A basketball bounces lower each time, a phone gets warm while charging, and bicycle brakes heat up when you stop quickly. At first, it can seem as if energy just disappears. But one of the most important ideas in science says the opposite: energy is always accounted for. If an object loses kinetic energy, that energy must show up somewhere else at the same time.
This idea helps explain motion, heating, cooling, weather, cooking, machines, and even why ice melts in your drink. Energy can move from place to place, and it can change from one form to another, but the total amount stays the same. Understanding where energy goes is the key to understanding many changes we see every day.
Conservation of energy means energy cannot be created or destroyed; it can only be transferred or transformed.
Energy transfer is energy moving from one object, place, or system to another.
Energy transformation is energy changing from one form to another, such as kinetic energy changing into thermal energy.
Scientists often track energy the way a detective tracks clues. If one kind of energy decreases, another kind must increase somewhere. Sometimes the change is easy to notice, like a moving ball stopping and making a sound. Sometimes it is harder to notice, like tiny increases in temperature in the ball, the floor, and the air.
When we say energy is conserved, we do not mean it is "saved up" in one place. We mean the total amount stays constant, even while it changes form. A flashlight, for example, changes chemical energy in its battery into light energy and thermal energy. The battery's stored energy decreases, while energy is transferred to the surroundings by light and heating.
This is why scientists are careful with the phrase "energy is lost." In everyday speech, people say that energy is lost when a machine slows down or when a ball stops rolling. In science, the energy is not truly gone. It has been transferred to the surroundings, often as sound or thermal energy. The total still balances.
"Energy cannot be created or destroyed, only transformed and transferred."
— A central idea of physics
Suppose a cart is rolling across the floor and then stops. Its kinetic energy becomes smaller. Where does that energy go? Some is transferred to the floor and wheels as thermal energy because of friction. Some is transferred to the air as sound. A tiny amount may also go into bending the wheels or floor for a moment. Even when the changes are too small to notice easily, they still happen.
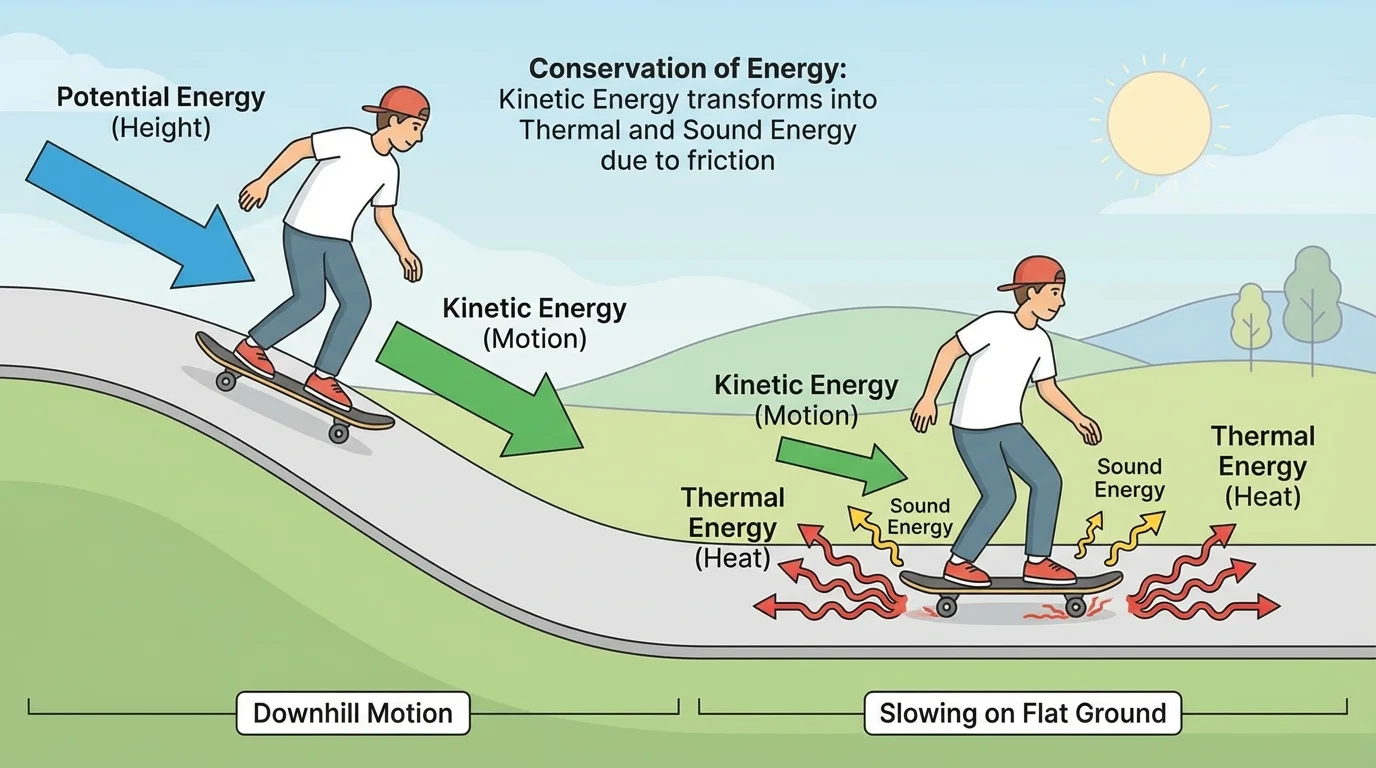
Kinetic energy is the energy an object has because it is moving. The faster an object moves, the more kinetic energy it has. When motion changes, as shown in [Figure 1], energy must be transferred or transformed at the same time. A skateboarder who slows down does not make energy vanish; the energy changes form.
