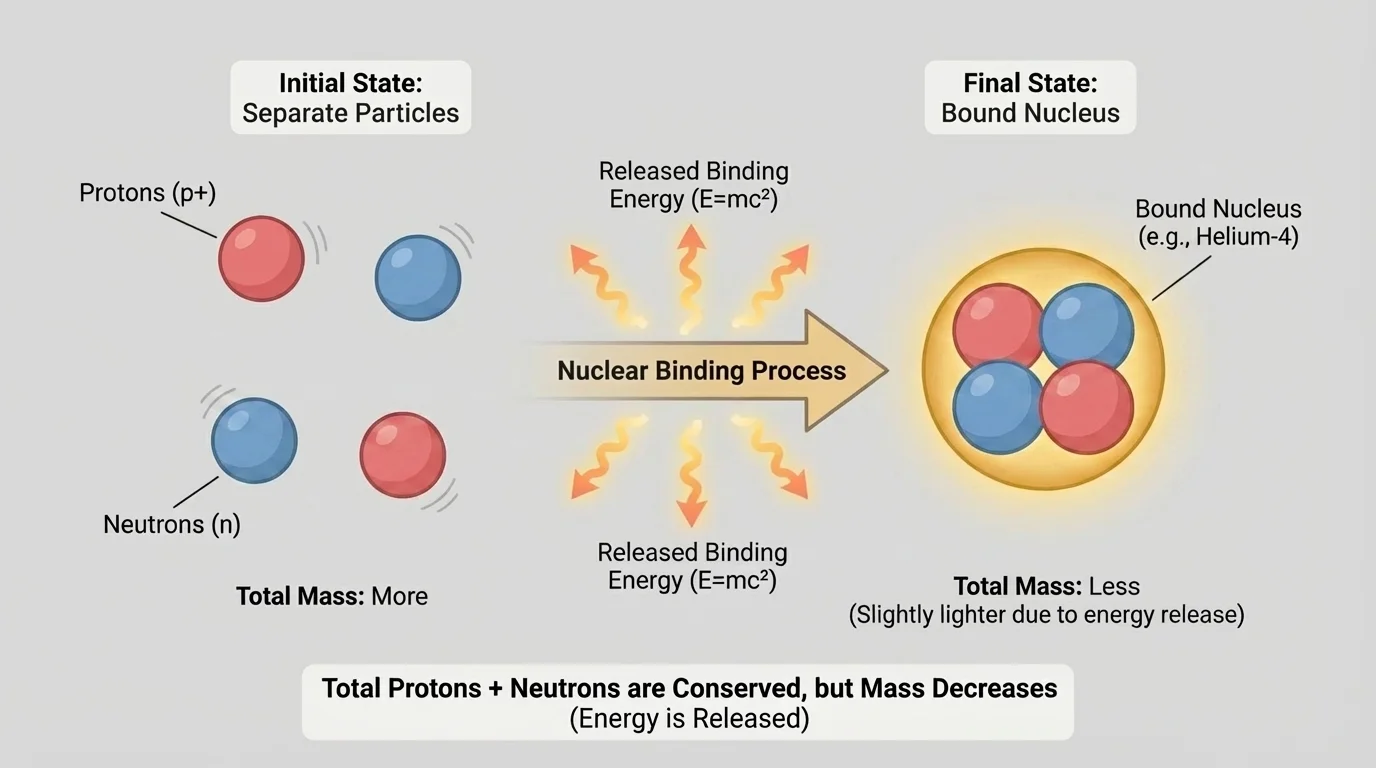
Why do some nuclear reactions release so much energy? The answer involves mass defect and binding energy. A bound nucleus has slightly less mass than the total mass of the separate protons and neutrons that make it up. The missing mass has been converted into energy associated with binding the nucleus together.
[Figure 4] This is where Einstein's famous relationship applies:
\(E = mc^2\)
Because \(c\), the speed of light, is extremely large, even a small amount of mass corresponds to a very large amount of energy.
Here is a simple numerical example. Suppose \(1.0 \times 10^{-6} \textrm{ kg}\) of mass is converted into energy. Then
\(E = mc^2 = (1.0 \times 10^{-6})(3.0 \times 10^8)^2\)
\((3.0 \times 10^8)^2 = 9.0 \times 10^{16}\), so
\[E = 9.0 \times 10^{10} \textrm{ J}\]
That is an enormous amount of energy from a tiny mass. This helps explain why nuclear fuels release far more energy per kilogram than chemical fuels.
Why conservation still works in nuclear reactions
In nuclear processes, it may seem as if matter disappears because the products can have slightly less mass than the reactants. But when mass and energy are treated together, conservation still holds. Some mass becomes emitted energy, such as kinetic energy of particles or electromagnetic radiation. So the deeper conservation law is conservation of total mass-energy.
This does not mean mass vanishes without a trace. It means mass is one way energy can be stored. In nuclear changes, that stored energy can be released when nuclei move to a more stable arrangement.
Nuclear power plants use fission to release energy from heavy nuclei. That energy heats water, produces steam, spins turbines, and generates electricity. The chain from nuclear energy to electrical energy is another example of flow through a system. As with any power plant, some energy is transferred to the surroundings as waste heat. That is why cooling systems are necessary.
Stars are natural fusion reactors. The Sun transfers radiant energy to Earth, driving weather, photosynthesis, and most food webs. The energy in fossil fuels originally came from ancient sunlight captured by organisms. So even many energy resources on Earth connect back to energy flows from a star.
Medicine also uses nuclear processes. Radiation therapy can damage cancer cells. Medical imaging techniques can detect radiation from radioactive tracers inside the body. In each case, understanding how energy transfers into tissues and instruments is essential for both effectiveness and safety.
Engineers care about conservation because it sets hard limits. No machine can produce more energy output than the total energy input. Claims of "free energy" devices fail because they violate the conservation principle. Devices can store energy, transfer it, or transform it, but not create it from nothing.
Nuclear fusion in stars creates many of the elements heavier than hydrogen. The calcium in bones and the iron in blood ultimately come from ancient stellar processes.
Climate science also depends on energy accounting. Earth receives radiant energy from the Sun and emits infrared radiation back into space. If incoming and outgoing energy are not balanced, the planet warms or cools. Once again, understanding the system means tracking what enters, what leaves, and what is retained.
Looking back to the kettle in [Figure 2], the same system logic applies on every scale—from household appliances to planets. Boundaries, transfers, and transformations determine the behavior we observe.
One common mistake is saying that energy is destroyed when motion stops. If a ball rolls and then stops, its kinetic energy has not vanished. It has been transferred mainly into thermal energy in the ball, floor, and air, with a little sound.
Another mistake is thinking that atoms are always conserved in every process. That is true in chemical reactions, but not in nuclear ones. As we saw in [Figure 3], nuclear reactions can change one element into another because nuclei themselves change.
A third mistake is assuming conservation means all energy remains useful. Conservation only says the total amount stays constant. It does not guarantee that energy remains concentrated in a form we want. Spread-out thermal energy is still energy, but it is often less useful for powering machines.
Finally, some students think \(E = mc^2\) replaces conservation of energy. It actually strengthens it. The equation shows that mass can be part of the energy accounting, especially in nuclear processes.