If the skateboarder rolls along rough ground and comes to a stop, friction between the wheels and the ground transfers energy to the surroundings. The wheels, axles, and ground become slightly warmer. There may also be sound from the wheels rattling. So a decrease in kinetic energy happens together with increases in thermal energy and sound energy.
Collisions are another clear example. When a soccer ball hits a wall, its kinetic energy changes. Some energy remains as motion if the ball bounces back. Some becomes sound. Some becomes thermal energy in the ball, wall, and air. Some goes into changing the ball's shape for a moment as it squashes and springs back.
The same idea explains brakes on a car or bike. Brakes press against a moving wheel, creating friction. The vehicle's kinetic energy decreases, and the brake pads and wheels heat up. This is why brakes can become very hot after repeated stopping. Later, that thermal energy spreads into the surrounding air.
Example: A moving bike slows down
A bicycle rider stops by squeezing the brakes. Where does the bike's kinetic energy go?
Step 1: Identify the energy that decreases.
The bike's kinetic energy decreases because its speed decreases.
Step 2: Identify the cause of the change.
Friction between the brake pads and wheels transfers energy.
Step 3: Track where the energy goes.
Most of the energy becomes thermal energy in the brakes and wheels. A little becomes sound.
The important idea is that the bike's lost kinetic energy appears as other energy changes at the same time.
Even objects that speed up follow the same rule. If a roller coaster speeds down a hill, its kinetic energy increases, but another form of energy decreases. Usually that is gravitational potential energy due to its height. If a toy car speeds up because of a battery-powered motor, the battery's chemical energy decreases while the car's kinetic energy increases, along with some thermal energy in the motor.
So whenever you notice a change in speed, ask: What other energy changed too? That question is one of the best tools in physical science. It works for balls, cars, runners, machines, and planets.
Thermal energy is the total energy associated with the motion and arrangement of particles in matter. Temperature tells us about the average kinetic energy of those particles. These ideas are related, but they are not exactly the same.
A bathtub of warm water contains more thermal energy than a cup of hot water, even if the cup has a higher temperature. Why? Because the bathtub contains much more matter. There are far more water particles, so the total energy associated with them can be greater.
You already know that matter is made of tiny particles that are always moving. In solids, particles mainly vibrate in place. In liquids, they move past one another. In gases, they move more freely and spread out. Temperature connects to how energetic that particle motion is on average.
When something is heated, energy is transferred into it. Often the particles move faster on average, so the temperature rises. When something cools, energy is transferred out of it, and the temperature falls. But temperature change is not always the same for every material or every amount of matter.
For example, a metal spoon in hot soup quickly becomes hot to the touch. A similar-sized wooden spoon does not heat up as quickly. This does not mean the soup gives "more heat" to metal; it means metal transfers energy more readily than wood. Different materials transfer and store energy differently.
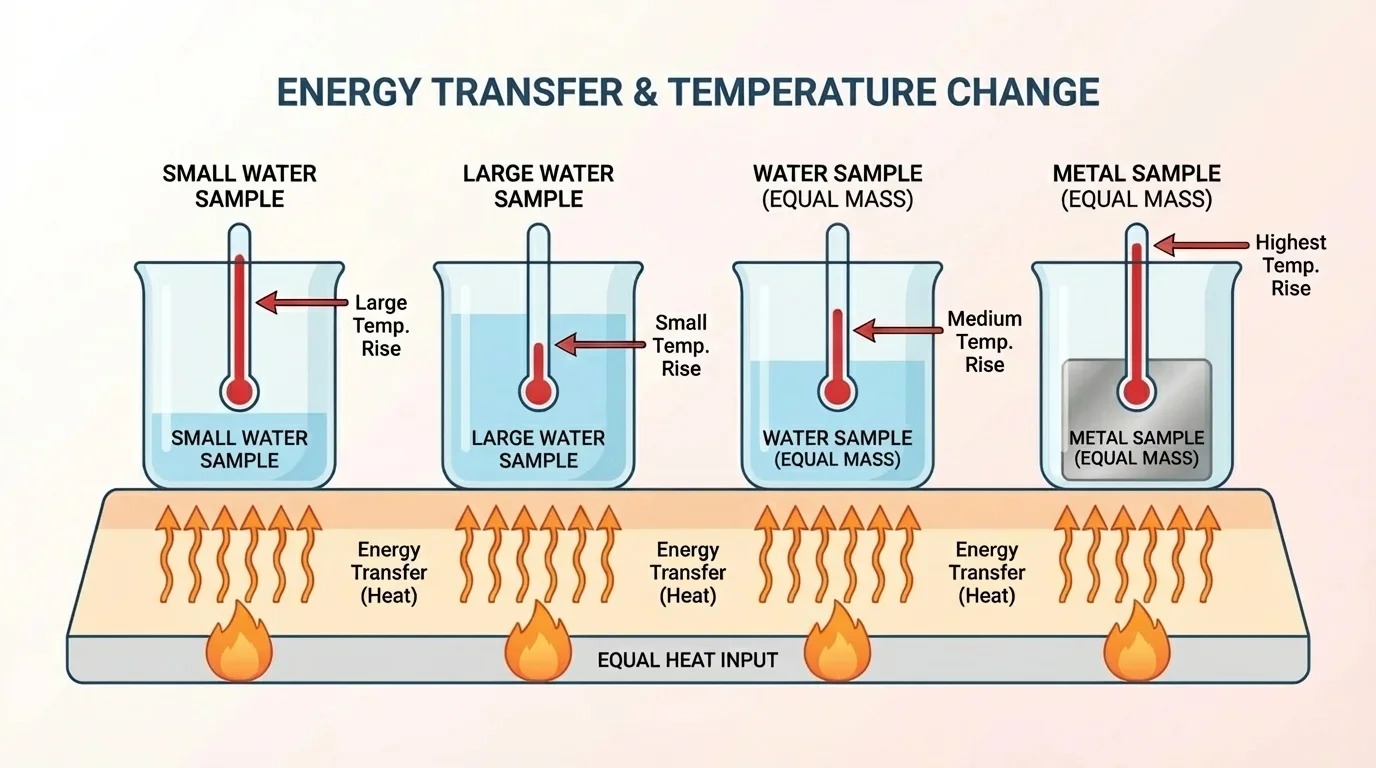
[Figure 2] helps show that the amount of energy transfer needed to change the temperature of a sample depends on several things. The most important are the nature of the material, the size of the sample, and the environment.
First, the size of the sample matters. If you give the same amount of energy to a small cup of water and to a large bucket of water, the cup's temperature will usually rise more. The same energy is spread through less matter, so the change is greater.
Second, the nature of the material matters. Different substances respond differently to energy transfer. Water takes a relatively large amount of energy to change temperature. Many metals need less energy for the same temperature change. That is one reason metal pans heat up quickly, while water in the pan warms more slowly.
Third, the environment matters. If you try to warm a cup of water outside on a cold, windy day, some energy escapes to the air while you are heating it. In a warmer room with less airflow, more of the transferred energy stays in the water. So the same heater used for the same length of time may not produce the same temperature change in different surroundings.
Think about two identical mugs of hot chocolate. One is placed in a warm room, and the other is placed outdoors on a cold day. The mug outside cools faster because energy is being transferred to the colder environment more quickly. Wind can increase the transfer even more by moving warmed air away from the mug.
Why equal energy does not always mean equal temperature change
If two samples receive the same energy transfer, they may warm by different amounts because one sample might have more mass, or it might be made of a material that needs more energy per gram for each degree of temperature increase. Energy transfer and temperature change are connected, but they are not in a simple one-size-fits-all relationship.
Scientists use a relationship for temperature change that includes mass and the type of material:
\[Q = mc\Delta T\]
In this equation, \(Q\) is the energy transferred as heating, \(m\) is mass, \(c\) is a value that depends on the material, and \(\Delta T\) is the change in temperature. For middle school science, the most important idea is not memorizing the formula but understanding what it means: larger samples and different materials need different amounts of energy for the same temperature change.
Example: Comparing two samples
Suppose sample A and sample B are made of the same material. Sample A has twice the mass of sample B. If both are warmed by the same temperature change, how much energy is needed?
Step 1: Use the relationship \(Q = mc\Delta T\).
Step 2: Notice what stays the same.
Both samples have the same material, so \(c\) is the same. They also have the same \(\Delta T\).
Step 3: Compare the masses.
If sample A has twice the mass, then its \(m\) value is twice as large, so the energy needed is also twice as large.
So the larger sample needs twice as much energy for the same temperature increase.
Here is a simple number example. If \(m = 100 \textrm{ g}\), \(c = 4\), and \(\Delta T = 5\), then \(Q = 100 \cdot 4 \cdot 5 = 2{,}000\). If the mass doubles to \(200 \textrm{ g}\) while the other values stay the same, then \(Q = 200 \cdot 4 \cdot 5 = 4{,}000\). Doubling the sample size doubles the required energy transfer.
Specific heat is the amount of energy needed to raise the temperature of a certain amount of a substance by a certain amount. You do not need to memorize units here to understand the big idea: some materials are harder to warm up than others.
Water has a high specific heat compared with many common substances. That means it takes a lot of energy to raise water's temperature. This has major effects on Earth. Large bodies of water warm and cool more slowly than land, which helps reduce extreme temperature changes near oceans and lakes.
| Material | Temperature change with same energy input | General idea |
|---|---|---|
| Water | Smaller | Needs more energy to warm |
| Metal | Larger | Needs less energy to warm |
| Large sample of any material | Smaller | Energy is spread through more matter |
| Small sample of same material | Larger | Energy is spread through less matter |
Table 1. Comparison of how sample size and material type affect temperature change for the same energy input.
A pan on a stove shows this clearly. The metal pan can become hot quickly, while the water inside takes longer to heat. Later, the hot pan can transfer energy into the water. When the water finally reaches its boiling point, added energy goes into changing state as well as particle motion, which is another reminder that energy can cause different kinds of changes.
Coastal places often have milder temperatures than inland places partly because large amounts of water warm up and cool down slowly. Water's high specific heat affects climate on a huge scale.
The idea from [Figure 2] also explains why a small metal object can become very hot very quickly, while a larger amount of water may change temperature only a little under the same heating conditions.
[Figure 3] shows one of the most important patterns in nature: energy is spontaneously transferred from hotter regions or objects to colder ones. This happens without needing someone to force it in that direction. A hot cup of tea cools down in a room. An ice cube in warm juice melts. Your warm hand can heat a cold snowball, but the snowball does not make your hand warmer by sending "coldness" into it.
Energy transfer from hotter to colder happens in three common ways: conduction, convection, and radiation.
Conduction is energy transfer by direct contact of particles. If you touch a cold metal railing, energy leaves your warmer hand and moves into the cooler railing. Metals are especially good conductors, which is why metal objects often feel colder than wood in the same room temperature: metal pulls energy from your hand more quickly.
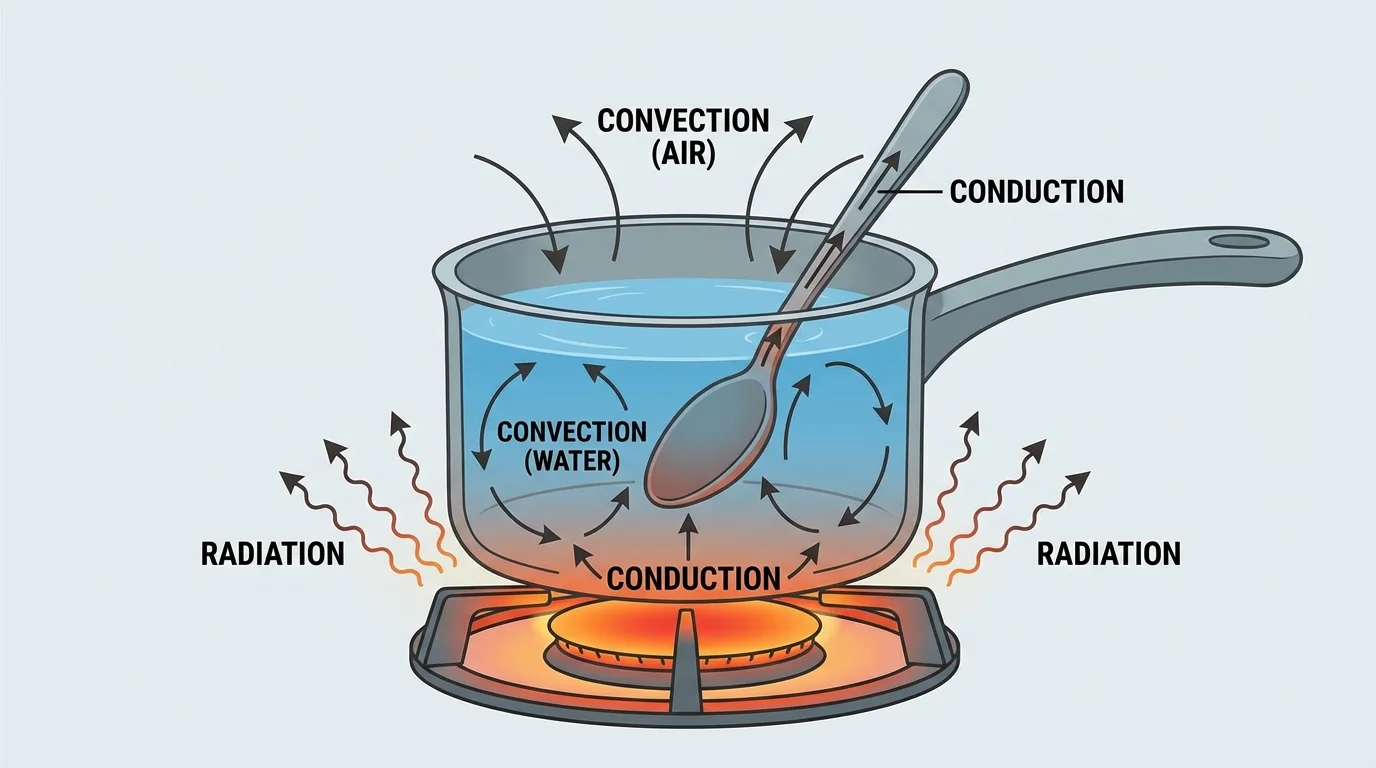
Convection happens in liquids and gases when warmer, less dense material moves and cooler, denser material sinks or moves in to replace it. In a pot of soup, the hotter liquid near the bottom rises while cooler liquid moves down. This circulation spreads energy through the liquid.
Radiation is energy transfer by electromagnetic waves. The Sun warms Earth through radiation across space. A campfire can warm your skin even if the air between you and the fire is not touched directly. Unlike conduction and convection, radiation does not require matter between the source and the receiver.
Example: Why an ice cube melts in warm water
Step 1: Compare temperatures.
The water is hotter than the ice cube.
Step 2: Determine the direction of energy transfer.
Energy moves from the warmer water to the colder ice.
Step 3: Describe the result.
The ice gains energy and melts. The water loses energy and cools slightly.
The transfer continues until the system moves toward the same temperature throughout.
This natural direction of transfer helps explain why refrigerators need energy from electricity. Energy does not spontaneously move from a colder place to a hotter one in everyday conditions. A refrigerator uses electrical energy to force energy out of the cold interior and release it into the warmer room.
The same direction rule appears again and again. Hot asphalt transfers energy to cooler night air. A freshly baked potato cools on a plate. Your body transfers energy to cold air in winter, which is why you wear layers to slow that transfer.
[Figure 4] connects these ideas to many technologies and daily experiences. In sports, friction between shoes and the ground helps athletes push off, but it also transfers energy and can warm surfaces slightly. In vehicles, engines transform chemical energy in fuel into kinetic energy, sound, and large amounts of thermal energy.
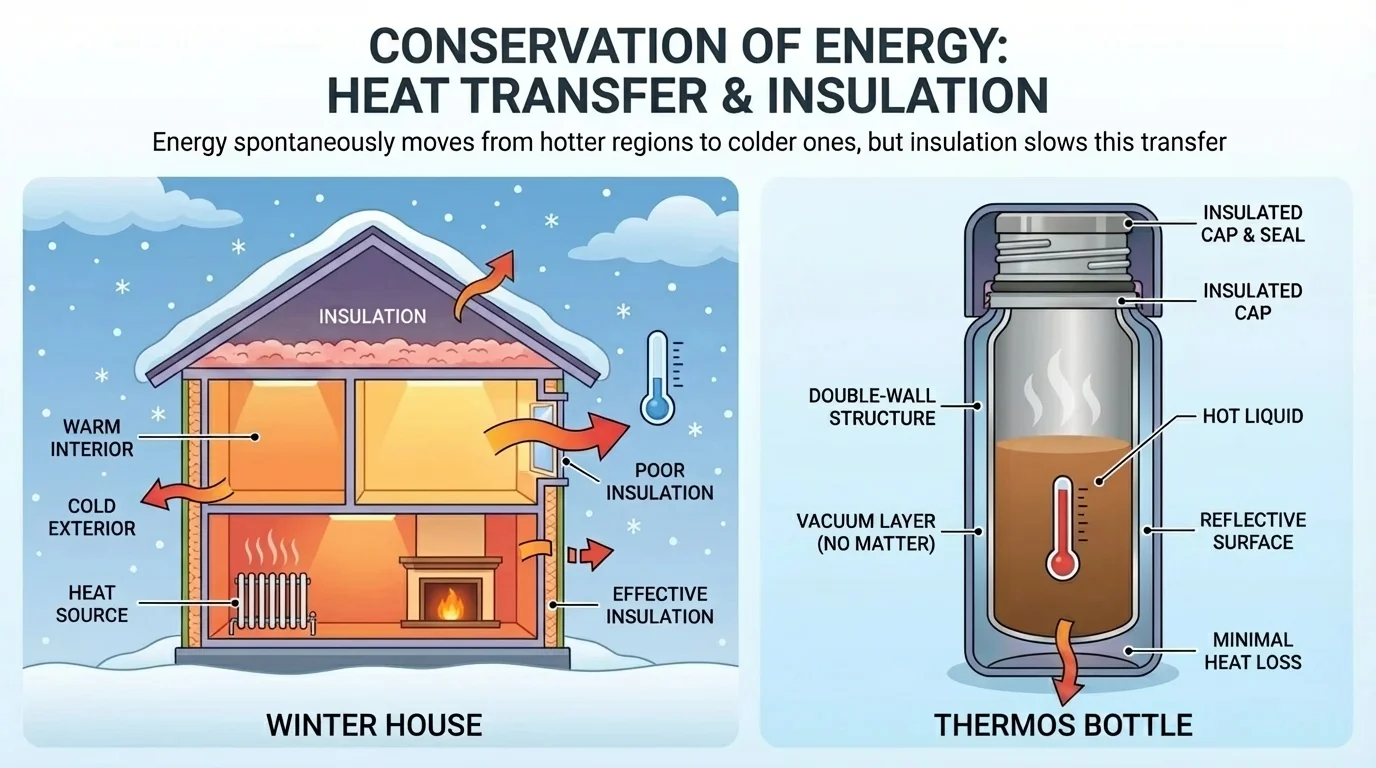
Insulation in buildings works by slowing energy transfer. In winter, a heated house is warmer than the outdoor air, so energy naturally flows outward. Good insulation, sealed windows, and thick clothing reduce the rate of transfer. They do not stop the direction of transfer, but they slow it.
A thermos bottle uses the same idea. It reduces conduction, convection, and radiation so hot drinks stay hot longer and cold drinks stay cold longer. The drink still tends to move toward room temperature, but the container slows that process.
Electronics also depend on careful energy transfer. A computer processor transforms electrical energy while working, and some of that energy becomes thermal energy. Cooling fans and heat sinks help move that thermal energy away so the device does not overheat. The processor is hotter than the surrounding air, so energy naturally flows outward, just as the rule predicts.
Weather and climate are affected too. Land warms and cools faster than water, partly because of differences in specific heat. That is one reason sea breezes form and why coastal climates are often less extreme. The pattern from [Figure 3] appears in the atmosphere as warm air rises and cooler air moves in.
Cooking provides everyday evidence. A metal pot conducts energy from the stove, convection currents move energy through boiling water, and radiation from an oven heats food. Several transfer methods often work together at the same time.
Place one metal spoon and one wooden spoon in the same bowl of hot water for a few minutes. Touch the handles carefully. The metal spoon usually feels warmer. This does not mean the metal "contains more heat". It means energy is transferred through the metal more effectively to your hand.
Another safe investigation is to compare how quickly equal amounts of warm water cool in different cups, such as a foam cup and a metal cup. Try to keep the starting temperature and amount of water the same. Over time, the cups often show different cooling rates because the environment and the cup material affect energy transfer.
What these investigations reveal
Energy transfer depends on more than just starting temperature. Material type, sample size, and contact with the environment all matter. These simple observations connect directly to the larger scientific ideas of conservation of energy and the natural flow of energy from hotter to colder places.
If you collect temperature readings, you may notice that the changes are faster at first and slower later. That happens because the temperature difference between the object and its surroundings becomes smaller over time, so the rate of energy transfer often decreases.
One common misunderstanding is saying that motion energy "turns into nothing" when an object stops. It does not. As we saw earlier with the skateboarder in [Figure 1], decreases in motion energy are matched by increases in other forms, often thermal energy and sound.
Another misunderstanding is thinking that "cold" flows from one object to another. In middle school science, it is better to describe this as energy transferring from the warmer object to the cooler one. When you hold an ice cube, energy moves from your hand into the ice, not cold moving into your hand.
A third misunderstanding is assuming that all objects heat up equally if they receive the same amount of energy. They do not. The result depends on mass, material, and surroundings. That is why the comparison shown earlier in [Figure 2] matters so much.
Once you begin tracking where energy comes from, where it goes, and what affects the rate of transfer, many everyday events make much more sense. A slowing ball, a steaming bowl of soup, a cooling room, and a warming sidewalk all follow the same connected set of principles.